+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | RUVBL1/2 in complex with ATP | |||||||||
 Map data Map data | CryoEM map (Refine3D) for RUVBL1/2-ATP complex | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | RUVBL1 / RUVBL2 / CB-6644 / Inhibitor / Chaperone | |||||||||
| Function / homology |  Function and homology information Function and homology informationpromoter-enhancer loop anchoring activity / telomerase RNA localization to Cajal body / regulation of DNA strand elongation / positive regulation of telomere maintenance in response to DNA damage / establishment of protein localization to chromatin / R2TP complex / dynein axonemal particle / RPAP3/R2TP/prefoldin-like complex / Swr1 complex / Ino80 complex ...promoter-enhancer loop anchoring activity / telomerase RNA localization to Cajal body / regulation of DNA strand elongation / positive regulation of telomere maintenance in response to DNA damage / establishment of protein localization to chromatin / R2TP complex / dynein axonemal particle / RPAP3/R2TP/prefoldin-like complex / Swr1 complex / Ino80 complex / regulation of double-strand break repair / box C/D snoRNP assembly / regulation of chromosome organization / NuA4 histone acetyltransferase complex / regulation of DNA replication / MLL1 complex / TFIID-class transcription factor complex binding / regulation of embryonic development / Telomere Extension By Telomerase / protein folding chaperone complex / regulation of DNA repair / RNA polymerase II core promoter sequence-specific DNA binding / positive regulation of double-strand break repair via homologous recombination / Deposition of new CENPA-containing nucleosomes at the centromere / TBP-class protein binding / telomere maintenance / positive regulation of DNA repair / DNA helicase activity / cellular response to estradiol stimulus / euchromatin / negative regulation of canonical Wnt signaling pathway / Formation of the beta-catenin:TCF transactivating complex / ADP binding / chromatin DNA binding / beta-catenin binding / DNA Damage Recognition in GG-NER / nuclear matrix / cellular response to UV / : / UCH proteinases / transcription corepressor activity / positive regulation of canonical Wnt signaling pathway / nucleosome / HATs acetylate histones / ATPase binding / protein folding / DNA recombination / regulation of apoptotic process / spermatogenesis / DNA helicase / transcription coactivator activity / regulation of cell cycle / Ub-specific processing proteases / nuclear speck / protein stabilization / ciliary basal body / RNA polymerase II cis-regulatory region sequence-specific DNA binding / cadherin binding / chromatin remodeling / ribonucleoprotein complex / cell division / DNA repair / regulation of DNA-templated transcription / regulation of transcription by RNA polymerase II / centrosome / positive regulation of DNA-templated transcription / protein homodimerization activity / ATP hydrolysis activity / positive regulation of transcription by RNA polymerase II / extracellular exosome / nucleoplasm / ATP binding / membrane / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
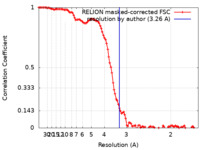
| Method | single particle reconstruction / cryo EM / Resolution: 3.26 Å | |||||||||
 Authors Authors | Lopez-Perrote A / Llorca O / Garcia-Martin C | |||||||||
| Funding support |  Spain, 1 items Spain, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Rep Phys Sci / Year: 2024 Journal: Cell Rep Phys Sci / Year: 2024Title: Mechanism of allosteric inhibition of RUVBL1-RUVBL2 by the small-molecule CB-6644 Authors: Garcia-Martin C / Lopez-Perrote A / Boskovic J / Llorca O | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_19817.map.gz emd_19817.map.gz | 79.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-19817-v30.xml emd-19817-v30.xml emd-19817.xml emd-19817.xml | 22.6 KB 22.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_19817_fsc.xml emd_19817_fsc.xml | 10.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_19817.png emd_19817.png | 585.7 KB | ||
| Filedesc metadata |  emd-19817.cif.gz emd-19817.cif.gz | 7.1 KB | ||
| Others |  emd_19817_additional_1.map.gz emd_19817_additional_1.map.gz emd_19817_half_map_1.map.gz emd_19817_half_map_1.map.gz emd_19817_half_map_2.map.gz emd_19817_half_map_2.map.gz | 10 MB 78.9 MB 78.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-19817 http://ftp.pdbj.org/pub/emdb/structures/EMD-19817 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19817 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19817 | HTTPS FTP |
-Related structure data
| Related structure data |  9emcMC  9emaC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_19817.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_19817.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | CryoEM map (Refine3D) for RUVBL1/2-ATP complex | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.8272 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Sharpened map (Postprocess) for RUVBL1/2-ATP complex
| File | emd_19817_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map (Postprocess) for RUVBL1/2-ATP complex | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 1 (unfiltered) for RUVBL1/2-ATP complex
| File | emd_19817_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 (unfiltered) for RUVBL1/2-ATP complex | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 2 (unfiltered) for RUVBL1/2-ATP complex
| File | emd_19817_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 (unfiltered) for RUVBL1/2-ATP complex | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Hetero-hexameric RUVBL1-RUVBL2 complex bound to ATP and the CB-66...
