+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cryo-EM structure of the Teriparatide-bound human PTH1R-Gs complex | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Teriparatide / PTH1R / Cryo-EM structure / MEMBRANE PROTEIN | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報macromolecule biosynthetic process / parathyroid hormone receptor binding / type 1 parathyroid hormone receptor binding / negative regulation of bone mineralization involved in bone maturation / negative regulation of apoptotic process in bone marrow cell / positive regulation of osteoclast proliferation / response to parathyroid hormone / positive regulation of cell proliferation in bone marrow / hormone-mediated apoptotic signaling pathway / adenylate cyclase-activating G protein-coupled cAMP receptor signaling pathway ...macromolecule biosynthetic process / parathyroid hormone receptor binding / type 1 parathyroid hormone receptor binding / negative regulation of bone mineralization involved in bone maturation / negative regulation of apoptotic process in bone marrow cell / positive regulation of osteoclast proliferation / response to parathyroid hormone / positive regulation of cell proliferation in bone marrow / hormone-mediated apoptotic signaling pathway / adenylate cyclase-activating G protein-coupled cAMP receptor signaling pathway / parathyroid hormone receptor activity / positive regulation of signal transduction / magnesium ion homeostasis / response to fibroblast growth factor / cAMP metabolic process / phosphate ion homeostasis / G-protein activation / Activation of the phototransduction cascade / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Ca2+ pathway / G alpha (z) signalling events / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / Class B/2 (Secretin family receptors) / Vasopressin regulates renal water homeostasis via Aquaporins / G protein-coupled peptide receptor activity / negative regulation of chondrocyte differentiation / Adrenaline,noradrenaline inhibits insulin secretion / ADP signalling through P2Y purinoceptor 12 / G alpha (q) signalling events / osteoblast development / response to vitamin D / G alpha (i) signalling events / Thrombin signalling through proteinase activated receptors (PARs) / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor / Presynaptic function of Kainate receptors / G beta:gamma signalling through CDC42 / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G alpha (12/13) signalling events / Glucagon-type ligand receptors / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Adrenaline,noradrenaline inhibits insulin secretion / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Ca2+ pathway / G alpha (z) signalling events / Thrombin signalling through proteinase activated receptors (PARs) / Extra-nuclear estrogen signaling / G alpha (s) signalling events / G alpha (q) signalling events / photoreceptor outer segment membrane / spectrin binding / G alpha (i) signalling events / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / peptide hormone receptor binding / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / Vasopressin regulates renal water homeostasis via Aquaporins / bone mineralization / alkylglycerophosphoethanolamine phosphodiesterase activity / positive regulation of inositol phosphate biosynthetic process / peptide hormone binding / positive regulation of glycogen biosynthetic process / chondrocyte differentiation / photoreceptor outer segment / positive regulation of bone mineralization / response to cadmium ion / bone resorption / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / Rho protein signal transduction / cardiac muscle cell apoptotic process / cell maturation / photoreceptor inner segment / homeostasis of number of cells within a tissue / positive regulation of D-glucose import across plasma membrane / skeletal system development / hormone activity / response to lead ion / G protein-coupled receptor activity / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / intracellular calcium ion homeostasis / cellular response to catecholamine stimulus / cell-cell signaling / adenylate cyclase-activating dopamine receptor signaling pathway / cellular response to prostaglandin E stimulus / heterotrimeric G-protein complex / G-protein beta-subunit binding / sensory perception of taste 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) / Homo sapiens (ヒト) /   | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 2.8 Å | |||||||||
