[English] 日本語
 Yorodumi
Yorodumi- EMDB-34318: A mechanism for SARS-CoV-2 RNA capping and its inhibition by nucl... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | A mechanism for SARS-CoV-2 RNA capping and its inhibition by nucleotide analogue inhibitors | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | SARS-CoV-2 / capping / nucleotide analogue inhibitor / cryo-EM / VIRAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationviral genome replication / methyltransferase activity / protein guanylyltransferase activity / RNA endonuclease activity, producing 3'-phosphomonoesters / mRNA guanylyltransferase activity / 5'-3' RNA helicase activity / Lyases; Phosphorus-oxygen lyases / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of TBK1 activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / Maturation of replicase proteins ...viral genome replication / methyltransferase activity / protein guanylyltransferase activity / RNA endonuclease activity, producing 3'-phosphomonoesters / mRNA guanylyltransferase activity / 5'-3' RNA helicase activity / Lyases; Phosphorus-oxygen lyases / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of TBK1 activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs / TRAF3-dependent IRF activation pathway / Translation of Replicase and Assembly of the Replication Transcription Complex / Replication of the SARS-CoV-2 genome / snRNP Assembly / double membrane vesicle viral factory outer membrane / Hydrolases; Acting on ester bonds; Exoribonucleases producing 5'-phosphomonoesters / SARS coronavirus main proteinase / host cell endoplasmic reticulum-Golgi intermediate compartment / 3'-5'-RNA exonuclease activity / 5'-3' DNA helicase activity / symbiont-mediated suppression of host NF-kappaB cascade / host cell endosome / symbiont-mediated suppression of host toll-like receptor signaling pathway / symbiont-mediated degradation of host mRNA / mRNA guanylyltransferase / symbiont-mediated suppression of host ISG15-protein conjugation / G-quadruplex RNA binding / mRNA (guanine-N7)-methyltransferase / omega peptidase activity / methyltransferase cap1 / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of IRF3 activity / SARS-CoV-2 modulates host translation machinery / host cell Golgi apparatus / symbiont-mediated perturbation of host ubiquitin-like protein modification / endonuclease activity / DNA helicase / mRNA (nucleoside-2'-O-)-methyltransferase activity / mRNA 5'-cap (guanine-N7-)-methyltransferase activity / ubiquitinyl hydrolase 1 / methylation / cysteine-type deubiquitinase activity / Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases / host cell perinuclear region of cytoplasm / single-stranded RNA binding / host cell endoplasmic reticulum membrane / viral protein processing / lyase activity / RNA helicase / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / induction by virus of host autophagy / copper ion binding / RNA-directed RNA polymerase / viral translational frameshifting / viral RNA genome replication / cysteine-type endopeptidase activity / RNA-dependent RNA polymerase activity / virus-mediated perturbation of host defense response / lipid binding / DNA-templated transcription / host cell nucleus / SARS-CoV-2 activates/modulates innate and adaptive immune responses / ATP hydrolysis activity / proteolysis / RNA binding / zinc ion binding / ATP binding / membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Yan LM / Huang YC / Ge J / Liu ZY / Gao Y / Rao ZH / Lou ZY | |||||||||
| Funding support |  China, 2 items China, 2 items
| |||||||||
