+ Open data
Open data
- Basic information
Basic information
| Entry | 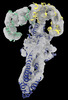 | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | GBP1 bound by 14-3-3sigma | ||||||||||||||||||||||||||||||
 Map data Map data | |||||||||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||||||||
 Keywords Keywords | Protein complex / Phosphorylation / IMMUNE SYSTEM | ||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationGDP phosphatase activity / non-canonical inflammasome complex assembly / protein localization to vacuole / negative regulation of substrate adhesion-dependent cell spreading / symbiont cell surface / cytolysis in another organism / positive regulation of pyroptotic inflammatory response / vesicle membrane / negative regulation of protein localization to plasma membrane / negative regulation of interleukin-2 production ...GDP phosphatase activity / non-canonical inflammasome complex assembly / protein localization to vacuole / negative regulation of substrate adhesion-dependent cell spreading / symbiont cell surface / cytolysis in another organism / positive regulation of pyroptotic inflammatory response / vesicle membrane / negative regulation of protein localization to plasma membrane / negative regulation of interleukin-2 production / negative regulation of T cell receptor signaling pathway / spectrin binding / regulation of epidermal cell division / protein kinase C inhibitor activity / cytokine binding / positive regulation of epidermal cell differentiation / keratinocyte development / keratinization / defense response to protozoan / Hydrolases; Acting on acid anhydrides; In phosphorus-containing anhydrides / Regulation of localization of FOXO transcription factors / keratinocyte proliferation / phosphoserine residue binding / Activation of BAD and translocation to mitochondria / negative regulation of keratinocyte proliferation / regulation of protein localization to plasma membrane / establishment of skin barrier / cellular response to interleukin-1 / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / negative regulation of stem cell proliferation / regulation of calcium-mediated signaling / RHO GTPases activate PKNs / protein kinase A signaling / protein sequestering activity / negative regulation of innate immune response / protein export from nucleus / TP53 Regulates Transcription of Genes Involved in G2 Cell Cycle Arrest / positive regulation of protein export from nucleus / release of cytochrome c from mitochondria / G protein activity / stem cell proliferation / Translocation of SLC2A4 (GLUT4) to the plasma membrane / lipopolysaccharide binding / TP53 Regulates Metabolic Genes / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / negative regulation of protein kinase activity / Hsp90 protein binding / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process / cytoplasmic vesicle membrane / negative regulation of ERK1 and ERK2 cascade / cellular response to type II interferon / GDP binding / intrinsic apoptotic signaling pathway in response to DNA damage / Interferon gamma signaling / actin cytoskeleton / cellular response to tumor necrosis factor / actin binding / cytoplasmic vesicle / positive regulation of cell growth / defense response to virus / regulation of cell cycle / defense response to bacterium / cadherin binding / Golgi membrane / innate immune response / GTPase activity / GTP binding / protein kinase binding / Golgi apparatus / enzyme binding / negative regulation of transcription by RNA polymerase II / signal transduction / protein homodimerization activity / extracellular space / extracellular exosome / extracellular region / identical protein binding / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 5.12 Å | ||||||||||||||||||||||||||||||
 Authors Authors | Pfleiderer MM / Liu X / Fisch D / Anastasakou E / Frickel EM / Galej WP | ||||||||||||||||||||||||||||||
| Funding support |  Germany, Germany,  France, European Union, France, European Union,  United Kingdom, 9 items United Kingdom, 9 items
| ||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Science / Year: 2023 Journal: Science / Year: 2023Title: PIM1 controls GBP1 activity to limit self-damage and to guard against pathogen infection. Authors: Daniel Fisch / Moritz M Pfleiderer / Eleni Anastasakou / Gillian M Mackie / Fabian Wendt / Xiangyang Liu / Barbara Clough / Samuel Lara-Reyna / Vesela Encheva / Ambrosius P Snijders / ...Authors: Daniel Fisch / Moritz M Pfleiderer / Eleni Anastasakou / Gillian M Mackie / Fabian Wendt / Xiangyang Liu / Barbara Clough / Samuel Lara-Reyna / Vesela Encheva / Ambrosius P Snijders / Hironori Bando / Masahiro Yamamoto / Andrew D Beggs / Jason Mercer / Avinash R Shenoy / Bernd Wollscheid / Kendle M Maslowski / Wojtek P Galej / Eva-Maria Frickel /      Abstract: Disruption of cellular activities by pathogen virulence factors can trigger innate immune responses. Interferon-γ (IFN-γ)-inducible antimicrobial factors, such as the guanylate binding proteins ...Disruption of cellular activities by pathogen virulence factors can trigger innate immune responses. Interferon-γ (IFN-γ)-inducible antimicrobial factors, such as the guanylate binding proteins (GBPs), promote cell-intrinsic defense by attacking intracellular pathogens and by inducing programmed cell death. Working in human macrophages, we discovered that GBP1 expression in the absence of IFN-γ killed the cells and induced Golgi fragmentation. IFN-γ exposure improved macrophage survival through the activity of the kinase PIM1. PIM1 phosphorylated GBP1, leading to its sequestration by 14-3-3σ, which thereby prevented GBP1 membrane association. During infection, the virulence protein TgIST interfered with IFN-γ signaling and depleted PIM1, thereby increasing GBP1 activity. Although infected cells can restrain pathogens in a GBP1-dependent manner, this mechanism can protect uninfected bystander cells. Thus, PIM1 can provide a bait for pathogen virulence factors, guarding the integrity of IFN-γ signaling. | ||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_18149.map.gz emd_18149.map.gz | 401.