+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Mycobacterial respiratory complex I, fully-inserted quinone | |||||||||||||||
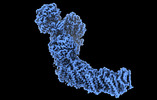
 Map data Map data | ||||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | Complex / Oxidative Phosphorylation / NADH-quinone oxidoreductase / Iron-sulfur Protein / MEMBRANE PROTEIN | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationNADH dehydrogenase (quinone) / NADH dehydrogenase (quinone) (non-electrogenic) activity / Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions / NADH dehydrogenase complex / molybdopterin cofactor binding / cellular respiration / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / phosphorelay signal transduction system / ubiquinone binding / electron transport coupled proton transport ...NADH dehydrogenase (quinone) / NADH dehydrogenase (quinone) (non-electrogenic) activity / Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions / NADH dehydrogenase complex / molybdopterin cofactor binding / cellular respiration / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / phosphorelay signal transduction system / ubiquinone binding / electron transport coupled proton transport / NADH dehydrogenase activity / respiratory chain complex I / NADH dehydrogenase (ubiquinone) activity / quinone binding / ATP synthesis coupled electron transport / endomembrane system / aerobic respiration / 2 iron, 2 sulfur cluster binding / NAD binding / FMN binding / 4 iron, 4 sulfur cluster binding / iron ion binding / membrane / metal ion binding / plasma membrane Similarity search - Function | |||||||||||||||
| Biological species |  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria) | |||||||||||||||
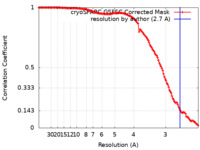
| Method | single particle reconstruction / cryo EM / Resolution: 2.7 Å | |||||||||||||||
 Authors Authors | Liang Y / Rubinstein JL | |||||||||||||||
| Funding support |  Canada, 4 items Canada, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2023 Journal: Proc Natl Acad Sci U S A / Year: 2023Title: Structure of mycobacterial respiratory complex I. Authors: Yingke Liang / Alicia Plourde / Stephanie A Bueler / Jun Liu / Peter Brzezinski / Siavash Vahidi / John L Rubinstein /   Abstract: Oxidative phosphorylation, the combined activity of the electron transport chain (ETC) and adenosine triphosphate synthase, has emerged as a valuable target for the treatment of infection by and ...Oxidative phosphorylation, the combined activity of the electron transport chain (ETC) and adenosine triphosphate synthase, has emerged as a valuable target for the treatment of infection by and other mycobacteria. The mycobacterial ETC is highly branched with multiple dehydrogenases transferring electrons to a membrane-bound pool of menaquinone and multiple oxidases transferring electrons from the pool. The proton-pumping type I nicotinamide adenine dinucleotide (NADH) dehydrogenase (Complex I) is found in low abundance in the plasma membranes of mycobacteria in typical in vitro culture conditions and is often considered dispensable. We found that growth of in carbon-limited conditions greatly increased the abundance of Complex I and allowed isolation of a rotenone-sensitive preparation of the enzyme. Determination of the structure of the complex by cryoEM revealed the "orphan" two-component response regulator protein MSMEG_2064 as a subunit of the assembly. MSMEG_2064 in the complex occupies a site similar to the proposed redox-sensing subunit NDUFA9 in eukaryotic Complex I. An apparent purine nucleoside triphosphate within the NuoG subunit resembles the GTP-derived molybdenum cofactor in homologous formate dehydrogenase enzymes. The membrane region of the complex binds acyl phosphatidylinositol dimannoside, a characteristic three-tailed lipid from the mycobacterial membrane. The structure also shows menaquinone, which is preferentially used over ubiquinone by gram-positive bacteria, in two different positions along the quinone channel, comparable to ubiquinone in other structures and suggesting a conserved quinone binding mechanism. #1:  Journal: Biorxiv / Year: 2022 Journal: Biorxiv / Year: 2022Title: Structure of mycobacterial respiratory Complex I Authors: Liang Y / Plourde A / Bueler SA / Liu J / Brzezinski P / Vahidi S / Rubinstein JL | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27964.map.gz emd_27964.map.gz | 108.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27964-v30.xml emd-27964-v30.xml emd-27964.xml emd-27964.xml | 41.4 KB 41.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_27964_fsc.xml emd_27964_fsc.xml | 12.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_27964.png emd_27964.png | 62.4 KB | ||
| Filedesc metadata |  emd-27964.cif.gz emd-27964.cif.gz | 10.5 KB | ||
| Others |  emd_27964_half_map_1.map.gz emd_27964_half_map_1.map.gz emd_27964_half_map_2.map.gz emd_27964_half_map_2.map.gz | 200.5 MB 200.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27964 http://ftp.pdbj.org/pub/emdb/structures/EMD-27964 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27964 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27964 | HTTPS FTP |
-Related structure data
| Related structure data |  8e9hMC  8e9gC  8e9iC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_27964.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27964.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.18021 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_27964_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_27964_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Mycobacterial respiratory complex I, fully-inserted quinone
+Supramolecule #1: Mycobacterial respiratory complex I, fully-inserted quinone
+Macromolecule #1: Two-component system response regulator
+Macromolecule #2: NADH-quinone oxidoreductase subunit B
+Macromolecule #3: NADH-quinone oxidoreductase subunit A
+Macromolecule #4: NADH-quinone oxidoreductase subunit C
+Macromolecule #5: NADH-quinone oxidoreductase subunit D
+Macromolecule #6: NADH-quinone oxidoreductase subunit E
+Macromolecule #7: NADH-quinone oxidoreductase subunit G
+Macromolecule #8: NADH-quinone oxidoreductase subunit F
+Macromolecule #9: NADH-quinone oxidoreductase subunit I
+Macromolecule #10: NADH-quinone oxidoreductase subunit H
+Macromolecule #11: NADH-quinone oxidoreductase subunit J
+Macromolecule #12: NADH-quinone oxidoreductase subunit K
+Macromolecule #13: NADH-quinone oxidoreductase, L subunit
+Macromolecule #14: NADH-quinone oxidoreductase subunit N
+Macromolecule #15: NADH-quinone oxidoreductase, M subunit
+Macromolecule #16: IRON/SULFUR CLUSTER
+Macromolecule #17: MENAQUINONE-9
+Macromolecule #18: FE2/S2 (INORGANIC) CLUSTER
+Macromolecule #19: GUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #20: FLAVIN MONONUCLEOTIDE
+Macromolecule #21: ZINC ION
+Macromolecule #22: (2R)-3-{[(R)-hydroxy({(1S,2R,3R,4R,5S,6S)-3,4,5-trihydroxy-2-(alp...
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL |
|---|---|
| Buffer | pH: 6 |
| Grid | Model: Homemade / Material: COPPER/RHODIUM / Mesh: 400 / Support film - Material: GOLD / Support film - topology: HOLEY / Support film - Film thickness: 36 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: LEICA EM GP |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 47.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.2 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 75000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)