[English] 日本語
 Yorodumi
Yorodumi- EMDB-15484: Structure of human DDB1-DCAF12 in complex with the C-terminus of CCT5 -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of human DDB1-DCAF12 in complex with the C-terminus of CCT5 | |||||||||
 Map data Map data | Locally sharpened map (LocScale). | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ubiquitin / E3 / TRiC / chaperonin / LIGASE | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of protein localization to Cajal body / positive regulation of telomerase RNA localization to Cajal body / chaperonin-containing T-complex / BBSome-mediated cargo-targeting to cilium / Formation of tubulin folding intermediates by CCT/TriC / binding of sperm to zona pellucida / Folding of actin by CCT/TriC / positive regulation by virus of viral protein levels in host cell / Prefoldin mediated transfer of substrate to CCT/TriC / spindle assembly involved in female meiosis ...positive regulation of protein localization to Cajal body / positive regulation of telomerase RNA localization to Cajal body / chaperonin-containing T-complex / BBSome-mediated cargo-targeting to cilium / Formation of tubulin folding intermediates by CCT/TriC / binding of sperm to zona pellucida / Folding of actin by CCT/TriC / positive regulation by virus of viral protein levels in host cell / Prefoldin mediated transfer of substrate to CCT/TriC / spindle assembly involved in female meiosis / epigenetic programming in the zygotic pronuclei / UV-damage excision repair / ubiquitin-dependent protein catabolic process via the C-end degron rule pathway / biological process involved in interaction with symbiont / regulation of mitotic cell cycle phase transition / WD40-repeat domain binding / Cul4A-RING E3 ubiquitin ligase complex / Cul4-RING E3 ubiquitin ligase complex / Cul4B-RING E3 ubiquitin ligase complex / Association of TriC/CCT with target proteins during biosynthesis / ubiquitin ligase complex scaffold activity / negative regulation of reproductive process / negative regulation of developmental process / viral release from host cell / cullin family protein binding / ectopic germ cell programmed cell death / positive regulation of viral genome replication / Hydrolases; Acting on acid anhydrides; In phosphorus-containing anhydrides / beta-tubulin binding / ubiquitin-like ligase-substrate adaptor activity / positive regulation of telomere maintenance via telomerase / proteasomal protein catabolic process / protein folding chaperone / positive regulation of gluconeogenesis / T cell activation / regulation of autophagy / nucleotide-excision repair / mRNA 3'-UTR binding / ATP-dependent protein folding chaperone / sperm end piece / Recognition of DNA damage by PCNA-containing replication complex / regulation of circadian rhythm / DNA Damage Recognition in GG-NER / mRNA 5'-UTR binding / Dual Incision in GG-NER / Transcription-Coupled Nucleotide Excision Repair (TC-NER) / Formation of TC-NER Pre-Incision Complex / Wnt signaling pathway / response to virus / Formation of Incision Complex in GG-NER / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / positive regulation of protein catabolic process / cellular response to UV / : / rhythmic process / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / G-protein beta-subunit binding / site of double-strand break / sperm principal piece / protein folding / Neddylation / cell body / sperm midpiece / ubiquitin-dependent protein catabolic process / damaged DNA binding / microtubule / proteasome-mediated ubiquitin-dependent protein catabolic process / protein-macromolecule adaptor activity / chromosome, telomeric region / protein stabilization / protein ubiquitination / DNA repair / apoptotic process / DNA damage response / centrosome / negative regulation of apoptotic process / protein-containing complex binding / nucleolus / ATP hydrolysis activity / protein-containing complex / : / DNA binding / extracellular exosome / nucleoplasm / ATP binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.83 Å | |||||||||
 Authors Authors | Pla-Prats C / Cavadini S / Kempf G / Thoma NH | |||||||||
| Funding support | European Union, 1 items
| |||||||||
