+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the ISFba1 TnpB-reRNA-dsDNA complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | RNA / Complex / RNA BINDING PROTEIN/RNA / RNA BINDING PROTEIN-RNA-DNA complex | |||||||||
| Function / homology | :  Function and homology information Function and homology information | |||||||||
| Biological species |  Firmicutes bacterium AM43-11BH (bacteria) Firmicutes bacterium AM43-11BH (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.0 Å | |||||||||
 Authors Authors | Yin M / Zhou F / Zhu Y / Huang Z | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Res / Year: 2024 Journal: Cell Res / Year: 2024Title: Discovery and structural mechanism of DNA endonucleases guided by RAGATH-18-derived RNAs. Authors: Kuan Ren / Fengxia Zhou / Fan Zhang / Mingyu Yin / Yuwei Zhu / Shouyu Wang / Yan Chen / Tengjin Huang / Zixuan Wu / Jiale He / Anqi Zhang / Changyou Guo / Zhiwei Huang /  Abstract: CRISPR-Cas systems and IS200/IS605 transposon-associated TnpBs have been utilized for the development of genome editing technologies. Using bioinformatics analysis and biochemical experiments, here ...CRISPR-Cas systems and IS200/IS605 transposon-associated TnpBs have been utilized for the development of genome editing technologies. Using bioinformatics analysis and biochemical experiments, here we present a new family of RNA-guided DNA endonucleases. Our bioinformatics analysis initially identifies the stable co-occurrence of conserved RAGATH-18-derived RNAs (reRNAs) and their upstream IS607 TnpBs with an average length of 390 amino acids. IS607 TnpBs form programmable DNases through interaction with reRNAs. We discover the robust dsDNA interference activity of IS607 TnpB systems in bacteria and human cells. Further characterization of the Firmicutes bacteria IS607 TnpB system (ISFba1 TnpB) reveals that its dsDNA cleavage activity is remarkably sensitive to single mismatches between the guide and target sequences in human cells. Our findings demonstrate that a length of 20 nt in the guide sequence of reRNA achieves the highest DNA cleavage activity for ISFba1 TnpB. A cryo-EM structure of the ISFba1 TnpB effector protein bound by its cognate RAGATH-18 motif-containing reRNA and a dsDNA target reveals the mechanisms underlying reRNA recognition by ISFba1 TnpB, reRNA-guided dsDNA targeting, and the sensitivity of the ISFba1 TnpB system to base mismatches between the guide and target DNA. Collectively, this study identifies the IS607 TnpB family of compact and specific RNA-guided DNases with great potential for application in gene editing. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_35323.map.gz emd_35323.map.gz | 59.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-35323-v30.xml emd-35323-v30.xml emd-35323.xml emd-35323.xml | 16.4 KB 16.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_35323.png emd_35323.png | 77.9 KB | ||
| Filedesc metadata |  emd-35323.cif.gz emd-35323.cif.gz | 5.9 KB | ||
| Others |  emd_35323_half_map_1.map.gz emd_35323_half_map_1.map.gz emd_35323_half_map_2.map.gz emd_35323_half_map_2.map.gz | 49.6 MB 49.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35323 http://ftp.pdbj.org/pub/emdb/structures/EMD-35323 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35323 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35323 | HTTPS FTP |
-Validation report
| Summary document |  emd_35323_validation.pdf.gz emd_35323_validation.pdf.gz | 822.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_35323_full_validation.pdf.gz emd_35323_full_validation.pdf.gz | 822.3 KB | Display | |
| Data in XML |  emd_35323_validation.xml.gz emd_35323_validation.xml.gz | 12.2 KB | Display | |
| Data in CIF |  emd_35323_validation.cif.gz emd_35323_validation.cif.gz | 14.3 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35323 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35323 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35323 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35323 | HTTPS FTP |
-Related structure data