| Entire | Name: Hetero-hexameric RUVBL1-RUVBL2 complex bound to ATP and the CB-6644 inhibitor |
|---|---|
| Components |
|
-Supramolecule #1: Hetero-hexameric RUVBL1-RUVBL2 complex bound to ATP and the CB-66...
| Supramolecule | Name: Hetero-hexameric RUVBL1-RUVBL2 complex bound to ATP and the CB-6644 inhibitor type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 310.47 kDa/nm |
-Macromolecule #1: RuvB-like 1
| Macromolecule | Name: RuvB-like 1 / type: protein_or_peptide / ID: 1 Details: Sequence contains three extra aminoacids (GSH) at the N-terminus due to protease cleavage that do not affect the structure and activity of the protein Number of copies: 3 / Enantiomer: LEVO / EC number: DNA helicase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 50.579188 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSHMKIEEVK STTKTQRIAS HSHVKGLGLD ESGLAKQAAS GLVGQENARE ACGVIVELIK SKKMAGRAVL LAGPPGTGKT ALALAIAQE LGSKVPFCPM VGSEVYSTEI KKTEVLMENF RRAIGLRIKE TKEVYEGEVT ELTPCETENP MGGYGKTISH V IIGLKTAK ...String: GSHMKIEEVK STTKTQRIAS HSHVKGLGLD ESGLAKQAAS GLVGQENARE ACGVIVELIK SKKMAGRAVL LAGPPGTGKT ALALAIAQE LGSKVPFCPM VGSEVYSTEI KKTEVLMENF RRAIGLRIKE TKEVYEGEVT ELTPCETENP MGGYGKTISH V IIGLKTAK GTKQLKLDPS IFESLQKERV EAGDVIYIEA NSGAVKRQGR CDTYATEFDL EAEEYVPLPK GDVHKKKEII QD VTLHDLD VANARPQGGQ DILSMMGQLM KPKKTEITDK LRGEINKVVN KYIDQGIAEL VPGVLFVDEV HMLDIECFTY LHR ALESSI APIVIFASNR GNCVIRGTED ITSPHGIPLD LLDRVMIIRT MLYTPQEMKQ IIKIRAQTEG INISEEALNH LGEI GTKTT LRYSVQLLTP ANLLAKINGK DSIEKEHVEE ISELFYDAKS SAKILADQQD KYMK UniProtKB: RuvB-like 1 |
-Macromolecule #2: RuvB-like 2
| Macromolecule | Name: RuvB-like 2 / type: protein_or_peptide / ID: 2 Details: A strectch of 18 extra residues is present at the N-terminus due to cloning design that do not affect the structure and activity of the protein Number of copies: 3 / Enantiomer: LEVO / EC number: DNA helicase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 53.047488 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MADLNWISAG HAIADVGTMA TVTATTKVPE IRDVTRIERI GAHSHIRGLG LDDALEPRQA SQGMVGQLAA RRAAGVVLEM IREGKIAGR AVLIAGQPGT GKTAIAMGMA QALGPDTPFT AIAGSEIFSL EMSKTEALTQ AFRRSIGVRI KEETEIIEGE V VEIQIDRP ...String: MADLNWISAG HAIADVGTMA TVTATTKVPE IRDVTRIERI GAHSHIRGLG LDDALEPRQA SQGMVGQLAA RRAAGVVLEM IREGKIAGR AVLIAGQPGT GKTAIAMGMA QALGPDTPFT AIAGSEIFSL EMSKTEALTQ AFRRSIGVRI KEETEIIEGE V VEIQIDRP ATGTGSKVGK LTLKTTEMET IYDLGTKMIE SLTKDKVQAG DVITIDKATG KISKLGRSFT RARDYDAMGS QT KFVQCPD GELQKRKEVV HTVSLHEIDV INSRTQGFLA LFSGDTGEIK SEVREQINAK VAEWREEGKA EIIPGVLFID EVH MLDIES FSFLNRALES DMAPVLIMAT NRGITRIRGT SYQSPHGIPI DLLDRLLIVS TTPYSEKDTK QILRIRCEEE DVEM SEDAY TVLTRIGLET SLRYAIQLIT AASLVCRKRK GTEVQVDDIK RVYSLFLDES RSTQYMKEYQ DAFLFNELKG ETMDT S UniProtKB: RuvB-like 2 |
-Macromolecule #3: ADENOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 3 / Number of copies: 6 / Formula: ATP |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-Macromolecule #4: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 4 / Number of copies: 6 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.8 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| |||||||||||||||
| Grid | Model: Quantifoil R0.6/1 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 10.0 kPa | |||||||||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 90 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOCONTINUUM (6k x 4k) / Average electron dose: 45.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.8000000000000003 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | Chain - Source name: Other / Chain - Initial model type: in silico model / Details: ModelAngelo |
|---|---|
| Refinement | Space: REAL / Protocol: BACKBONE TRACE |
| Output model |  PDB-9emc: |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)