+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-30296 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of yeast Ribonuclease MRP | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Ribonuclease MRP / RNA-protein complex / RNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationribonuclease MRP activity / nuclear-transcribed mRNA catabolic process, RNase MRP-dependent / intronic box C/D snoRNA processing / nucleolar ribonuclease P complex / ribonuclease MRP complex / ribonuclease P RNA binding / ribonuclease P complex / plasmid partitioning / ribonuclease P / nuclease activity ...ribonuclease MRP activity / nuclear-transcribed mRNA catabolic process, RNase MRP-dependent / intronic box C/D snoRNA processing / nucleolar ribonuclease P complex / ribonuclease MRP complex / ribonuclease P RNA binding / ribonuclease P complex / plasmid partitioning / ribonuclease P / nuclease activity / rRNA primary transcript binding / ribonuclease P activity / tRNA 5'-leader removal / maturation of 5.8S rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / telomerase holoenzyme complex / tRNA processing / maturation of 5.8S rRNA / mRNA processing / rRNA processing / nucleolus / RNA binding / nucleus / membrane / cytosol / cytoplasm Similarity search - Function | |||||||||
| Biological species |    | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.5 Å | |||||||||
 Authors Authors | Lan P / Wu J | |||||||||
 Citation Citation |  Journal: Science / Year: 2020 Journal: Science / Year: 2020Title: Structural insight into precursor ribosomal RNA processing by ribonuclease MRP. Authors: Pengfei Lan / Bin Zhou / Ming Tan / Shaobai Li / Mi Cao / Jian Wu / Ming Lei /  Abstract: Ribonuclease (RNase) MRP is a conserved eukaryotic ribonucleoprotein complex that plays essential roles in precursor ribosomal RNA (pre-rRNA) processing and cell cycle regulation. In contrast to ...Ribonuclease (RNase) MRP is a conserved eukaryotic ribonucleoprotein complex that plays essential roles in precursor ribosomal RNA (pre-rRNA) processing and cell cycle regulation. In contrast to RNase P, which selectively cleaves transfer RNA-like substrates, it has remained a mystery how RNase MRP recognizes its diverse substrates. To address this question, we determined cryo-electron microscopy structures of RNase MRP alone and in complex with a fragment of pre-rRNA. These structures and the results of biochemical studies reveal that coevolution of both protein and RNA subunits has transformed RNase MRP into a distinct ribonuclease that processes single-stranded RNAs by recognizing a short, loosely defined consensus sequence. This broad substrate specificity suggests that RNase MRP may have myriad yet unrecognized substrates that could play important roles in various cellular contexts. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_30296.map.gz emd_30296.map.gz | 202.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-30296-v30.xml emd-30296-v30.xml emd-30296.xml emd-30296.xml | 21.5 KB 21.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_30296.png emd_30296.png | 103 KB | ||
| Filedesc metadata |  emd-30296.cif.gz emd-30296.cif.gz | 7.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-30296 http://ftp.pdbj.org/pub/emdb/structures/EMD-30296 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30296 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30296 | HTTPS FTP |
-Validation report
| Summary document |  emd_30296_validation.pdf.gz emd_30296_validation.pdf.gz | 496.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_30296_full_validation.pdf.gz emd_30296_full_validation.pdf.gz | 496.3 KB | Display | |
| Data in XML |  emd_30296_validation.xml.gz emd_30296_validation.xml.gz | 6.9 KB | Display | |
| Data in CIF |  emd_30296_validation.cif.gz emd_30296_validation.cif.gz | 7.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-30296 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-30296 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-30296 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-30296 | HTTPS FTP |
-Related structure data
| Related structure data |  7c79MC  7c7aC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_30296.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_30296.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.1 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
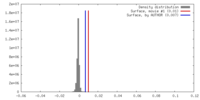
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Ribonuclease MRP
+Supramolecule #1: Ribonuclease MRP
+Macromolecule #1: Ribonuclease MRP RNA subunit NME1
+Macromolecule #2: Ribonucleases P/MRP protein subunit POP1
+Macromolecule #3: Ribonucleases P/MRP protein subunit POP3
+Macromolecule #4: RNases MRP/P 32.9 kDa subunit
+Macromolecule #5: Ribonuclease P/MRP protein subunit POP5
+Macromolecule #6: Ribonucleases P/MRP protein subunit POP6
+Macromolecule #7: Ribonucleases P/MRP protein subunit POP7
+Macromolecule #8: Ribonucleases P/MRP protein subunit POP8
+Macromolecule #9: Ribonuclease P/MRP protein subunit RPP1
+Macromolecule #10: Ribonuclease MRP protein subunit SNM1
+Macromolecule #11: Ribonuclease MRP protein subunit RMP1
+Macromolecule #12: MAGNESIUM ION
+Macromolecule #13: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 2.5 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 1232761 |
| Initial angle assignment | Type: COMMON LINE |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)