+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of R2 with 5'ORF | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | R2 complex / RNA BINDING PROTEIN/RNA / RNA BINDING PROTEIN-RNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.47 Å | |||||||||
 Authors Authors | Deng P / Tan S / Wang J / Liu JJ | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Cell / Year: 2023 Journal: Cell / Year: 2023Title: Structural RNA components supervise the sequential DNA cleavage in R2 retrotransposon. Authors: Pujuan Deng / Shun-Qing Tan / Qi-Yu Yang / Liangzheng Fu / Yachao Wu / Han-Zhou Zhu / Lei Sun / Zhangbin Bao / Yi Lin / Qiangfeng Cliff Zhang / Haoyi Wang / Jia Wang / Jun-Jie Gogo Liu /  Abstract: Retroelements are the widespread jumping elements considered as major drivers for genome evolution, which can also be repurposed as gene-editing tools. Here, we determine the cryo-EM structures of ...Retroelements are the widespread jumping elements considered as major drivers for genome evolution, which can also be repurposed as gene-editing tools. Here, we determine the cryo-EM structures of eukaryotic R2 retrotransposon with ribosomal DNA target and regulatory RNAs. Combined with biochemical and sequencing analysis, we reveal two essential DNA regions, Drr and Dcr, required for recognition and cleavage. The association of 3' regulatory RNA with R2 protein accelerates the first-strand cleavage, blocks the second-strand cleavage, and initiates the reverse transcription starting from the 3'-tail. Removing 3' regulatory RNA by reverse transcription allows the association of 5' regulatory RNA and initiates the second-strand cleavage. Taken together, our work explains the DNA recognition and RNA supervised sequential retrotransposition mechanisms by R2 machinery, providing insights into the retrotransposon and application reprogramming. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_35349.map.gz emd_35349.map.gz | 14.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-35349-v30.xml emd-35349-v30.xml emd-35349.xml emd-35349.xml | 17.6 KB 17.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_35349.png emd_35349.png | 94.7 KB | ||
| Others |  emd_35349_half_map_1.map.gz emd_35349_half_map_1.map.gz emd_35349_half_map_2.map.gz emd_35349_half_map_2.map.gz | 14.5 MB 14.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35349 http://ftp.pdbj.org/pub/emdb/structures/EMD-35349 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35349 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35349 | HTTPS FTP |
-Related structure data
| Related structure data |  8ibyMC  8ibwC  8ibxC  8ibzC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_35349.map.gz / Format: CCP4 / Size: 15.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_35349.map.gz / Format: CCP4 / Size: 15.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0979 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_35349_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
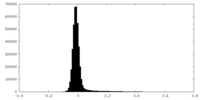
| Density Histograms |
-Half map: #1
| File | emd_35349_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
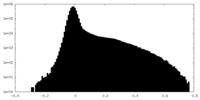
| Density Histograms |
- Sample components
Sample components
-Entire : R2 complex
| Entire | Name: R2 complex |
|---|---|
| Components |
|
-Supramolecule #1: R2 complex
| Supramolecule | Name: R2 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 177.28 KDa |
-Macromolecule #1: 5'ORF RNA
| Macromolecule | Name: 5'ORF RNA / type: rna / ID: 1 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 72.943172 KDa |
| Sequence | String: UGUACACGUG GUAAACACGU GACAGCAGCC CCGAUGGACG GACCGCGAGG ACCGUCAAGC CUAGCAGGUA CCUUCGGGUG GGGCCUUGC GAUACCUGCG GGCGAACCCU GUGGUCGGGU UUGCAGCCCG GCCACAGUGG GUUUUUUUCC UGUUGCAAAA A AGUCAAAU ...String: UGUACACGUG GUAAACACGU GACAGCAGCC CCGAUGGACG GACCGCGAGG ACCGUCAAGC CUAGCAGGUA CCUUCGGGUG GGGCCUUGC GAUACCUGCG GGCGAACCCU GUGGUCGGGU UUGCAGCCCG GCCACAGUGG GUUUUUUUCC UGUUGCAAAA A AGUCAAAU AAAGAAAAUA GACCUACUGU GCGGGGUUCC GCCGGCGCUU GGACCUGCCA GUUCUGCG |
-Macromolecule #2: Reverse transcriptase-like protein
| Macromolecule | Name: Reverse transcriptase-like protein / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 123.36243 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MMASTALSLM GRCNPDGCTR GKHVTAAPMD GPRGPSSLAG TFGWGLAIPA GEPCGRVCSP ATVGFFPVAK KSNKENRPEA SGLPLESER TGDNPTVRGS AGADPVGQDA PGWTCQFCER TFSTNRGLGV HKRRAHPVET NTDAAPMMVK RRWHGEEIDL L ARTEARLL ...String: MMASTALSLM GRCNPDGCTR GKHVTAAPMD GPRGPSSLAG TFGWGLAIPA GEPCGRVCSP ATVGFFPVAK KSNKENRPEA SGLPLESER TGDNPTVRGS AGADPVGQDA PGWTCQFCER TFSTNRGLGV HKRRAHPVET NTDAAPMMVK RRWHGEEIDL L ARTEARLL AERGQCSGGD LFGALPGFGR TLEAIKGQRR REPYRALVQA HLARFGSQPG PSSGGCSAEP DFRRASGAEE AG EERCAED AAAYDPSAVG QMSPDAARVL SELLEGAGRR RACRAMRPKT AGRRNDLHDD RTASAHKTSR QKRRAEYARV QEL YKKCRS RAAAEVIDGA CGGVGHSLEE METYWRPILE RVSDAPGPTP EALHALGRAE WHGGNRDYTQ LWKPISVEEI KASR FDWRT SPGPDGIRSG QWRAVPVHLK AEMFNAWMAR GEIPEILRQC RTVFVPKVER PGGPGEYRPI SIASIPLRHF HSILA RRLL ACCPPDARQR GFICADGTLE NSAVLDAVLG DSRKKLRECH VAVLDFAKAF DTVSHEALVE LLRLRGMPEQ FCGYIA HLY DTASTTLAVN NEMSSPVKVG RGVRQGDPLS PILFNVVMDL ILASLPERVG YRLEMELVSA LAYAYDLVLL AGSKVGM QE SISAVDCVGR QMGLRLNCRK SAVLSMIPDG HRKKHHYLTE RTFNIGGKPL RQVSCVERWR YLGVDFEASG CVTLEHSI S SALNNISRAP LKPQQRLEIL RAHLIPRFQH GFVLGNISDD RLRMLDVQIR KAVGQWLRLP ADVPKAYYHA AVQDGGLAI PSVRATIPDL IVRRFGGLDS SPWSVARAAA KSDKIRKKLR WAWKQLRRFS RVDSTTQRPS VRLFWREHLH ASVDGRELRE STRTPTSTK WIRERCAQIT GRDFVQFVHT HINALPSRIR GSRGRRGGGE SSLTCRAGCK VRETTAHILQ QCHRTHGGRI L RHNKIVSF VAKAMEENKW TVELEPRLRT SVGLRKPAII ASRDGVGVIV DVQVVSGQRS LDELHREKRN KYGNHGELVE LV AGRLGLP KAECVRATSC TISWRGVWSL TSYKELRSII GLREPTLQIV PILALRGSHM NWTRFNQMTS VMGGGVG UniProtKB: Reverse transcriptase-like protein |
-Macromolecule #3: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 3 / Number of copies: 1 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 400 / Support film - Material: GRAPHENE OXIDE / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 0.01 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 64000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: BACKBONE TRACE |
|---|---|
| Output model |  PDB-8iby: |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)