| Related structure data |  8iazMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_35323.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_35323.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.86 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_35323_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
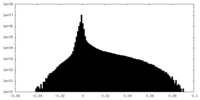
| Projections & Slices |
| ||||||||||||
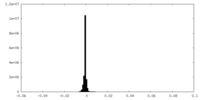
| Density Histograms |
-Half map: #2
| File | emd_35323_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
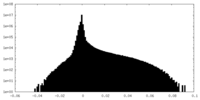
| Projections & Slices |
| ||||||||||||
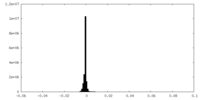
| Density Histograms |
- Sample components
Sample components
-Entire : The complex of protein with gRNA and target DNA
| Entire | Name: The complex of protein with gRNA and target DNA |
|---|---|
| Components |
|
-Supramolecule #1: The complex of protein with gRNA and target DNA
| Supramolecule | Name: The complex of protein with gRNA and target DNA / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:  Firmicutes bacterium AM43-11BH (bacteria) Firmicutes bacterium AM43-11BH (bacteria) |
-Macromolecule #1: Transposase
| Macromolecule | Name: Transposase / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Firmicutes bacterium AM43-11BH (bacteria) Firmicutes bacterium AM43-11BH (bacteria) |
| Molecular weight | Theoretical: 44.549645 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: LLKSFKTEIN PSEEQKVKIH KTIGTCRFIY NFYLAHNKEL YDKGEKFMSG KSFSVWLNNE YLPQNPDKLW IKEVSSKSVK HSIENGCIA FTRFFKHQSA FPNLKKKGKS DVKMYFVKNN PKDCRCERHR INIPSLGWVR IKEKGYIPTT KDGYVIKSGT V SMKADRYY ...String: LLKSFKTEIN PSEEQKVKIH KTIGTCRFIY NFYLAHNKEL YDKGEKFMSG KSFSVWLNNE YLPQNPDKLW IKEVSSKSVK HSIENGCIA FTRFFKHQSA FPNLKKKGKS DVKMYFVKNN PKDCRCERHR INIPSLGWVR IKEKGYIPTT KDGYVIKSGT V SMKADRYY VSVLVEISNN KIANNSNAGI GIDLGLKDFA IVSNGKTYKN INKSARLKKH EKQLIREQRS LSRKYENLKK GE STQKANI QKQRLKVQKL HHRMDNIRTD YINKTIAEIV KTKPSYITIE DLNVKGMMKN RHLSKAVASQ KFYEFRTKLQ AKC NENGIE LRVVDRWYPS SKTCHCCGAV KKDLKLSDRI FKCSCGYVED RAFNAALNLR DAITYEVA UniProtKB: UNIPROTKB: A0A417B524 |
-Macromolecule #2: RNA (207-MER)
| Macromolecule | Name: RNA (207-MER) / type: rna / ID: 2 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  Firmicutes bacterium AM43-11BH (bacteria) Firmicutes bacterium AM43-11BH (bacteria) |
| Molecular weight | Theoretical: 66.616352 KDa |
| Sequence | String: CUAAAACGCA AACGUAAGUA UGUACCGAAG GCUAUUUUCG GGAAUUUACG ACUAUGGAGU GUACAAGAAC UUGUGAGUAG AUAUGAUUU CGGUCAAUCC AAAGCAUACA CGAUGAAAUA GUAAGUAAUG UUCGUGAGAA CUUACAUUAU CUCGAUGUGA G CAUAUUUA ...String: CUAAAACGCA AACGUAAGUA UGUACCGAAG GCUAUUUUCG GGAAUUUACG ACUAUGGAGU GUACAAGAAC UUGUGAGUAG AUAUGAUUU CGGUCAAUCC AAAGCAUACA CGAUGAAAUA GUAAGUAAUG UUCGUGAGAA CUUACAUUAU CUCGAUGUGA G CAUAUUUA AUCACAUUUU GAGUGGCAGC UGAUGGUCCA UGUCUGUUA |
-Macromolecule #3: DNA (5'-D(P*AP*CP*AP*TP*GP*GP*AP*CP*CP*AP*TP*CP*AP*GP*CP*TP*CP*CP...
| Macromolecule | Name: DNA (5'-D(P*AP*CP*AP*TP*GP*GP*AP*CP*CP*AP*TP*CP*AP*GP*CP*TP*CP*CP*TP*AP*AP*TP*GP*G)-3') type: dna / ID: 3 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Firmicutes bacterium AM43-11BH (bacteria) Firmicutes bacterium AM43-11BH (bacteria) |
| Molecular weight | Theoretical: 7.338756 KDa |
| Sequence | String: (DA)(DC)(DA)(DT)(DG)(DG)(DA)(DC)(DC)(DA) (DT)(DC)(DA)(DG)(DC)(DT)(DC)(DC)(DT)(DA) (DA)(DT)(DG)(DG) |
-Macromolecule #4: DNA (5'-D(P*CP*CP*AP*TP*TP*AP*GP*GP*AP*GP*CP*TP*GP*AP*TP*G)-3')
| Macromolecule | Name: DNA (5'-D(P*CP*CP*AP*TP*TP*AP*GP*GP*AP*GP*CP*TP*GP*AP*TP*G)-3') type: dna / ID: 4 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Firmicutes bacterium AM43-11BH (bacteria) Firmicutes bacterium AM43-11BH (bacteria) |
| Molecular weight | Theoretical: 4.938216 KDa |
| Sequence | String: (DC)(DC)(DA)(DT)(DT)(DA)(DG)(DG)(DA)(DG) (DC)(DT)(DG)(DA)(DT)(DG) |
-Macromolecule #5: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 5 / Number of copies: 1 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.2 |
|---|---|
| Vitrification | Cryogen name: NITROGEN |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.5 µm / Nominal defocus min: 1.2 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.0 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 369379 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller




 Z
Z Y
Y X
X