+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Masked refinement of the IFTB1 subcomplex within anterograde intraflagellar trains in Chlamydomonas reinhardtii cilia | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Cilia / IFT / Intraflagellar / transport / PROTEIN TRANSPORT | |||||||||
| 生物種 |  | |||||||||
| 手法 | サブトモグラム平均法 / クライオ電子顕微鏡法 / 解像度: 9.9 Å | |||||||||
 データ登録者 データ登録者 | Lacey SE / Pigino G | |||||||||
| 資金援助 | European Union,  イタリア, 2件 イタリア, 2件
| |||||||||
 引用 引用 |  ジャーナル: Nat Struct Mol Biol / 年: 2023 ジャーナル: Nat Struct Mol Biol / 年: 2023タイトル: The molecular structure of IFT-A and IFT-B in anterograde intraflagellar transport trains. 著者: Samuel E Lacey / Helen E Foster / Gaia Pigino /  要旨: Anterograde intraflagellar transport (IFT) trains are essential for cilia assembly and maintenance. These trains are formed of 22 IFT-A and IFT-B proteins that link structural and signaling cargos to ...Anterograde intraflagellar transport (IFT) trains are essential for cilia assembly and maintenance. These trains are formed of 22 IFT-A and IFT-B proteins that link structural and signaling cargos to microtubule motors for import into cilia. It remains unknown how the IFT-A/-B proteins are arranged into complexes and how these complexes polymerize into functional trains. Here we use in situ cryo-electron tomography of Chlamydomonas reinhardtii cilia and AlphaFold2 protein structure predictions to generate a molecular model of the entire anterograde train. We show how the conformations of both IFT-A and IFT-B are dependent on lateral interactions with neighboring repeats, suggesting that polymerization is required to cooperatively stabilize the complexes. Following three-dimensional classification, we reveal how IFT-B extends two flexible tethers to maintain a connection with IFT-A that can withstand the mechanical stresses present in actively beating cilia. Overall, our findings provide a framework for understanding the fundamental processes that govern cilia assembly. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_15978.map.gz emd_15978.map.gz | 3.8 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-15978-v30.xml emd-15978-v30.xml emd-15978.xml emd-15978.xml | 17.1 KB 17.1 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_15978.png emd_15978.png | 74.7 KB | ||
| マスクデータ |  emd_15978_msk_1.map emd_15978_msk_1.map | 64 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-15978.cif.gz emd-15978.cif.gz | 4.4 KB | ||
| その他 |  emd_15978_additional_1.map.gz emd_15978_additional_1.map.gz emd_15978_half_map_1.map.gz emd_15978_half_map_1.map.gz emd_15978_half_map_2.map.gz emd_15978_half_map_2.map.gz | 1.5 MB 49.5 MB 49.5 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15978 http://ftp.pdbj.org/pub/emdb/structures/EMD-15978 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15978 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15978 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_15978.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_15978.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 3.03 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-マスク #1
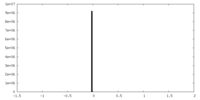
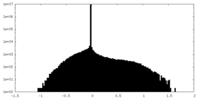
| ファイル |  emd_15978_msk_1.map emd_15978_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: A masked refinement on the IFTB1 periphery region,...
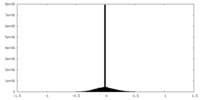
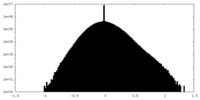
| ファイル | emd_15978_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | A masked refinement on the IFTB1 periphery region, containing the regions corresponding to IFT56 and IFT52/46 heterodimer. Lowpassed to 16 angstrom and with a -1600 A^2 bfactor | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_15978_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #2
| ファイル | emd_15978_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Two IFTB repeats from anterograde intraflagellar transport trains...
| 全体 | 名称: Two IFTB repeats from anterograde intraflagellar transport trains within native Chlamydomonas reinhardtii cilia |
|---|---|
| 要素 |
|
-超分子 #1: Two IFTB repeats from anterograde intraflagellar transport trains...
| 超分子 | 名称: Two IFTB repeats from anterograde intraflagellar transport trains within native Chlamydomonas reinhardtii cilia タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#13 |
|---|---|
| 由来(天然) | 生物種:  |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | サブトモグラム平均法 |
| 試料の集合状態 | helical array |
- 試料調製
試料調製
| 緩衝液 | pH: 7 / 詳細: TAP (Tris-Acetate-Phosphate) Media |
|---|---|
| 凍結 | 凍結剤: ETHANE |
| 詳細 | Chlamydomonas reinhardtii cells applied to quantifoil grids and plunge frozen; cilia project out from cell bodies and traverse holes in the carbon film. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | TFS KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: FEI FALCON IV (4k x 4k) 平均電子線量: 2.6 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 5.0 µm / 最小 デフォーカス(公称値): 2.5 µm |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 最終 再構成 | 想定した対称性 - 点群: C1 (非対称) / 解像度のタイプ: BY AUTHOR / 解像度: 9.9 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア: (名称: RELION (ver. 3.1.3), Warp) / 使用したサブトモグラム数: 18216 |
|---|---|
| 抽出 | トモグラム数: 600 / 使用した粒子像数: 18216 |
| 最終 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
-原子モデル構築 1
| 精密化 | プロトコル: FLEXIBLE FIT |
|---|
 ムービー
ムービー コントローラー
コントローラー










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)