[English] 日本語
 Yorodumi
Yorodumi- EMDB-16389: Cryo-EM structure of photosystem II C2S2 supercomplex from Norway... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of photosystem II C2S2 supercomplex from Norway spruce (Picea babies) at 2.8 Angstrom resolution | ||||||||||||||||||||||||||||||
 Map data Map data | |||||||||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||||||||
 Keywords Keywords | Membrane protein complex / Photosynthesis / Photosystem / Gymnosperm / MEMBRANE PROTEIN | ||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationphotosynthesis, light harvesting / photosystem II oxygen evolving complex / photosystem II assembly / oxygen evolving activity / photosystem II stabilization / photosystem II / photosystem II reaction center / photosystem I / oxidoreductase activity, acting on diphenols and related substances as donors, oxygen as acceptor / photosynthetic electron transport chain ...photosynthesis, light harvesting / photosystem II oxygen evolving complex / photosystem II assembly / oxygen evolving activity / photosystem II stabilization / photosystem II / photosystem II reaction center / photosystem I / oxidoreductase activity, acting on diphenols and related substances as donors, oxygen as acceptor / photosynthetic electron transport chain / response to herbicide / photosystem II / chlorophyll binding / photosynthesis, light reaction / electron transporter, transferring electrons within the cyclic electron transport pathway of photosynthesis activity / phosphate ion binding / chloroplast thylakoid membrane / photosynthetic electron transport in photosystem II / photosynthesis / electron transfer activity / protein stabilization / iron ion binding / heme binding / metal ion binding Similarity search - Function | ||||||||||||||||||||||||||||||
| Biological species |  | ||||||||||||||||||||||||||||||
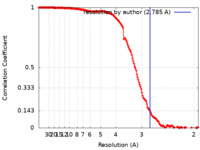
| Method | single particle reconstruction / cryo EM / Resolution: 2.785 Å | ||||||||||||||||||||||||||||||
 Authors Authors | Kopecny D / Semchonok DA / Kouril R | ||||||||||||||||||||||||||||||
| Funding support |  Czech Republic, Czech Republic,  Germany, 9 items Germany, 9 items
| ||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Plants / Year: 2023 Journal: Nat Plants / Year: 2023Title: Cryo-EM structure of a plant photosystem II supercomplex with light-harvesting protein Lhcb8 and α-tocopherol. Authors: Monika Opatíková / Dmitry A Semchonok / David Kopečný / Petr Ilík / Pavel Pospíšil / Iva Ilíková / Pavel Roudnický / Sanja Ćavar Zeljković / Petr Tarkowski / Fotis L Kyrilis / ...Authors: Monika Opatíková / Dmitry A Semchonok / David Kopečný / Petr Ilík / Pavel Pospíšil / Iva Ilíková / Pavel Roudnický / Sanja Ćavar Zeljković / Petr Tarkowski / Fotis L Kyrilis / Farzad Hamdi / Panagiotis L Kastritis / Roman Kouřil /    Abstract: The heart of oxygenic photosynthesis is the water-splitting photosystem II (PSII), which forms supercomplexes with a variable amount of peripheral trimeric light-harvesting complexes (LHCII). Our ...The heart of oxygenic photosynthesis is the water-splitting photosystem II (PSII), which forms supercomplexes with a variable amount of peripheral trimeric light-harvesting complexes (LHCII). Our knowledge of the structure of green plant PSII supercomplex is based on findings obtained from several representatives of green algae and flowering plants; however, data from a non-flowering plant are currently missing. Here we report a cryo-electron microscopy structure of PSII supercomplex from spruce, a representative of non-flowering land plants, at 2.8 Å resolution. Compared with flowering plants, PSII supercomplex in spruce contains an additional Ycf12 subunit, Lhcb4 protein is replaced by Lhcb8, and trimeric LHCII is present as a homotrimer of Lhcb1. Unexpectedly, we have found α-tocopherol (α-Toc)/α-tocopherolquinone (α-TQ) at the boundary between the LHCII trimer and the inner antenna CP43. The molecule of α-Toc/α-TQ is located close to chlorophyll a614 of one of the Lhcb1 proteins and its chromanol/quinone head is exposed to the thylakoid lumen. The position of α-Toc in PSII supercomplex makes it an ideal candidate for the sensor of excessive light, as α-Toc can be oxidized to α-TQ by high-light-induced singlet oxygen at low lumenal pH. The molecule of α-TQ appears to shift slightly into the PSII supercomplex, which could trigger important structure-functional modifications in PSII supercomplex. Inspection of the previously reported cryo-electron microscopy maps of PSII supercomplexes indicates that α-Toc/α-TQ can be present at the same site also in PSII supercomplexes from flowering plants, but its identification in the previous studies has been hindered by insufficient resolution. | ||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_16389.map.gz emd_16389.map.gz | 323.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-16389-v30.xml emd-16389-v30.xml emd-16389.xml emd-16389.xml | 52.9 KB 52.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_16389_fsc.xml emd_16389_fsc.xml | 15.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_16389.png emd_16389.png | 91.8 KB | ||
