+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of TRPV5 in detergent in the presence of PKA | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Calcium / Ion channel / Kidney / MEMBRANE PROTEIN | |||||||||
| Biological species |   | |||||||||
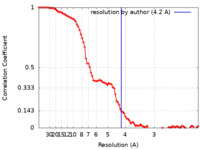
| Method | single particle reconstruction / cryo EM / Resolution: 4.2 Å | |||||||||
 Authors Authors | Fluck EC / Yazici AT / Rohacs T / Moiseenkova-Bell VY | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2022 Journal: Cell Rep / Year: 2022Title: Structural basis of TRPV5 regulation by physiological and pathophysiological modulators. Authors: Edwin C Fluck / Aysenur Torun Yazici / Tibor Rohacs / Vera Y Moiseenkova-Bell /  Abstract: Transient receptor potential vanilloid 5 (TRPV5) is a kidney-specific Ca-selective ion channel that plays a key role in Ca homeostasis. The basal activity of TRPV5 is balanced through activation by ...Transient receptor potential vanilloid 5 (TRPV5) is a kidney-specific Ca-selective ion channel that plays a key role in Ca homeostasis. The basal activity of TRPV5 is balanced through activation by phosphatidylinositol 4,5-bisphosphate (PI(4,5)P) and inhibition by Ca-bound calmodulin (CaM). Parathyroid hormone (PTH), the key extrinsic regulator of Ca homeostasis, increases the activity of TRPV5 via protein kinase A (PKA)-mediated phosphorylation. Metabolic acidosis leads to reduced TRPV5 activity independent of PTH, causing hypercalciuria. Using cryoelectron microscopy (cryo-EM), we show that low pH inhibits TRPV5 by precluding PI(4,5)P activation. We capture intermediate conformations at low pH, revealing a transition from open to closed state. In addition, we demonstrate that PI(4,5)P is the primary modulator of channel gating, yet PKA controls TRPV5 activity by preventing CaM binding and channel inactivation. Our data provide detailed molecular mechanisms for regulation of TRPV5 by two key extrinsic modulators, low pH and PKA. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_25722.map.gz emd_25722.map.gz | 59.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-25722-v30.xml emd-25722-v30.xml emd-25722.xml emd-25722.xml | 15.2 KB 15.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_25722_fsc.xml emd_25722_fsc.xml | 8.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_25722.png emd_25722.png | 70.1 KB | ||
| Filedesc metadata |  emd-25722.cif.gz emd-25722.cif.gz | 5.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-25722 http://ftp.pdbj.org/pub/emdb/structures/EMD-25722 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25722 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25722 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_25722.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_25722.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Tetrameric assembly of TRPV5 ion channel reconstituted in deterge...
| Entire | Name: Tetrameric assembly of TRPV5 ion channel reconstituted in detergent with catalytic subunit of PKA |
|---|---|
| Components |
|
-Supramolecule #1: Tetrameric assembly of TRPV5 ion channel reconstituted in deterge...
| Supramolecule | Name: Tetrameric assembly of TRPV5 ion channel reconstituted in detergent with catalytic subunit of PKA type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Transient receptor potential cation channel subfamily V member 5
| Macromolecule | Name: Transient receptor potential cation channel subfamily V member 5 type: protein_or_peptide / ID: 1 / Details: Rabbit TRPV5 with c-terminal 1D4 epitope tag / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MGACPPKAKG PWAQLQKLLI SWPVGEQDWE QYRDRVNMLQ QERIRDSPLL QAAKENDLRL LKILLLNQSC DFQQRGAVGE TALHVAALY DNLEAATLLM EAAPELAKEP ALCEPFVGQT ALHIAVMNQN LNLVRALLAR GASVSARATG AAFRRSPHNL I YYGEHPLS ...String: MGACPPKAKG PWAQLQKLLI SWPVGEQDWE QYRDRVNMLQ QERIRDSPLL QAAKENDLRL LKILLLNQSC DFQQRGAVGE TALHVAALY DNLEAATLLM EAAPELAKEP ALCEPFVGQT ALHIAVMNQN LNLVRALLAR GASVSARATG AAFRRSPHNL I YYGEHPLS FAACVGSEEI VRLLIEHGAD IRAQDSLGNT VLHILILQPN KTFACQMYNL LLSYDEHSDH LQSLELVPNH QG LTPFKLA GVEGNTVMFQ HLMQKRKHVQ WTCGPLTSTL YDLTEIDSWG EELSFLELVV SSKKREARQI LEQTPVKELV SFK WKKYGR PYFCVLASLY ILYMICFTTC CIYRPLKLRD DNRTDPRDIT ILQQKLLQEA YVTHQDNIRL VGELVTVTGA VIIL LLEIP DIFRVGASRY FGQTILGGPF HVIIITYASL VLLTMVMRLT NMNGEVVPLS FALVLGWCSV MYFARGFQML GPFTI MIQK MIFGDLMRFC WLMAVVILGF ASAFHITFQT EDPNNLGEFS DYPTALFSTF ELFLTIIDGP ANYSVDLPFM YCITYA AFA IIATLLMLNL FIAMMGDTHW RVAQERDELW RAQVVATTVM LERKMPRFLW PRSGICGYEY GLGDRWFLRV ENHHDQN PL RVLRYVEAFK CSDKEDGQEQ LSEKRPSTVE SGMLSRASVA FQTPSLSRTT SQSSNSHRGW EILRRNTLGH LNLGLDLG E GDGEEVYHFT ETSQVAPA |
-Macromolecule #2: Protein kinase A catalytic subunit
| Macromolecule | Name: Protein kinase A catalytic subunit / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MGSSHHHHHH SSGLVPRGSH MGNAAAAKKG SEQESVKEFL AKAKEDFLKK WETPSQNTAQ LDQFDRIKTL GTGSFGRVML VKHKESGNHY AMKILDKQKV VKLKQIEHTL NEKRILQAVN FPFLVKLEFS FKDNSNLYMV MEYVAGGEMF SHLRRIGRFS EPHARFYAAQ ...String: MGSSHHHHHH SSGLVPRGSH MGNAAAAKKG SEQESVKEFL AKAKEDFLKK WETPSQNTAQ LDQFDRIKTL GTGSFGRVML VKHKESGNHY AMKILDKQKV VKLKQIEHTL NEKRILQAVN FPFLVKLEFS FKDNSNLYMV MEYVAGGEMF SHLRRIGRFS EPHARFYAAQ IVLTFEYLHS LDLIYRDLKP ENLLIDQQGY IQVTDFGFAK RVKGRTWTLC GTPEYLAPEI ILSKGYNKAV DWWALGVLIY EMAAGYPPFF ADQPIQIYEK IVSGKVRFPS HFSSDLKDLL RNLLQVDLTK RFGNLKNGVN DIKNHKWFAT TDWIAIYQRK VEAPFIPKFK GPGDTSNFDD YEEEEIRVSI NEKCGKEFTE F |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Support film - Film thickness: 12 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Number grids imaged: 2 / Number real images: 12801 / Average electron dose: 72.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.2 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)