[English] 日本語
 Yorodumi
Yorodumi- EMDB-15979: Masked refinement of the IFTB2 subcomplex within anterograde intr... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Masked refinement of the IFTB2 subcomplex within anterograde intraflagellar trains in Chlamydomonas reinhardtii cilia | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Cilia / IFT / Intraflagellar / transport / PROTEIN TRANSPORT | |||||||||
| Biological species |  | |||||||||
| Method | subtomogram averaging / cryo EM / Resolution: 11.5 Å | |||||||||
 Authors Authors | Lacey SE / Pigino G | |||||||||
| Funding support | European Union,  Italy, 2 items Italy, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: The molecular structure of IFT-A and IFT-B in anterograde intraflagellar transport trains. Authors: Samuel E Lacey / Helen E Foster / Gaia Pigino /  Abstract: Anterograde intraflagellar transport (IFT) trains are essential for cilia assembly and maintenance. These trains are formed of 22 IFT-A and IFT-B proteins that link structural and signaling cargos to ...Anterograde intraflagellar transport (IFT) trains are essential for cilia assembly and maintenance. These trains are formed of 22 IFT-A and IFT-B proteins that link structural and signaling cargos to microtubule motors for import into cilia. It remains unknown how the IFT-A/-B proteins are arranged into complexes and how these complexes polymerize into functional trains. Here we use in situ cryo-electron tomography of Chlamydomonas reinhardtii cilia and AlphaFold2 protein structure predictions to generate a molecular model of the entire anterograde train. We show how the conformations of both IFT-A and IFT-B are dependent on lateral interactions with neighboring repeats, suggesting that polymerization is required to cooperatively stabilize the complexes. Following three-dimensional classification, we reveal how IFT-B extends two flexible tethers to maintain a connection with IFT-A that can withstand the mechanical stresses present in actively beating cilia. Overall, our findings provide a framework for understanding the fundamental processes that govern cilia assembly. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15979.map.gz emd_15979.map.gz | 2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15979-v30.xml emd-15979-v30.xml emd-15979.xml emd-15979.xml | 16.9 KB 16.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_15979.png emd_15979.png | 42.9 KB | ||
| Filedesc metadata |  emd-15979.cif.gz emd-15979.cif.gz | 4.3 KB | ||
| Others |  emd_15979_additional_1.map.gz emd_15979_additional_1.map.gz emd_15979_half_map_1.map.gz emd_15979_half_map_1.map.gz emd_15979_half_map_2.map.gz emd_15979_half_map_2.map.gz | 1.2 MB 27.1 MB 27.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15979 http://ftp.pdbj.org/pub/emdb/structures/EMD-15979 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15979 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15979 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_15979.map.gz / Format: CCP4 / Size: 35.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15979.map.gz / Format: CCP4 / Size: 35.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 4.04 Å | ||||||||||||||||||||||||||||||||||||
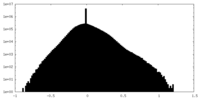
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Masked classification of the periphery of IFTB2 complex,...
| File | emd_15979_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Masked classification of the periphery of IFTB2 complex, in the region corrresponding to IFT172 (WD domains) and IFT57 CH domain. Lowpass filtered to 18 angstrom and a b factor of -1300 A^2 applied. | ||||||||||||
| Projections & Slices |
| ||||||||||||
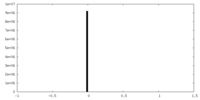
| Density Histograms |
-Half map: #1
| File | emd_15979_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
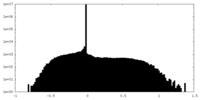
| Density Histograms |
-Half map: #2
| File | emd_15979_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
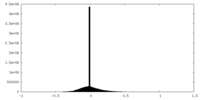
| Density Histograms |
- Sample components
Sample components
-Entire : Two IFTB repeats from anterograde intraflagellar transport trains...
| Entire | Name: Two IFTB repeats from anterograde intraflagellar transport trains within native Chlamydomonas reinhardtii cilia |
|---|---|
| Components |
|
-Supramolecule #1: Two IFTB repeats from anterograde intraflagellar transport trains...
| Supramolecule | Name: Two IFTB repeats from anterograde intraflagellar transport trains within native Chlamydomonas reinhardtii cilia type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#13 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | subtomogram averaging |
| Aggregation state | helical array |
- Sample preparation
Sample preparation
| Buffer | pH: 7 / Details: TAP (Tris-Acetate-Phosphate) Media |
|---|---|
| Vitrification | Cryogen name: ETHANE |
| Details | Chlamydomonas reinhardtii cells applied to quantifoil grids and plunge frozen; cilia project out from cell bodies and traverse holes in the carbon film. |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 2.6 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 5.0 µm / Nominal defocus min: 2.5 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 11.5 Å / Resolution method: FSC 0.143 CUT-OFF / Software: (Name: RELION (ver. 3.1.3), Warp) / Number subtomograms used: 18216 |
|---|---|
| Extraction | Number tomograms: 600 / Number images used: 18216 |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
-Atomic model buiding 1
| Refinement | Protocol: FLEXIBLE FIT |
|---|
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)