 データ登録者 データ登録者 | Zhai X / Mao C / Shen Q / Zang S / Shen D / Zhang H / Chen Z / Wang G / Zhang C / Zhang Y / Liu Z | |||||||||
| 資金援助 | 1件
| |||||||||
 引用 引用 |  ジャーナル: to be published ジャーナル: to be publishedタイトル: Cryo-EM structure of the Teriparatide-bound human PTH1R-Gs complex 著者: Zhai X / Mao C / Shen Q / Zang S / Shen D / Zhang H / Chen Z / Wang G / Zhang C / Zhang Y / Liu Z | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_33590.map.gz emd_33590.map.gz | 33 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-33590-v30.xml emd-33590-v30.xml emd-33590.xml emd-33590.xml | 29.3 KB 29.3 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_33590.png emd_33590.png | 44.1 KB | ||
| Filedesc metadata |  emd-33590.cif.gz emd-33590.cif.gz | 8.2 KB | ||
| その他 |  emd_33590_additional_1.map.gz emd_33590_additional_1.map.gz emd_33590_half_map_1.map.gz emd_33590_half_map_1.map.gz emd_33590_half_map_2.map.gz emd_33590_half_map_2.map.gz | 33 MB 33.1 MB 33.1 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33590 http://ftp.pdbj.org/pub/emdb/structures/EMD-33590 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33590 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33590 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7y36MC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_33590.map.gz / 形式: CCP4 / 大きさ: 42.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_33590.map.gz / 形式: CCP4 / 大きさ: 42.9 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.014 Å | ||||||||||||||||||||||||||||||||||||
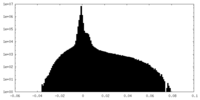
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-追加マップ: half1 map of main map
| ファイル | emd_33590_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half1 map of main map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
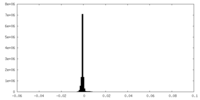
| 密度ヒストグラム |
-ハーフマップ: half1 map of main map
| ファイル | emd_33590_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half1 map of main map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
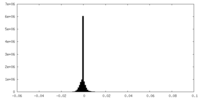
| 密度ヒストグラム |
-ハーフマップ: half2 map of main map
| ファイル | emd_33590_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half2 map of main map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
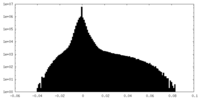
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Cryo-EM structure of the Teriparatide-bound human PTH1R-Gs complex
| 全体 | 名称: Cryo-EM structure of the Teriparatide-bound human PTH1R-Gs complex |
|---|---|
| 要素 |
|
-超分子 #1: Cryo-EM structure of the Teriparatide-bound human PTH1R-Gs complex
| 超分子 | 名称: Cryo-EM structure of the Teriparatide-bound human PTH1R-Gs complex タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
-分子 #1: Parathyroid hormone/parathyroid hormone-related peptide receptor
| 分子 | 名称: Parathyroid hormone/parathyroid hormone-related peptide receptor タイプ: protein_or_peptide / ID: 1 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 81.730609 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: DADDVMTKEE QIFLLHRAQA QCEKRLKEVL QRPASIMESD KGWTSASTSG KPRKDKASGK LYPESEEDKE APTGSRYRGR PCLPEWDHI LCWPLGAPGE VVAVPCPDYI YDFNHKGHAY RRCDRNGSWE LVPGHNRTWA NYSECVKFLT NETREREVFD R LAMIYTVG ...文字列: DADDVMTKEE QIFLLHRAQA QCEKRLKEVL QRPASIMESD KGWTSASTSG KPRKDKASGK LYPESEEDKE APTGSRYRGR PCLPEWDHI LCWPLGAPGE VVAVPCPDYI YDFNHKGHAY RRCDRNGSWE LVPGHNRTWA NYSECVKFLT NETREREVFD R LAMIYTVG YSVSLASLTV AVLILAYFRR LHCTRNYIHM HLFLSFMLRA VSIFVKDAVL YSGATLDEAE RLTEEELRAI AQ APPPPAT AAAGYAGCRV AVTFFLYFLA TNYYWILVEG LYLHSLIFMA FFSEKKYLWG FTVFGWGLPA VFVAVWVSVR ATL ANTGCW DLSSGNKKWI IQVPILASIV LNFILFINIV RVLATKLRET NAGRCDTRQQ YRKLLKSTLV LMPLFGVHYI VFMA TPYTE VSGTLWQVQM HYEMLFNSFQ GFFVAIIYCF CNGEVQAEIK KSWSRWTLAL DFKRKARSGS SSYSYGPMVS HTSVT NVGP RVGLGLPLSP RLLPTATTNG HPQLPGHAKP GTPALETLET TPPAMAAPKD DGFLNGSCSG LDEEASGPER PPALLQ EEW ETVMEFVFTL EDFVGDWEQT AAYNLDQVLE QGGVSSLLQN LAVSVTPIQR IVRSGENALK IDIHVIIPYE GLSADQM AQ IEEVFKVVYP VDDHHFKVIL PYGTLVIDGV TPNMLNYFGR PYEGIAVFDG KKITVTGTLW NGNKIIDERL ITPDGSML F RVTINS UniProtKB: Parathyroid hormone/parathyroid hormone-related peptide receptor |
-分子 #2: Parathyroid hormone
| 分子 | 名称: Parathyroid hormone / タイプ: protein_or_peptide / ID: 2 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 4.125778 KDa |
| 配列 | 文字列: SVSEIQLMHN LGKHLNSMER VEWLRKKLQD VHNF UniProtKB: Parathyroid hormone |
-分子 #3: Isoform Gnas-2 of Guanine nucleotide-binding protein G(s) subunit...
