[English] 日本語
 Yorodumi
Yorodumi- EMDB-32978: Cryo-EM structure of RNC-RAC complex in presence of Ssb from S. c... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of RNC-RAC complex in presence of Ssb from S. cerevisiae 2 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | RAC / co-translational folding / TRANSLATION / TRANSLATION-RNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationtranslational frameshifting / 'de novo' cotranslational protein folding / Regulation of HSF1-mediated heat shock response / protein folding chaperone complex / ribosomal subunit export from nucleus / regulation of translational fidelity / Hsp70 protein binding / rRNA processing / protein folding / ribosome binding ...translational frameshifting / 'de novo' cotranslational protein folding / Regulation of HSF1-mediated heat shock response / protein folding chaperone complex / ribosomal subunit export from nucleus / regulation of translational fidelity / Hsp70 protein binding / rRNA processing / protein folding / ribosome binding / transcription coactivator activity / intracellular signal transduction / ribosome / nucleolus / mitochondrion / DNA binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / negative staining / Resolution: 3.1 Å | |||||||||
 Authors Authors | Chen Y / Gao N | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Structural remodeling of ribosome associated Hsp40-Hsp70 chaperones during co-translational folding. Authors: Yan Chen / Bin Tsai / Ningning Li / Ning Gao /  Abstract: Ribosome associated complex (RAC), an obligate heterodimer of HSP40 and HSP70 (Zuo1 and Ssz1 in yeast), is conserved in eukaryotes and functions as co-chaperone for another HSP70 (Ssb1/2 in yeast) to ...Ribosome associated complex (RAC), an obligate heterodimer of HSP40 and HSP70 (Zuo1 and Ssz1 in yeast), is conserved in eukaryotes and functions as co-chaperone for another HSP70 (Ssb1/2 in yeast) to facilitate co-translational folding of nascent polypeptides. Many mechanistic details, such as the coordination of one HSP40 with two HSP70s and the dynamic interplay between RAC-Ssb and growing nascent chains, remain unclear. Here, we report three sets of structures of RAC-containing ribosomal complexes isolated from Saccharomyces cerevisiae. Structural analyses indicate that RAC on the nascent-chain-free ribosome is in an autoinhibited conformation, and in the presence of a nascent chain at the peptide tunnel exit (PTE), RAC undergoes large-scale structural remodeling to make Zuo1 J-Domain more accessible to Ssb. Our data also suggest a role of Zuo1 in orienting Ssb-SBD proximal to the PTE for easy capture of the substrate. Altogether, in accordance with previous data, our work suggests a sequence of structural remodeling events for RAC-Ssb during co-translational folding, triggered by the binding and passage of growing nascent chain from one to another. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_32978.map.gz emd_32978.map.gz | 140.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-32978-v30.xml emd-32978-v30.xml emd-32978.xml emd-32978.xml | 16.4 KB 16.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_32978.png emd_32978.png | 70.3 KB | ||
| Filedesc metadata |  emd-32978.cif.gz emd-32978.cif.gz | 5.5 KB | ||
| Others |  emd_32978_half_map_1.map.gz emd_32978_half_map_1.map.gz emd_32978_half_map_2.map.gz emd_32978_half_map_2.map.gz | 140.9 MB 140.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-32978 http://ftp.pdbj.org/pub/emdb/structures/EMD-32978 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32978 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32978 | HTTPS FTP |
-Related structure data
| Related structure data |  7x34MC  7x3kC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_32978.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_32978.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.356 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_32978_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
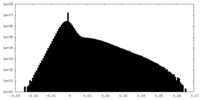
| Projections & Slices |
| ||||||||||||
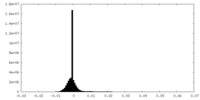
| Density Histograms |
-Half map: #1
| File | emd_32978_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
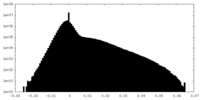
| Projections & Slices |
| ||||||||||||
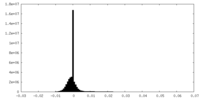
| Density Histograms |
- Sample components
Sample components
-Entire : RNC-RAC complex in presence of Ssb from S. cerevisiae 2
| Entire | Name: RNC-RAC complex in presence of Ssb from S. cerevisiae 2 |
|---|---|
| Components |
|
-Supramolecule #1: RNC-RAC complex in presence of Ssb from S. cerevisiae 2
| Supramolecule | Name: RNC-RAC complex in presence of Ssb from S. cerevisiae 2 type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Zuotin
| Macromolecule | Name: Zuotin / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 10.619055 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: ASAKADKKKA KEAAKAAKKK NKRAIRNSAK EADYFGDADK ATTIDEQVGL IVDSLNDEEL VSTADKIKAN AAGAKEVLKE SAKTIVDSG KLPSSLLSYF V UniProtKB: Zuotin |
-Macromolecule #2: RNA (130-mer)
| Macromolecule | Name: RNA (130-mer) / type: rna / ID: 2 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 42.010996 KDa |
| Sequence | String: CGCCCGUCGC UAGUACCGAU UGAAUGGCUU AGUGAGGCCU CAGGAUCUGC UUAGAGAAGG GGGCAACUCC AUCUCAGAGC GGAGAAUUU GGACAAACUU GGUCAUUUAG AGGAACUAAA AGUCGUAACA A GENBANK: GENBANK: CP011821.1 |
-Experimental details
-Structure determination
| Method | negative staining, cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Staining | Type: NEGATIVE / Material: Uranyl Acetate |
| Vitrification | Cryogen name: NITROGEN |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 35.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: DARK FIELD / Nominal defocus max: 1.6 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: RIGID BODY FIT |
|---|---|
| Output model |  PDB-7x34: |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)