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-18149-v30.xml emd-18149-v30.xml emd-18149.xml emd-18149.xml | 20 KB 20 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_18149.png emd_18149.png | 90.9 KB | ||
| Filedesc metadata |  emd-18149.cif.gz emd-18149.cif.gz | 6.7 KB | ||
| Others |  emd_18149_half_map_1.map.gz emd_18149_half_map_1.map.gz emd_18149_half_map_2.map.gz emd_18149_half_map_2.map.gz | 763.8 MB 763.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-18149 http://ftp.pdbj.org/pub/emdb/structures/EMD-18149 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18149 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18149 | HTTPS FTP |
-Validation report
| Summary document |  emd_18149_validation.pdf.gz emd_18149_validation.pdf.gz | 937 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_18149_full_validation.pdf.gz emd_18149_full_validation.pdf.gz | 936.5 KB | Display | |
| Data in XML |  emd_18149_validation.xml.gz emd_18149_validation.xml.gz | 21.2 KB | Display | |
| Data in CIF |  emd_18149_validation.cif.gz emd_18149_validation.cif.gz | 25.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18149 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18149 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18149 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18149 | HTTPS FTP |
-Related structure data
| Related structure data |  8q4lMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_18149.map.gz / Format: CCP4 / Size: 824 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_18149.map.gz / Format: CCP4 / Size: 824 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.638 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
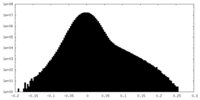
-Half map: #2
| File | emd_18149_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
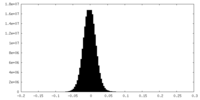
| Density Histograms |
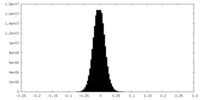
-Half map: #1
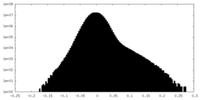
| File | emd_18149_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : GBP1 bound by 14-3-3sigma
| Entire | Name: GBP1 bound by 14-3-3sigma |
|---|---|
| Components |
|
-Supramolecule #1: GBP1 bound by 14-3-3sigma
| Supramolecule | Name: GBP1 bound by 14-3-3sigma / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 120 KDa |
-Macromolecule #1: Guanylate-binding protein 1
| Macromolecule | Name: Guanylate-binding protein 1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 66.275539 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTGPMCLIEN TNGRLMANPE ALKILSAITQ PMVVVAIVGL YRTGKSYLMN KLAGKKKGFS LGSTVQSHTK GIWMWCVPHP KKPGHILVL LDTEGLGDVE KGDNQNDSWI FALAVLLSST FVYNSIGTIN QQAMDQLYYV TELTHRIRSK SSPDENENEV E DSADFVSF ...String: MTGPMCLIEN TNGRLMANPE ALKILSAITQ PMVVVAIVGL YRTGKSYLMN KLAGKKKGFS LGSTVQSHTK GIWMWCVPHP KKPGHILVL LDTEGLGDVE KGDNQNDSWI FALAVLLSST FVYNSIGTIN QQAMDQLYYV TELTHRIRSK SSPDENENEV E DSADFVSF FPDFVWTLRD FSLDLEADGQ PLTPDEYLTY SLKLKKGTSQ KDETFNLPRL CIRKFFPKKK CFVFDRPVHR RK LAQLEKL QDEELDPEFV QQVADFCSYI FSNSKTKTLS GGIQVNGPRL ESLVLTYVNA ISSGDLPCME NAVLALAQIE NSA AVQKAI AHYEQQMGQK VQLPTETLQE LLDLHRDSER EAIEVFIRSS FKDVDHLFQK ELAAQLEKKR DDFCKQNQEA SSDR CSALL QVIFSPLEEE VKAGIYSKPG GYRLFVQKLQ DLKKKYYEEP RKGIQAEEIL QTYLKSKESM TDAILQTDQT LTEKE KEIE VERVKAESAQ ASAKMLQEMQ RKNEQMMEQK ERSYQEHLKQ LTEKMENDRV QLLKEQERTL ALKLQEQEQL LKEGFQ KES RIMKNEIQDL QTKM UniProtKB: Guanylate-binding protein 1 |
-Macromolecule #2: 14-3-3 protein sigma
| Macromolecule | Name: 14-3-3 protein sigma / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 26.139461 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MERASLIQKA KLAEQAERYE DMAAFMKGAV EKGEELSCEE RNLLSVAYKN VVGGQRAAWR VLSSIEQKSN EEGSEEKGPE VREYREKVE TELQGVCDTV LGLLDSHLIK EAGDAESRVF YLKMKGDYYR YLAEVATGDD KKRIIDSARS AYQEAMDISK K EMPPTNPI ...String: MERASLIQKA KLAEQAERYE DMAAFMKGAV EKGEELSCEE RNLLSVAYKN VVGGQRAAWR VLSSIEQKSN EEGSEEKGPE VREYREKVE TELQGVCDTV LGLLDSHLIK EAGDAESRVF YLKMKGDYYR YLAEVATGDD KKRIIDSARS AYQEAMDISK K EMPPTNPI RLGLALNFSV FHYEIANSPE EAISLAKTTF DEAMADLHTL SEDSYKDSTL IMQLLRDNLT LWT UniProtKB: 14-3-3 protein sigma |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.25 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.8 Component:
Details: 150 mM KCl, 20 mM HEPES-KOH pH 7.8 | |||||||||
| Grid | Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 20 sec. / Pretreatment - Pressure: 30.0 kPa Details: Glow discharged for 20 seconds at 25 mA and 0.3 bar using a Pelco EasyGlow device. | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 14974 / Average exposure time: 8.0 sec. / Average electron dose: 65.5 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



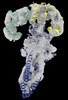











 Z
Z Y
Y X
X