| 分子 | 名称: Isoform Gnas-2 of Guanine nucleotide-binding protein G(s) subunit alpha isoforms short タイプ: protein_or_peptide / ID: 3 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 44.257051 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKSTIVKQM RILHVNGFNG DSEKATKVQD IKNNLKEAI ETIVAAMSNL VPPVELANPE NQFRVDYILS VMNVPDFDFP PEFYEHAKAL WEDEGVRACY ERSNEYQLID C AQYFLDKI ...文字列: MGCLGNSKTE DQRNEEKAQR EANKKIEKQL QKDKQVYRAT HRLLLLGAGE SGKSTIVKQM RILHVNGFNG DSEKATKVQD IKNNLKEAI ETIVAAMSNL VPPVELANPE NQFRVDYILS VMNVPDFDFP PEFYEHAKAL WEDEGVRACY ERSNEYQLID C AQYFLDKI DVIKQADYVP SDQDLLRCRV LTSGIFETKF QVDKVNFHMF DVGAQRDERR KWIQCFNDVT AIIFVVASSS YN MVIREDN QTNRLQAALK LFDSIWNNKW LRDTSVILFL NKQDLLAEKV LAGKSKIEDY FPEFARYTTP EDATPEPGED PRV TRAKYF IRDEFLRIST ASGDGRHYCY PHFTCAVDTE NIRRVFNDCR DIIQRMHLRQ YELL UniProtKB: Isoform Gnas-2 of Guanine nucleotide-binding protein G(s) subunit alpha isoforms short |
-分子 #4: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| 分子 | 名称: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 タイプ: protein_or_peptide / ID: 4 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 41.44132 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MHHHHHHLEV LFQGPSELDQ LRQEAEQLKN QIRDARKACA DATLSQITNN IDPVGRIQMR TRRTLRGHLA KIYAMHWGTD SRLLVSASQ DGKLIIWDSY TTNKVHAIPL RSSWVMTCAY APSGNYVACG GLDNICSIYN LKTREGNVRV SRELAGHTGY L SCCRFLDD ...文字列: MHHHHHHLEV LFQGPSELDQ LRQEAEQLKN QIRDARKACA DATLSQITNN IDPVGRIQMR TRRTLRGHLA KIYAMHWGTD SRLLVSASQ DGKLIIWDSY TTNKVHAIPL RSSWVMTCAY APSGNYVACG GLDNICSIYN LKTREGNVRV SRELAGHTGY L SCCRFLDD NQIVTSSGDT TCALWDIETG QQTTTFTGHT GDVMSLSLAP DTRLFVSGAC DASAKLWDVR EGMCRQTFTG HE SDINAIC FFPNGNAFAT GSDDATCRLF DLRADQELMT YSHDNIICGI TSVSFSKSGR LLLAGYDDFN CNVWDALKAD RAG VLAGHD NRVSCLGVTD DGMAVATGSW DSFLKIWNGS SGGGGSGGGG SSGVSGWRLF KKIS UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-分子 #5: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| 分子 | 名称: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 タイプ: protein_or_peptide / ID: 5 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 7.729947 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: ASNNTASIAQ ARKLVEQLKM EANIDRIKVS KAAADLMAYC EAHAKEDPLL TPVPASENPF REKKFFCAIL UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-分子 #6: NanoBody 35
| 分子 | 名称: NanoBody 35 / タイプ: protein_or_peptide / ID: 6 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種: synthetic construct (人工物) |
| 分子量 | 理論値: 13.711284 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: QVQLQESGGG LVQPGGSLRL SCAASGFTFS NYKMNWVRQA PGKGLEWVSD ISQSGASISY TGSVKGRFTI SRDNAKNTLY LQMNSLKPE DTAVYYCARC PAPFTRDCFD VTSTTYAYRG QGTQVTV |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 |
|---|---|
| グリッド | モデル: Quantifoil R1.2/1.3 / 支持フィルム - 材質: CARBON / 支持フィルム - トポロジー: HOLEY |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 277 K |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) デジタル化 - 画像ごとのフレーム数: 1-40 / 実像数: 6947 / 平均電子線量: 64.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 倍率(補正後): 49310 / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.0 µm / 最小 デフォーカス(公称値): 0.5 µm / 倍率(公称値): 29000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)