+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
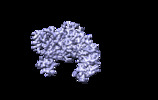
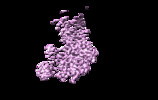
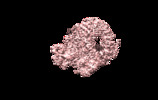
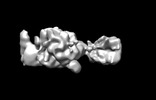
| Title | The cryo-EM map of the DDX42-SF3b complex core region | |||||||||
 Map data Map data | The EM map of the DDX42-SF3b complex | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | DDX32 / SF3B1 / SF3b / U2 snRNP / SPLICING | |||||||||
| Function / homology |  Function and homology information Function and homology informationU11/U12 snRNP / B-WICH complex / splicing factor binding / U12-type spliceosomal complex / RNA splicing, via transesterification reactions / U2-type spliceosomal complex / U2-type precatalytic spliceosome / U2-type prespliceosome assembly / U2 snRNP / SAGA complex ...U11/U12 snRNP / B-WICH complex / splicing factor binding / U12-type spliceosomal complex / RNA splicing, via transesterification reactions / U2-type spliceosomal complex / U2-type precatalytic spliceosome / U2-type prespliceosome assembly / U2 snRNP / SAGA complex / positive regulation of transcription by RNA polymerase III / U2-type prespliceosome / positive regulation of transcription by RNA polymerase I / precatalytic spliceosome / spliceosomal complex assembly / mRNA Splicing - Minor Pathway / regulation of RNA splicing / U2 snRNA binding / regulation of DNA repair / Cajal body / catalytic step 2 spliceosome / mRNA Splicing - Major Pathway / RNA splicing / stem cell differentiation / spliceosomal complex / B-WICH complex positively regulates rRNA expression / negative regulation of protein catabolic process / protein localization / nuclear matrix / mRNA splicing, via spliceosome / regulation of apoptotic process / RNA helicase activity / RNA helicase / nuclear speck / chromatin remodeling / mRNA binding / protein-containing complex binding / nucleolus / positive regulation of DNA-templated transcription / ATP hydrolysis activity / positive regulation of transcription by RNA polymerase II / DNA binding / RNA binding / zinc ion binding / nucleoplasm / ATP binding / membrane / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.6 Å | |||||||||
 Authors Authors | Zhang X / Zhan X | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat.Struct.Mol.Biol. / Year: 2024 Journal: Nat.Struct.Mol.Biol. / Year: 2024Title: Structural insights into branch site proofreading by human spliceosome Authors: Zhang X / Zhan X / Bian T / Yang F / Li P / Lu Y / Xing Z / Fan R / Zhang QC / Shi Y | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_31330.map.gz emd_31330.map.gz | 49.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-31330-v30.xml emd-31330-v30.xml emd-31330.xml emd-31330.xml | 17.2 KB 17.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_31330.png emd_31330.png | 87.6 KB | ||
| Filedesc metadata |  emd-31330.cif.gz emd-31330.cif.gz | 7.5 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-31330 http://ftp.pdbj.org/pub/emdb/structures/EMD-31330 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-31330 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-31330 | HTTPS FTP |
-Validation report
| Summary document |  emd_31330_validation.pdf.gz emd_31330_validation.pdf.gz | 584 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_31330_full_validation.pdf.gz emd_31330_full_validation.pdf.gz | 583.5 KB | Display | |
| Data in XML |  emd_31330_validation.xml.gz emd_31330_validation.xml.gz | 5.9 KB | Display | |