| Masks |  emd_16389_msk_1.map emd_16389_msk_1.map | 343 MB |  Mask map Mask map | |
| Others |  emd_16389_half_map_1.map.gz emd_16389_half_map_1.map.gz emd_16389_half_map_2.map.gz emd_16389_half_map_2.map.gz | 318.3 MB 318.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-16389 http://ftp.pdbj.org/pub/emdb/structures/EMD-16389 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16389 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16389 | HTTPS FTP |
-Validation report
| Summary document |  emd_16389_validation.pdf.gz emd_16389_validation.pdf.gz | 1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_16389_full_validation.pdf.gz emd_16389_full_validation.pdf.gz | 1 MB | Display | |
| Data in XML |  emd_16389_validation.xml.gz emd_16389_validation.xml.gz | 24.2 KB | Display | |
| Data in CIF |  emd_16389_validation.cif.gz emd_16389_validation.cif.gz | 31.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16389 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16389 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16389 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16389 | HTTPS FTP |
-Related structure data
| Related structure data |  8c29MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_16389.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_16389.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.96127 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_16389_msk_1.map emd_16389_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
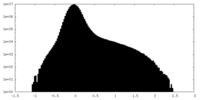
| Density Histograms |
-Half map: #1
| File | emd_16389_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
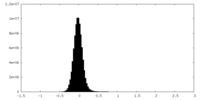
| Density Histograms |
-Half map: #2
| File | emd_16389_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : The C2S2-type PSII-LHCII supercomplex
+Supramolecule #1: The C2S2-type PSII-LHCII supercomplex
+Supramolecule #2: Photosystem II core complex (C2)
+Supramolecule #3: Photosystem II light-harvesting complex
+Macromolecule #1: Photosystem II protein D1
+Macromolecule #2: Photosystem II CP47 reaction center protein
+Macromolecule #3: Photosystem II CP43 reaction center protein
+Macromolecule #4: Photosystem II D2 protein
+Macromolecule #5: Cytochrome b559 subunit alpha
+Macromolecule #6: Cytochrome b559 subunit beta
+Macromolecule #7: Chlorophyll a-b binding protein, chloroplastic
+Macromolecule #8: Photosystem II reaction center protein H
+Macromolecule #9: Photosystem II reaction center protein I
+Macromolecule #10: Photosystem II reaction center protein K
+Macromolecule #11: Photosystem II reaction center protein L
+Macromolecule #12: Photosystem II reaction center protein M
+Macromolecule #13: Oxygen-evolving enhancer protein 1, chloroplastic
+Macromolecule #14: Chlorophyll a-b binding protein, chloroplastic
+Macromolecule #15: Chlorophyll a-b binding protein, chloroplastic
+Macromolecule #16: Photosystem II reaction center protein T
+Macromolecule #17: Photosystem II 5 kDa protein, chloroplastic
+Macromolecule #18: Photosystem II reaction center protein Ycf12
+Macromolecule #19: PSII 6.1 kDa protein
+Macromolecule #20: Photosystem II PsbX
+Macromolecule #21: Photosystem II reaction center protein Z
+Macromolecule #22: FE (II) ION
+Macromolecule #23: CHLOROPHYLL A
+Macromolecule #24: PHEOPHYTIN A
+Macromolecule #25: BETA-CAROTENE
+Macromolecule #26: 1,2-DI-O-ACYL-3-O-[6-DEOXY-6-SULFO-ALPHA-D-GLUCOPYRANOSYL]-SN-GLYCEROL
+Macromolecule #27: 1,2-DISTEAROYL-MONOGALACTOSYL-DIGLYCERIDE
+Macromolecule #28: 1,2-DIACYL-GLYCEROL-3-SN-PHOSPHATE
+Macromolecule #29: 2,3-DIMETHYL-5-(3,7,11,15,19,23,27,31,35-NONAMETHYL-2,6,10,14,18,...
+Macromolecule #30: 1,2-DIPALMITOYL-PHOSPHATIDYL-GLYCEROLE
+Macromolecule #31: ALPHA-LINOLENIC ACID
+Macromolecule #32: PALMITOLEIC ACID
+Macromolecule #33: DIGALACTOSYL DIACYL GLYCEROL (DGDG)
+Macromolecule #34: MAGNESIUM ION
+Macromolecule #35: (2R)-2,5,7,8-TETRAMETHYL-2-[(4R,8R)-4,8,12-TRIMETHYLTRIDECYL]CHRO...
+Macromolecule #36: BICARBONATE ION
+Macromolecule #37: DIACYL GLYCEROL
+Macromolecule #38: PROTOPORPHYRIN IX CONTAINING FE
+Macromolecule #39: CHLOROPHYLL B
+Macromolecule #40: (3R,3'R,6S)-4,5-DIDEHYDRO-5,6-DIHYDRO-BETA,BETA-CAROTENE-3,3'-DIOL
+Macromolecule #41: (1R,3R)-6-{(3E,5E,7E,9E,11E,13E,15E,17E)-18-[(1S,4R,6R)-4-HYDROXY...
+Macromolecule #42: (3S,5R,6S,3'S,5'R,6'S)-5,6,5',6'-DIEPOXY-5,6,5',6'- TETRAHYDRO-BE...
+Macromolecule #43: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| |||||||||
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 25 sec. / Pretreatment - Atmosphere: AIR / Details: PELCO easiGlow, 15 mA | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV | |||||||||
| Details | 3 mg of chlorophylls/ml |
- Electron microscopy
Electron microscopy
| Microscope | TFS GLACIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 4096 pixel / Digitization - Dimensions - Height: 4096 pixel / Number grids imaged: 1 / Number real images: 2392 / Average electron dose: 120.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Specimen holder model: OTHER / Cooling holder cryogen: NITROGEN |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-8c29: |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)