[English] 日本語
 Yorodumi
Yorodumi- EMDB-33329: High resolution cry-EM structure of the human 80S ribosome from S... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | High resolution cry-EM structure of the human 80S ribosome from SNORD127+/+ Kasumi-1 cells | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ribosome / rRNA ribomethylome | |||||||||
| Function / homology |  Function and homology information Function and homology informationtranslation at presynapse / exit from mitosis / optic nerve development / response to insecticide / regulation of translation involved in cellular response to UV / eukaryotic 80S initiation complex / negative regulation of formation of translation preinitiation complex / axial mesoderm development / negative regulation of endoplasmic reticulum unfolded protein response / ribosomal protein import into nucleus ...translation at presynapse / exit from mitosis / optic nerve development / response to insecticide / regulation of translation involved in cellular response to UV / eukaryotic 80S initiation complex / negative regulation of formation of translation preinitiation complex / axial mesoderm development / negative regulation of endoplasmic reticulum unfolded protein response / ribosomal protein import into nucleus / regulation of G1 to G0 transition / retinal ganglion cell axon guidance / oxidized pyrimidine DNA binding / response to TNF agonist / positive regulation of base-excision repair / positive regulation of ubiquitin-protein transferase activity / protein-DNA complex disassembly / positive regulation of respiratory burst involved in inflammatory response / positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator / positive regulation of gastrulation / positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage / protein tyrosine kinase inhibitor activity / 90S preribosome assembly / IRE1-RACK1-PP2A complex / positive regulation of Golgi to plasma membrane protein transport / nucleolus organization / positive regulation of DNA-templated transcription initiation / alpha-beta T cell differentiation / TNFR1-mediated ceramide production / positive regulation of DNA damage response, signal transduction by p53 class mediator / GAIT complex / negative regulation of RNA splicing / TORC2 complex binding / neural crest cell differentiation / supercoiled DNA binding / NF-kappaB complex / negative regulation of DNA repair / G1 to G0 transition / cytoplasmic translational initiation / oxidized purine DNA binding / cysteine-type endopeptidase activator activity involved in apoptotic process / middle ear morphogenesis / negative regulation of intrinsic apoptotic signaling pathway in response to hydrogen peroxide / rRNA modification in the nucleus and cytosol / negative regulation of bicellular tight junction assembly / ubiquitin-like protein conjugating enzyme binding / regulation of establishment of cell polarity / negative regulation of phagocytosis / erythrocyte homeostasis / cytoplasmic side of rough endoplasmic reticulum membrane / Formation of the ternary complex, and subsequently, the 43S complex / ion channel inhibitor activity / laminin receptor activity / protein kinase A binding / homeostatic process / pigmentation / Ribosomal scanning and start codon recognition / positive regulation of mitochondrial depolarization / Translation initiation complex formation / macrophage chemotaxis / lung morphogenesis / negative regulation of Wnt signaling pathway / positive regulation of natural killer cell proliferation / fibroblast growth factor binding / monocyte chemotaxis / BH3 domain binding / Protein hydroxylation / negative regulation of translational frameshifting / regulation of adenylate cyclase-activating G protein-coupled receptor signaling pathway / TOR signaling / positive regulation of GTPase activity / SARS-CoV-1 modulates host translation machinery / mTORC1-mediated signalling / iron-sulfur cluster binding / regulation of cell division / Peptide chain elongation / cellular response to ethanol / Selenocysteine synthesis / positive regulation of intrinsic apoptotic signaling pathway by p53 class mediator / Formation of a pool of free 40S subunits / negative regulation of protein binding / protein serine/threonine kinase inhibitor activity / Eukaryotic Translation Termination / blastocyst development / ubiquitin ligase inhibitor activity / SRP-dependent cotranslational protein targeting to membrane / Response of EIF2AK4 (GCN2) to amino acid deficiency / negative regulation of respiratory burst involved in inflammatory response / endonucleolytic cleavage to generate mature 3'-end of SSU-rRNA from (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / Viral mRNA Translation / positive regulation of signal transduction by p53 class mediator / protein localization to nucleus / negative regulation of ubiquitin-dependent protein catabolic process / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / GTP hydrolysis and joining of the 60S ribosomal subunit / L13a-mediated translational silencing of Ceruloplasmin expression / protein targeting / Major pathway of rRNA processing in the nucleolus and cytosol / regulation of translational fidelity / positive regulation of microtubule polymerization Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