| Data in CIF |  emd_31330_validation.cif.gz emd_31330_validation.cif.gz | 6.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-31330 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-31330 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-31330 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-31330 | HTTPS FTP |
-Related structure data
| Related structure data |  7evnMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_31330.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_31330.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The EM map of the DDX42-SF3b complex | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.087 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : The DDX42-SF3b core complex
| Entire | Name: The DDX42-SF3b core complex |
|---|---|
| Components |
|
-Supramolecule #1: The DDX42-SF3b core complex
| Supramolecule | Name: The DDX42-SF3b core complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Splicing factor 3B subunit 5
| Macromolecule | Name: Splicing factor 3B subunit 5 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 10.149369 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MTDRYTIHSQ LEHLQSKYIG TGHADTTKWE WLVNQHRDSY CSYMGHFDLL NYFAIAENES KARVRFNLME KMLQPCGPPA DKPEEN UniProtKB: Splicing factor 3B subunit 5 |
-Macromolecule #2: Splicing factor 3B subunit 1
| Macromolecule | Name: Splicing factor 3B subunit 1 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 98.747867 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MASDYKDDDD KASDEVDAGT MKSVNDQPSG NLPFLKPDDI QYFDKLLVDV DESTLSPEEQ KERKIMKLLL KIKNGTPPMR KAALRQITD KAREFGAGPL FNQILPLLMS PTLEDQERHL LVKVIDRILY KLDDLVRPYV HKILVVIEPL LIDEDYYARV E GREIISNL ...String: MASDYKDDDD KASDEVDAGT MKSVNDQPSG NLPFLKPDDI QYFDKLLVDV DESTLSPEEQ KERKIMKLLL KIKNGTPPMR KAALRQITD KAREFGAGPL FNQILPLLMS PTLEDQERHL LVKVIDRILY KLDDLVRPYV HKILVVIEPL LIDEDYYARV E GREIISNL AKAAGLATMI STMRPDIDNM DEYVRNTTAR AFAVVASALG IPSLLPFLKA VCKSKKSWQA RHTGIKIVQQ IA ILMGCAI LPHLRSLVEI IEHGLVDEQQ KVRTISALAI AALAEAATPY GIESFDSVLK PLWKGIRQHR GKGLAAFLKA IGY LIPLMD AEYANYYTRE VMLILIREFQ SPDEEMKKIV LKVVKQCCGT DGVEANYIKT EILPPFFKHF WQHRMALDRR NYRQ LVDTT VELANKVGAA EIISRIVDDL KDEAEQYRKM VMETIEKIMG NLGAADIDHK LEEQLIDGIL YAFQEQTTED SVMLN GFGT VVNALGKRVK PYLPQICGTV LWRLNNKSAK VRQQAADLIS RTAVVMKTCQ EEKLMGHLGV VLYEYLGEEY PEVLGS ILG ALKAIVNVIG MHKMTPPIKD LLPRLTPILK NRHEKVQENC IDLVGRIADR GAEYVSAREW MRICFELLEL LKAHKKA IR RATVNTFGYI AKAIGPHDVL ATLLNNLKVQ ERQNRVCTTV AIAIVAETCS PFTVLPALMN EYRVPELNVQ NGVLKSLS F LFEYIGEMGK DYIYAVTPLL EDALMDRDLV HRQTASAVVQ HMSLGVYGFG CEDSLNHLLN YVWPNVFETS PHVIQAVMG ALEGLRVAIG PCRMLQYCLQ GLFHPARKVR DVYWKIYNSI YIGSQDALIA HYPRIYNDDK NTYIRYELDY IL UniProtKB: Splicing factor 3B subunit 1 |
-Macromolecule #3: PHD finger-like domain-containing protein 5A
| Macromolecule | Name: PHD finger-like domain-containing protein 5A / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 12.427524 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MAKHHPDLIF CRKQAGVAIG RLCEKCDGKC VICDSYVRPC TLVRICDECN YGSYQGRCVI CGGPGVSDAY YCKECTIQEK DRDGCPKIV NLGSSKTDLF YERKKYGFKK R UniProtKB: PHD finger-like domain-containing protein 5A |
-Macromolecule #4: Splicing factor 3B subunit 3
