+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Allosteric Regulation of Human Plastins | |||||||||
 Map data Map data | F-actin/Plastin 3-ABD1 | |||||||||
 Sample Sample |
| |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 5.1 Å | |||||||||
 Authors Authors | Zheng W / Schwebach CL / Kudryashova E / Egelman EH / Kudryashov DS | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2022 Journal: Nat Struct Mol Biol / Year: 2022Title: Allosteric regulation controls actin-bundling properties of human plastins. Authors: Christopher L Schwebach / Elena Kudryashova / Richa Agrawal / Weili Zheng / Edward H Egelman / Dmitri S Kudryashov /  Abstract: Plastins/fimbrins are conserved actin-bundling proteins contributing to motility, cytokinesis and other cellular processes by organizing strikingly different actin assemblies as in aligned bundles ...Plastins/fimbrins are conserved actin-bundling proteins contributing to motility, cytokinesis and other cellular processes by organizing strikingly different actin assemblies as in aligned bundles and branched networks. We propose that this ability of human plastins stems from an allosteric communication between their actin-binding domains (ABD1/2) engaged in a tight spatial association. Here we show that ABD2 can bind actin three orders of magnitude stronger than ABD1, unless the domains are involved in an equally strong inhibitory engagement. A mutation mimicking physiologically relevant phosphorylation at the ABD1-ABD2 interface greatly weakened their association, dramatically potentiating actin cross-linking. Cryo-EM reconstruction revealed the ABD1-actin interface and enabled modeling of the plastin bridge and domain separation in parallel bundles. We predict that a strong and tunable allosteric inhibition between the domains allows plastins to modulate the cross-linking strength, contributing to remodeling of actin assemblies of different morphologies defining the unique place of plastins in actin organization. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_25371.map.gz emd_25371.map.gz | 10.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-25371-v30.xml emd-25371-v30.xml emd-25371.xml emd-25371.xml | 10.6 KB 10.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_25371.png emd_25371.png | 52.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-25371 http://ftp.pdbj.org/pub/emdb/structures/EMD-25371 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25371 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25371 | HTTPS FTP |
-Validation report
| Summary document |  emd_25371_validation.pdf.gz emd_25371_validation.pdf.gz | 350.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_25371_full_validation.pdf.gz emd_25371_full_validation.pdf.gz | 349.9 KB | Display | |
| Data in XML |  emd_25371_validation.xml.gz emd_25371_validation.xml.gz | 6.3 KB | Display | |
| Data in CIF |  emd_25371_validation.cif.gz emd_25371_validation.cif.gz | 7.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25371 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25371 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25371 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25371 | HTTPS FTP |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_25371.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_25371.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | F-actin/Plastin 3-ABD1 | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.4 Å | ||||||||||||||||||||||||||||||||||||
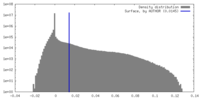
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Complex of Plastin3-ABD1 with F-actin
| Entire | Name: Complex of Plastin3-ABD1 with F-actin |
|---|---|
| Components |
|
-Supramolecule #1: Complex of Plastin3-ABD1 with F-actin
| Supramolecule | Name: Complex of Plastin3-ABD1 with F-actin / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|
-Macromolecule #1: Actin, alpha skeletal muscle
| Macromolecule | Name: Actin, alpha skeletal muscle / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Sequence | String: MCDEDETTAL VCDNGSGLVK AGFAGDDAPR AVFPSIVGRP RHQGVMVGMG QKDSYVGDEA QSKRGILTL KYPIEHGIIT NWDDMEKIWH HTFYNELRVA PEEHPTLLTE APLNPKANRE K MTQIMFET FNVPAMYVAI QAVLSLYASG RTTGIVLDSG DGVTHNVPIY ...String: MCDEDETTAL VCDNGSGLVK AGFAGDDAPR AVFPSIVGRP RHQGVMVGMG QKDSYVGDEA QSKRGILTL KYPIEHGIIT NWDDMEKIWH HTFYNELRVA PEEHPTLLTE APLNPKANRE K MTQIMFET FNVPAMYVAI QAVLSLYASG RTTGIVLDSG DGVTHNVPIY EGYALPHAIM RL DLAGRDL TDYLMKILTE RGYSFVTTAE REIVRDIKEK LCYVALDFEN EMATAASSSS LEK SYELPD GQVITIGNER FRCPETLFQP SFIGMESAGI HETTYNSIMK CDIDIRKDLY ANNV MSGGT TMYPGIADRM QKEITALAPS TMKIKIIAPP ERKYSVWIGG SILASLSTFQ QMWIT KQEY DEAGPSIVHR KCF |
-Macromolecule #2: Plastin 3
| Macromolecule | Name: Plastin 3 / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MDEMATTQIS KDELDELKEA FAKVDLNSNG FICDYELHEL FKEANMPLPG YKVREIIQKL MLDGDRNKDG KISFDEFVYI FQEVKSSDIA KTFRKAINRK EGICALGGTS ELSSEGTQHS YSEEEKYAFV NWINKALEND PDCRHVIPMN PNTDDLFKAV GDGIVLCKMI ...String: MDEMATTQIS KDELDELKEA FAKVDLNSNG FICDYELHEL FKEANMPLPG YKVREIIQKL MLDGDRNKDG KISFDEFVYI FQEVKSSDIA KTFRKAINRK EGICALGGTS ELSSEGTQHS YSEEEKYAFV NWINKALEND PDCRHVIPMN PNTDDLFKAV GDGIVLCKMI NLSVPDTIDE RAINKKKLTP FIIQENLNLA LNSASAIGCH VVNIGAEDLR AGKPHLVLGL LWQIIKIGLF ADIELSRNEA LAALLRDGET LEELMKLSPE ELLLRWANFH LENSGWQKIN NFSADIKDSK AYFHLLNQIA PKGQKEGEPR IDINMSGFNE TDDLKRAESM LQQADKLGCR QFVTPADVVS GNPKLNLAFV ANLFNKYPAL TKPENQDIDW TLLEGETREE RTFRNWMNSL GVNPHVNHLY ADLQDALVIL QLYERIKVPV DWSKVNKPPY PKLGANMKKL ENCNYAVELG KHPAKFSLVG IGGQDLNDGN QTLTLALVWQ LMRRYTLNVL EDLGDGQKAN DDIIVNWVNR TLSEAGKSTS IQSFKDKTIS SSLAVVDLID AIQPGCINYD LVKSGNLTED DKHNNAKYAV SMARRIGARV YALPEDLVEV KPKMVMTVFA CLMGRGMKRV |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Model: PELCO Ultrathin Carbon with Lacey Carbon / Pretreatment - Type: PLASMA CLEANING |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average electron dose: 20.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 28.0 Å Applied symmetry - Helical parameters - Δ&Phi: -166.5 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Resolution.type: BY AUTHOR / Resolution: 5.1 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION / Number images used: 155939 |
|---|---|
| CTF correction | Software - Name: RELION |
| Final angle assignment | Type: NOT APPLICABLE / Software - Name: RELION |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)