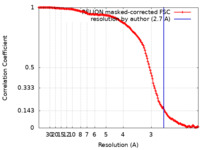
| Method | single particle reconstruction / cryo EM / Resolution: 2.7 Å | |||||||||
 Authors Authors | Cheng J / Beckmann R | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Cancer Discov / Year: 2023 Journal: Cancer Discov / Year: 2023Title: A Dynamic rRNA Ribomethylome Drives Stemness in Acute Myeloid Leukemia. Authors: Fengbiao Zhou / Nesrine Aroua / Yi Liu / Christian Rohde / Jingdong Cheng / Anna-Katharina Wirth / Daria Fijalkowska / Stefanie Göllner / Michelle Lotze / Haiyang Yun / Xiaobing Yu / ...Authors: Fengbiao Zhou / Nesrine Aroua / Yi Liu / Christian Rohde / Jingdong Cheng / Anna-Katharina Wirth / Daria Fijalkowska / Stefanie Göllner / Michelle Lotze / Haiyang Yun / Xiaobing Yu / Caroline Pabst / Tim Sauer / Thomas Oellerich / Hubert Serve / Christoph Röllig / Martin Bornhäuser / Christian Thiede / Claudia Baldus / Michaela Frye / Simon Raffel / Jeroen Krijgsveld / Irmela Jeremias / Roland Beckmann / Andreas Trumpp / Carsten Müller-Tidow /  Abstract: The development and regulation of malignant self-renewal remain unresolved issues. Here, we provide biochemical, genetic, and functional evidence that dynamics in ribosomal RNA (rRNA) 2'-O- ...The development and regulation of malignant self-renewal remain unresolved issues. Here, we provide biochemical, genetic, and functional evidence that dynamics in ribosomal RNA (rRNA) 2'-O-methylation regulate leukemia stem cell (LSC) activity in vivo. A comprehensive analysis of the rRNA 2'-O-methylation landscape of 94 patients with acute myeloid leukemia (AML) revealed dynamic 2'-O-methylation specifically at exterior sites of ribosomes. The rRNA 2'-O-methylation pattern is closely associated with AML development stage and LSC gene expression signature. Forced expression of the 2'-O-methyltransferase fibrillarin (FBL) induced an AML stem cell phenotype and enabled engraftment of non-LSC leukemia cells in NSG mice. Enhanced 2'-O-methylation redirected the ribosome translation program toward amino acid transporter mRNAs enriched in optimal codons and subsequently increased intracellular amino acid levels. Methylation at the single site 18S-guanosine 1447 was instrumental for LSC activity. Collectively, our work demonstrates that dynamic 2'-O-methylation at specific sites on rRNAs shifts translational preferences and controls AML LSC self-renewal. SIGNIFICANCE: We establish the complete rRNA 2'-O-methylation landscape in human AML. Plasticity of rRNA 2'-O-methylation shifts protein translation toward an LSC phenotype. This dynamic process ...SIGNIFICANCE: We establish the complete rRNA 2'-O-methylation landscape in human AML. Plasticity of rRNA 2'-O-methylation shifts protein translation toward an LSC phenotype. This dynamic process constitutes a novel concept of how cancers reprogram cell fate and function. This article is highlighted in the In This Issue feature, p. 247. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33329.map.gz emd_33329.map.gz | 157.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33329-v30.xml emd-33329-v30.xml emd-33329.xml emd-33329.xml | 102 KB 102 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_33329_fsc.xml emd_33329_fsc.xml | 14.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_33329.png emd_33329.png | 236.9 KB | ||
| Filedesc metadata |  emd-33329.cif.gz emd-33329.cif.gz | 20.4 KB | ||
| Others |  emd_33329_additional_1.map.gz emd_33329_additional_1.map.gz emd_33329_half_map_1.map.gz emd_33329_half_map_1.map.gz emd_33329_half_map_2.map.gz emd_33329_half_map_2.map.gz | 15.8 MB 225.2 MB 225.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33329 http://ftp.pdbj.org/pub/emdb/structures/EMD-33329 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33329 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33329 | HTTPS FTP |
-Related structure data