| Macromolecule | Name: Splicing factor 3B subunit 3 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 138.949188 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: WSHPQFEKGG GSGGGSGGSA WSHPQFEKGS AAAMFLYNLT LQRATGISFA IHGNFSGTKQ QEIVVSRGKI LELLRPDPNT GKVHTLLTV EVFGVIRSLM AFRLTGGTKD YIVVGSDSGR IVILEYQPSK NMFEKIHQET FGKSGCRRIV PGQFLAVDPK G RAVMISAI ...String: WSHPQFEKGG GSGGGSGGSA WSHPQFEKGS AAAMFLYNLT LQRATGISFA IHGNFSGTKQ QEIVVSRGKI LELLRPDPNT GKVHTLLTV EVFGVIRSLM AFRLTGGTKD YIVVGSDSGR IVILEYQPSK NMFEKIHQET FGKSGCRRIV PGQFLAVDPK G RAVMISAI EKQKLVYILN RDAAARLTIS SPLEAHKANT LVYHVVGVDV GFENPMFACL EMDYEEADND PTGEAAANTQ QT LTFYELD LGLNHVVRKY SEPLEEHGNF LITVPGGSDG PSGVLICSEN YITYKNFGDQ PDIRCPIPRR RNDLDDPERG MIF VCSATH KTKSMFFFLA QTEQGDIFKI TLETDEDMVT EIRLKYFDTV PVAAAMCVLK TGFLFVASEF GNHYLYQIAH LGDD DEEPE FSSAMPLEEG DTFFFQPRPL KNLVLVDELD SLSPILFCQI ADLANEDTPQ LYVACGRGPR SSLRVLRHGL EVSEM AVSE LPGNPNAVWT VRRHIEDEFD AYIIVSFVNA TLVLSIGETV EEVTDSGFLG TTPTLSCSLL GDDALVQVYP DGIRHI RAD KRVNEWKTPG KKTIVKCAVN QRQVVIALTG GELVYFEMDP SGQLNEYTER KEMSADVVCM SLANVPPGEQ RSRFLAV GL VDNTVRIISL DPSDCLQPLS MQALPAQPES LCIVEMGGTE KQDELGERGS IGFLYLNIGL QNGVLLRTVL DPVTGDLS D TRTRYLGSRP VKLFRVRMQG QEAVLAMSSR SWLSYSYQSR FHLTPLSYET LEFASGFASE QCPEGIVAIS TNTLRILAL EKLGAVFNQV AFPLQYTPRK FVIHPESNNL IIIETDHNAY TEATKAQRKQ QMAEEMVEAA GEDERELAAE MAAAFLNENL PESIFGAPK AGNGQWASVI RVMNPIQGNT LDLVQLEQNE AAFSVAVCRF SNTGEDWYVL VGVAKDLILN PRSVAGGFVY T YKLVNNGE KLEFLHKTPV EEVPAAIAPF QGRVLIGVGK LLRVYDLGKK KLLRKCENKH IANYISGIQT IGHRVIVSDV QE SFIWVRY KRNENQLIIF ADDTYPRWVT TASLLDYDTV AGADKFGNIC VVRLPPNTND EVDEDPTGNK ALWDRGLLNG ASQ KAEVIM NYHVGETVLS LQKTTLIPGG SESLVYTTLS GGIGILVPFT SHEDHDFFQH VEMHLRSEHP PLCGRDHLSF RSYY FPVKN VIDGDLCEQF NSMEPNKQKN VSEELDRTPP EVSKKLEDIR TRYAF UniProtKB: Splicing factor 3B subunit 3 |
-Macromolecule #5: ATP-dependent RNA helicase DDX42
| Macromolecule | Name: ATP-dependent RNA helicase DDX42 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO / EC number: RNA helicase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 105.261625 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MASDYKDDDD KASDEVDAGT MNWNKGGPGT KRGFGFGGFA ISAGKKEEPK LPQQSHSAFG ATSSSSGFGK SAPPQLPSFY KIGSKRANF DEENAYFEDE EEDSSNVDLP YIPAENSPTR QQFHSKPVDS DSDDDPLEAF MAEVEDQAAR DMKRLEEKDK E RKNVKGIR ...String: MASDYKDDDD KASDEVDAGT MNWNKGGPGT KRGFGFGGFA ISAGKKEEPK LPQQSHSAFG ATSSSSGFGK SAPPQLPSFY KIGSKRANF DEENAYFEDE EEDSSNVDLP YIPAENSPTR QQFHSKPVDS DSDDDPLEAF MAEVEDQAAR DMKRLEEKDK E RKNVKGIR DDIEEEDDQE AYFRYMAENP TAGVVQEEEE DNLEYDSDGN PIAPTKKIID PLPPIDHSEI DYPPFEKNFY NE HEEITNL TPQQLIDLRH KLNLRVSGAA PPRPGSSFAH FGFDEQLMHQ IRKSEYTQPT PIQCQGVPVA LSGRDMIGIA KTG SGKTAA FIWPMLIHIM DQKELEPGDG PIAVIVCPTR ELCQQIHAEC KRFGKAYNLR SVAVYGGGSM WEQAKALQEG AEIV VCTPG RLIDHVKKKA TNLQRVSYLV FDEADRMFDM GFEYQVRSIA SHVRPDRQTL LFSATFRKKI EKLARDILID PIRVV QGDI GEANEDVTQI VEILHSGPSK WNWLTRRLVE FTSSGSVLLF VTKKANAEEL ANNLKQEGHN LGLLHGDMDQ SERNKV ISD FKKKDIPVLV ATDVAARGLD IPSIKTVINY DVARDIDTHT HRIGRTGRAG EKGVAYTLLT PKDSNFAGDL VRNLEGA NQ HVSKELLDLA MQNAWFRKSR FKGGKGKKLN IGGGGLGYRE RPGLGSENMD RGNNNVMSNY EAYKPSTGAM GDRLTAMK A AFQSQYKSHF VAASLSNQKA GSSAAGASGW TSAGSLNSVP TNSAQQGHNS PDSPVTSAAK GIPGFGNTGN ISGAPVTYP SAGAQGVNNT ASGNNSREGT GGSNGKRERY TENRGSSRHS HGETGNRHSD SPRHGDGGRH GDGYRHPESS SRHTDGHRHG ENRHGGSAG RHGENRGAND GRNGESRKEA FNRESKMEPK MEPKVDSSKM DKVDSKTDKT ADGFAVPEPP KRKKSRWDS UniProtKB: ATP-dependent RNA helicase DDX42 |
-Macromolecule #6: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 6 / Number of copies: 3 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.5 mg/mL |
|---|---|
| Buffer | pH: 7.9 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: EMDB MAP |
|---|---|
| Final reconstruction | Algorithm: FOURIER SPACE / Resolution.type: BY AUTHOR / Resolution: 2.6 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 234800 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller