[English] 日本語
 Yorodumi
Yorodumi- EMDB-28155: The complex of phosphorylated human delta F508 cystic fibrosis tr... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | The complex of phosphorylated human delta F508 cystic fibrosis transmembrane conductance regulator (CFTR) with elexacaftor (VX-445) and ATP/Mg | |||||||||
 Map data Map data | d508/E1371Q VX-445 ATP/Mg | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ABC transporter / ion channel / folding correction / membrane protein / ATP-Binding Protein | |||||||||
| Function / homology |  Function and homology information Function and homology informationSec61 translocon complex binding / channel-conductance-controlling ATPase / intracellularly ATP-gated chloride channel activity / positive regulation of enamel mineralization / RHO GTPases regulate CFTR trafficking / transepithelial water transport / intracellular pH elevation / amelogenesis / chloride channel inhibitor activity / : ...Sec61 translocon complex binding / channel-conductance-controlling ATPase / intracellularly ATP-gated chloride channel activity / positive regulation of enamel mineralization / RHO GTPases regulate CFTR trafficking / transepithelial water transport / intracellular pH elevation / amelogenesis / chloride channel inhibitor activity / : / multicellular organismal-level water homeostasis / water transport / chloride channel regulator activity / Golgi-associated vesicle membrane / cholesterol transport / bicarbonate transmembrane transporter activity / bicarbonate transport / chloride transmembrane transporter activity / membrane hyperpolarization / cholesterol biosynthetic process / sperm capacitation / RHOQ GTPase cycle / chloride channel activity / chloride channel complex / ABC-type transporter activity / 14-3-3 protein binding / establishment of localization in cell / cellular response to forskolin / response to endoplasmic reticulum stress / cellular response to cAMP / chloride transmembrane transport / Developmental Lineage of Pancreatic Ductal Cells / PDZ domain binding / clathrin-coated endocytic vesicle membrane / Late endosomal microautophagy / Defective CFTR causes cystic fibrosis / recycling endosome / transmembrane transport / ABC-family protein mediated transport / recycling endosome membrane / Chaperone Mediated Autophagy / Aggrephagy / Cargo recognition for clathrin-mediated endocytosis / Clathrin-mediated endocytosis / protein-folding chaperone binding / early endosome membrane / basolateral plasma membrane / early endosome / endosome membrane / apical plasma membrane / Ub-specific processing proteases / lysosomal membrane / endoplasmic reticulum membrane / enzyme binding / cell surface / ATP hydrolysis activity / protein-containing complex / ATP binding / membrane / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Fiedorczuk K / Chen J | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2022 Journal: Science / Year: 2022Title: Molecular structures reveal synergistic rescue of Δ508 CFTR by Trikafta modulators. Authors: Karol Fiedorczuk / Jue Chen /  Abstract: The predominant mutation causing cystic fibrosis, a deletion of phenylalanine 508 (Δ508) in the cystic fibrosis transmembrane conductance regulator (CFTR), leads to severe defects in CFTR biogenesis ...The predominant mutation causing cystic fibrosis, a deletion of phenylalanine 508 (Δ508) in the cystic fibrosis transmembrane conductance regulator (CFTR), leads to severe defects in CFTR biogenesis and function. The advanced therapy Trikafta combines the folding corrector tezacaftor (VX-661), the channel potentiator ivacaftor (VX-770), and the dual-function modulator elexacaftor (VX-445). However, it is unclear how elexacaftor exerts its effects, in part because the structure of Δ508 CFTR is unknown. Here, we present cryo-electron microscopy structures of Δ508 CFTR in the absence and presence of CFTR modulators. When used alone, elexacaftor partially rectified interdomain assembly defects in Δ508 CFTR, but when combined with a type I corrector, did so fully. These data illustrate how the different modulators in Trikafta synergistically rescue Δ508 CFTR structure and function. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_28155.map.gz emd_28155.map.gz | 304.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-28155-v30.xml emd-28155-v30.xml emd-28155.xml emd-28155.xml | 20.1 KB 20.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_28155_fsc.xml emd_28155_fsc.xml | 15.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_28155.png emd_28155.png | 103.9 KB | ||
| Masks |  emd_28155_msk_1.map emd_28155_msk_1.map | 325 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-28155.cif.gz emd-28155.cif.gz | 7.2 KB | ||
| Others |  emd_28155_half_map_1.map.gz emd_28155_half_map_1.map.gz emd_28155_half_map_2.map.gz emd_28155_half_map_2.map.gz | 260.1 MB 259.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-28155 http://ftp.pdbj.org/pub/emdb/structures/EMD-28155 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28155 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28155 | HTTPS FTP |
-Related structure data
| Related structure data |  8eigMC  8eioC  8eiqC  8ej1C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_28155.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_28155.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | d508/E1371Q VX-445 ATP/Mg | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.676 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_28155_msk_1.map emd_28155_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map 1
| File | emd_28155_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map 2
| File | emd_28155_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : The complex of phosphorylated human delta F508 cystic fibrosis tr...
| Entire | Name: The complex of phosphorylated human delta F508 cystic fibrosis transmembrane conductance regulator (CFTR) with elexacaftor (VX-445) and ATP/Mg |
|---|---|
| Components |
|
-Supramolecule #1: The complex of phosphorylated human delta F508 cystic fibrosis tr...
| Supramolecule | Name: The complex of phosphorylated human delta F508 cystic fibrosis transmembrane conductance regulator (CFTR) with elexacaftor (VX-445) and ATP/Mg type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Cystic fibrosis transmembrane conductance regulator
| Macromolecule | Name: Cystic fibrosis transmembrane conductance regulator / type: protein_or_peptide / ID: 1 Details: Construct used for expression is the human CFTR with a deletion of F508 and E1371Q substitution. Number of copies: 1 / Enantiomer: LEVO / EC number: channel-conductance-controlling ATPase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 168.187297 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MQRSPLEKAS VVSKLFFSWT RPILRKGYRQ RLELSDIYQI PSVDSADNLS EKLEREWDRE LASKKNPKLI NALRRCFFWR FMFYGIFLY LGEVTKAVQP LLLGRIIASY DPDNKEERSI AIYLGIGLCL LFIVRTLLLH PAIFGLHHIG MQMRIAMFSL I YKKTLKLS ...String: MQRSPLEKAS VVSKLFFSWT RPILRKGYRQ RLELSDIYQI PSVDSADNLS EKLEREWDRE LASKKNPKLI NALRRCFFWR FMFYGIFLY LGEVTKAVQP LLLGRIIASY DPDNKEERSI AIYLGIGLCL LFIVRTLLLH PAIFGLHHIG MQMRIAMFSL I YKKTLKLS SRVLDKISIG QLVSLLSNNL NKFDEGLALA HFVWIAPLQV ALLMGLIWEL LQASAFCGLG FLIVLALFQA GL GRMMMKY RDQRAGKISE RLVITSEMIE NIQSVKAYCW EEAMEKMIEN LRQTELKLTR KAAYVRYFNS SAFFFSGFFV VFL SVLPYA LIKGIILRKI FTTISFCIVL RMAVTRQFPW AVQTWYDSLG AINKIQDFLQ KQEYKTLEYN LTTTEVVMEN VTAF WEEGF GELFEKAKQN NNNRKTSNGD DSLFFSNFSL LGTPVLKDIN FKIERGQLLA VAGSTGAGKT SLLMVIMGEL EPSEG KIKH SGRISFCSQF SWIMPGTIKE NIIGVSYDEY RYRSVIKACQ LEEDISKFAE KDNIVLGEGG ITLSGGQRAR ISLARA VYK DADLYLLDSP FGYLDVLTEK EIFESCVCKL MANKTRILVT SKMEHLKKAD KILILHEGSS YFYGTFSELQ NLQPDFS SK LMGCDSFDQF SAERRNSILT ETLHRFSLEG DAPVSWTETK KQSFKQTGEF GEKRKNSILN PINSIRKFSI VQKTPLQM N GIEEDSDEPL ERRLSLVPDS EQGEAILPRI SVISTGPTLQ ARRRQSVLNL MTHSVNQGQN IHRKTTASTR KVSLAPQAN LTELDIYSRR LSQETGLEIS EEINEEDLKE CFFDDMESIP AVTTWNTYLR YITVHKSLIF VLIWCLVIFL AEVAASLVVL WLLGNTPLQ DKGNSTHSRN NSYAVIITST SSYYVFYIYV GVADTLLAMG FFRGLPLVHT LITVSKILHH KMLHSVLQAP M STLNTLKA GGILNRFSKD IAILDDLLPL TIFDFIQLLL IVIGAIAVVA VLQPYIFVAT VPVIVAFIML RAYFLQTSQQ LK QLESEGR SPIFTHLVTS LKGLWTLRAF GRQPYFETLF HKALNLHTAN WFLYLSTLRW FQMRIEMIFV IFFIAVTFIS ILT TGEGEG RVGIILTLAM NIMSTLQWAV NSSIDVDSLM RSVSRVFKFI DMPTEGKPTK STKPYKNGQL SKVMIIENSH VKKD DIWPS GGQMTVKDLT AKYTEGGNAI LENISFSISP GQRVGLLGRT GSGKSTLLSA FLRLLNTEGE IQIDGVSWDS ITLQQ WRKA FGVIPQKVFI FSGTFRKNLD PYEQWSDQEI WKVADEVGLR SVIEQFPGKL DFVLVDGGCV LSHGHKQLMC LARSVL SKA KILLLDQPSA HLDPVTYQII RRTLKQAFAD CTVILCEHRI EAMLECQQFL VIEENKVRQY DSIQKLLNER SLFRQAI SP SDRVKLFPHR NSSKCKSKPQ IAALKEETEE EVQDTRL UniProtKB: Cystic fibrosis transmembrane conductance regulator |
-Macromolecule #2: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 2 / Number of copies: 2 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #3: ADENOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 3 / Number of copies: 2 / Formula: ATP |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-Macromolecule #4: CHOLESTEROL
| Macromolecule | Name: CHOLESTEROL / type: ligand / ID: 4 / Number of copies: 1 / Formula: CLR |
|---|---|
| Molecular weight | Theoretical: 386.654 Da |
| Chemical component information |  ChemComp-CLR: |
-Macromolecule #5: (6P)-N-(1,3-dimethyl-1H-pyrazole-4-sulfonyl)-6-[3-(3,3,3-trifluor...
| Macromolecule | Name: (6P)-N-(1,3-dimethyl-1H-pyrazole-4-sulfonyl)-6-[3-(3,3,3-trifluoro-2,2-dimethylpropoxy)-1H-pyrazol-1-yl]-2-[(4S)-2,2,4-trimethylpyrrolidin-1-yl]pyridine-3-carboxamide type: ligand / ID: 5 / Number of copies: 1 / Formula: WJX |
|---|---|
| Molecular weight | Theoretical: 597.653 Da |
| Chemical component information | 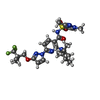 ChemComp-WJX: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 65.6 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)