| Related structure data |  7xnxMC  7xnyC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_33329.map.gz / Format: CCP4 / Size: 282.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33329.map.gz / Format: CCP4 / Size: 282.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.059 Å | ||||||||||||||||||||||||||||||||||||
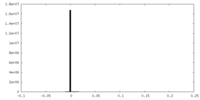
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_33329_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
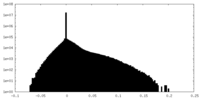
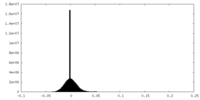
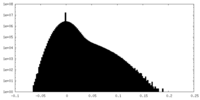
| Density Histograms |
-Half map: #2
| File | emd_33329_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_33329_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : ribosome
+Supramolecule #1: ribosome
+Macromolecule #1: 28S rRNA
+Macromolecule #2: 5S rRNA
+Macromolecule #3: 5.8S rRNA
+Macromolecule #45: 18S rRNA
+Macromolecule #79: tRNA
+Macromolecule #4: 60S ribosomal protein L8
+Macromolecule #5: 60S ribosomal protein L3
+Macromolecule #6: 60S ribosomal protein L4
+Macromolecule #7: 60S ribosomal protein L5
+Macromolecule #8: 60S ribosomal protein L6
+Macromolecule #9: 60S ribosomal protein L7
+Macromolecule #10: 60S ribosomal protein L7a
+Macromolecule #11: 60S ribosomal protein L9
+Macromolecule #12: Ribosomal protein L10 isoform A
+Macromolecule #13: 60S ribosomal protein L11
+Macromolecule #14: 60S ribosomal protein L13
+Macromolecule #15: 60S ribosomal protein L14
+Macromolecule #16: 60S ribosomal protein L15
+Macromolecule #17: 60S ribosomal protein L13a
+Macromolecule #18: 60S ribosomal protein L17
+Macromolecule #19: 60S ribosomal protein L18
+Macromolecule #20: 60S ribosomal protein L19
+Macromolecule #21: 60S ribosomal protein L18a
+Macromolecule #22: 60S ribosomal protein L21
+Macromolecule #23: 60S ribosomal protein L22
+Macromolecule #24: 60S ribosomal protein L23
+Macromolecule #25: 60S ribosomal protein L24
+Macromolecule #26: 60S ribosomal protein L23a
+Macromolecule #27: 60S ribosomal protein L26
+Macromolecule #28: 60S ribosomal protein L27
+Macromolecule #29: 60S ribosomal protein L29
+Macromolecule #30: 60S ribosomal protein L30
+Macromolecule #31: 60S ribosomal protein L31
+Macromolecule #32: 60S ribosomal protein L32
+Macromolecule #33: 60S ribosomal protein L35a
+Macromolecule #34: 60S ribosomal protein L34
+Macromolecule #35: 60S ribosomal protein L35
+Macromolecule #36: 60S ribosomal protein L36
+Macromolecule #37: 60S ribosomal protein L37
+Macromolecule #38: 60S ribosomal protein L38
+Macromolecule #39: 60S ribosomal protein L39
+Macromolecule #40: Ubiquitin-60S ribosomal protein L40
+Macromolecule #41: 60S ribosomal protein L41
+Macromolecule #42: 60S ribosomal protein L36a
+Macromolecule #43: 60S ribosomal protein L37a
+Macromolecule #44: 60S ribosomal protein L28
+Macromolecule #46: 40S ribosomal protein SA
+Macromolecule #47: 40S ribosomal protein S3a
+Macromolecule #48: 40S ribosomal protein S3
+Macromolecule #49: 40S ribosomal protein S4, X isoform
+Macromolecule #50: 40S ribosomal protein S5
+Macromolecule #51: 40S ribosomal protein S7
+Macromolecule #52: 40S ribosomal protein S8
+Macromolecule #53: 40S ribosomal protein S10
+Macromolecule #54: 40S ribosomal protein S11
+Macromolecule #55: 40S ribosomal protein S15
+Macromolecule #56: 40S ribosomal protein S16
+Macromolecule #57: 40S ribosomal protein S17
+Macromolecule #58: 40S ribosomal protein S18
+Macromolecule #59: 40S ribosomal protein S19
+Macromolecule #60: 40S ribosomal protein S20
+Macromolecule #61: 40S ribosomal protein S21
+Macromolecule #62: 40S ribosomal protein S23
+Macromolecule #63: 40S ribosomal protein S26
+Macromolecule #64: 40S ribosomal protein S28
+Macromolecule #65: 40S ribosomal protein S29
+Macromolecule #66: Receptor of activated protein C kinase 1
+Macromolecule #67: 40S ribosomal protein S2
+Macromolecule #68: 40S ribosomal protein S6
+Macromolecule #69: 40S ribosomal protein S9
+Macromolecule #70: 40S ribosomal protein S12
+Macromolecule #71: 40S ribosomal protein S13
+Macromolecule #72: 40S ribosomal protein S14
+Macromolecule #73: 40S ribosomal protein S15a
+Macromolecule #74: 40S ribosomal protein S24
+Macromolecule #75: 40S ribosomal protein S25
+Macromolecule #76: 40S ribosomal protein S27
+Macromolecule #77: 40S ribosomal protein S30
+Macromolecule #78: Ubiquitin-40S ribosomal protein S27a
+Macromolecule #80: MAGNESIUM ION
+Macromolecule #81: POTASSIUM ION
+Macromolecule #82: ZINC ION
+Macromolecule #83: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 44.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source: TUNGSTEN HAIRPIN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)