 Citation Citation |  Journal: Cell / Year: 2022 Journal: Cell / Year: 2022Title: A mechanism for SARS-CoV-2 RNA capping and its inhibition by nucleotide analog inhibitors. Authors: Liming Yan / Yucen Huang / Ji Ge / Zhenyu Liu / Pengchi Lu / Bo Huang / Shan Gao / Junbo Wang / Liping Tan / Sihan Ye / Fengxi Yu / Weiqi Lan / Shiya Xu / Feng Zhou / Lei Shi / Luke W Guddat ...Authors: Liming Yan / Yucen Huang / Ji Ge / Zhenyu Liu / Pengchi Lu / Bo Huang / Shan Gao / Junbo Wang / Liping Tan / Sihan Ye / Fengxi Yu / Weiqi Lan / Shiya Xu / Feng Zhou / Lei Shi / Luke W Guddat / Yan Gao / Zihe Rao / Zhiyong Lou /    Abstract: Decoration of cap on viral RNA plays essential roles in SARS-CoV-2 proliferation. Here, we report a mechanism for SARS-CoV-2 RNA capping and document structural details at atomic resolution. The ...Decoration of cap on viral RNA plays essential roles in SARS-CoV-2 proliferation. Here, we report a mechanism for SARS-CoV-2 RNA capping and document structural details at atomic resolution. The NiRAN domain in polymerase catalyzes the covalent link of RNA 5' end to the first residue of nsp9 (termed as RNAylation), thus being an intermediate to form cap core (GpppA) with GTP catalyzed again by NiRAN. We also reveal that triphosphorylated nucleotide analog inhibitors can be bonded to nsp9 and fit into a previously unknown "Nuc-pocket" in NiRAN, thus inhibiting nsp9 RNAylation and formation of GpppA. S-loop (residues 50-KTN-52) in NiRAN presents a remarkable conformational shift observed in RTC bound with sofosbuvir monophosphate, reasoning an "induce-and-lock" mechanism to design inhibitors. These findings not only improve the understanding of SARS-CoV-2 RNA capping and the mode of action of NAIs but also provide a strategy to design antiviral drugs. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_34318.map.gz emd_34318.map.gz | 483.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-34318-v30.xml emd-34318-v30.xml emd-34318.xml emd-34318.xml | 23.9 KB 23.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_34318.png emd_34318.png | 27.1 KB | ||
| Filedesc metadata |  emd-34318.cif.gz emd-34318.cif.gz | 7.2 KB | ||
| Others |  emd_34318_half_map_1.map.gz emd_34318_half_map_1.map.gz emd_34318_half_map_2.map.gz emd_34318_half_map_2.map.gz | 475.6 MB 475.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34318 http://ftp.pdbj.org/pub/emdb/structures/EMD-34318 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34318 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34318 | HTTPS FTP |
-Validation report
| Summary document |  emd_34318_validation.pdf.gz emd_34318_validation.pdf.gz | 1.1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_34318_full_validation.pdf.gz emd_34318_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  emd_34318_validation.xml.gz emd_34318_validation.xml.gz | 18.8 KB | Display | |
| Data in CIF |  emd_34318_validation.cif.gz emd_34318_validation.cif.gz | 22.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34318 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34318 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34318 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34318 | HTTPS FTP |
-Related structure data
| Related structure data |  8gwoMC  8gw1C  8gwbC  8gweC  8gwfC  8gwgC  8gwiC  8gwkC  8gwmC  8gwnC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_34318.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_34318.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.82 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_34318_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
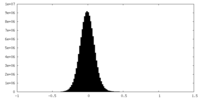
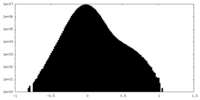
| Density Histograms |
-Half map: #2
| File | emd_34318_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
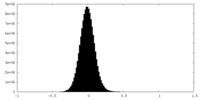
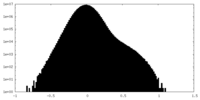
| Density Histograms |
- Sample components
Sample components
+Entire : E-RTC_UMP-nsp9_GMPPNP
+Supramolecule #1: E-RTC_UMP-nsp9_GMPPNP
+Supramolecule #2: Replicase polyprotein
+Supramolecule #3: RNA
+Macromolecule #1: RNA-directed RNA polymerase
+Macromolecule #2: Non-structural protein 8
+Macromolecule #3: Non-structural protein 7
+Macromolecule #6: Helicase
+Macromolecule #7: Non-structural protein 9
+Macromolecule #4: RNA (25-MER)
+Macromolecule #5: template
+Macromolecule #8: ZINC ION
+Macromolecule #9: PHOSPHOAMINOPHOSPHONIC ACID-GUANYLATE ESTER
+Macromolecule #10: URIDINE-5'-MONOPHOSPHATE
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.2 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.8 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 2058771 |
| Initial angle assignment | Type: NOT APPLICABLE |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)