 Citation Citation |  Journal: EMBO J / Year: 2023 Journal: EMBO J / Year: 2023Title: Recognition of the CCT5 di-Glu degron by CRL4 is dependent on TRiC assembly. Authors: Carlos Pla-Prats / Simone Cavadini / Georg Kempf / Nicolas H Thomä /  Abstract: Assembly Quality Control (AQC) E3 ubiquitin ligases target incomplete or incorrectly assembled protein complexes for degradation. The CUL4-RBX1-DDB1-DCAF12 (CRL4 ) E3 ligase preferentially ...Assembly Quality Control (AQC) E3 ubiquitin ligases target incomplete or incorrectly assembled protein complexes for degradation. The CUL4-RBX1-DDB1-DCAF12 (CRL4 ) E3 ligase preferentially ubiquitinates proteins that carry a C-terminal double glutamate (di-Glu) motif. Reported CRL4 di-Glu-containing substrates include CCT5, a subunit of the TRiC chaperonin. How DCAF12 engages its substrates and the functional relationship between CRL4 and CCT5/TRiC is currently unknown. Here, we present the cryo-EM structure of the DDB1-DCAF12-CCT5 complex at 2.8 Å resolution. DCAF12 serves as a canonical WD40 DCAF substrate receptor and uses a positively charged pocket at the center of the β-propeller to bind the C-terminus of CCT5. DCAF12 specifically reads out the CCT5 di-Glu side chains, and contacts other visible degron amino acids through Van der Waals interactions. The CCT5 C-terminus is inaccessible in an assembled TRiC complex, and functional assays demonstrate that DCAF12 binds and ubiquitinates monomeric CCT5, but not CCT5 assembled into TRiC. Our biochemical and structural results suggest a previously unknown role for the CRL4 E3 ligase in overseeing the assembly of a key cellular complex. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15484.map.gz emd_15484.map.gz | 41.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15484-v30.xml emd-15484-v30.xml emd-15484.xml emd-15484.xml | 19.9 KB 19.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_15484.png emd_15484.png | 69.8 KB | ||
| Filedesc metadata |  emd-15484.cif.gz emd-15484.cif.gz | 6.7 KB | ||
| Others |  emd_15484_additional_1.map.gz emd_15484_additional_1.map.gz emd_15484_half_map_1.map.gz emd_15484_half_map_1.map.gz emd_15484_half_map_2.map.gz emd_15484_half_map_2.map.gz | 65.3 MB 65.5 MB 65.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15484 http://ftp.pdbj.org/pub/emdb/structures/EMD-15484 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15484 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15484 | HTTPS FTP |
-Related structure data
| Related structure data |  8ajmMC  8ajnC  8ajoC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_15484.map.gz / Format: CCP4 / Size: 83.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15484.map.gz / Format: CCP4 / Size: 83.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Locally sharpened map (LocScale). | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.86 Å | ||||||||||||||||||||||||||||||||||||
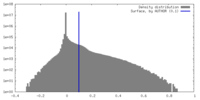
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Full map.
| File | emd_15484_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Full map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
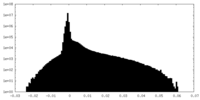
| Density Histograms |
-Half map: #2
| File | emd_15484_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
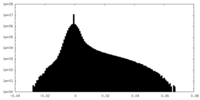
| Density Histograms |
-Half map: #1
| File | emd_15484_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of DDB1-DCAF12-CCT5
| Entire | Name: Complex of DDB1-DCAF12-CCT5 |
|---|---|
| Components |
|
-Supramolecule #1: Complex of DDB1-DCAF12-CCT5
| Supramolecule | Name: Complex of DDB1-DCAF12-CCT5 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: DNA damage-binding protein 1
| Macromolecule | Name: DNA damage-binding protein 1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 129.784258 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGSSHHHHHH SAVDENLYFQ GGGRMSYNYV VTAQKPTAVN GCVTGHFTSA EDLNLLIAKN TRLEIYVVTA EGLRPVKEVG MYGKIAVME LFRPKGESKD LLFILTAKYN ACILEYKQSG ESIDIITRAH GNVQDRIGRP SETGIIGIID PECRMIGLRL Y DGLFKVIP ...String: MGSSHHHHHH SAVDENLYFQ GGGRMSYNYV VTAQKPTAVN GCVTGHFTSA EDLNLLIAKN TRLEIYVVTA EGLRPVKEVG MYGKIAVME LFRPKGESKD LLFILTAKYN ACILEYKQSG ESIDIITRAH GNVQDRIGRP SETGIIGIID PECRMIGLRL Y DGLFKVIP LDRDNKELKA FNIRLEELHV IDVKFLYGCQ APTICFVYQD PQGRHVKTYE VSLREKEFNK GPWKQENVEA EA SMVIAVP EPFGGAIIIG QESITYHNGD KYLAIAPPII KQSTIVCHNR VDPNGSRYLL GDMEGRLFML LLEKEEQMDG TVT LKDLRV ELLGETSIAE CLTYLDNGVV FVGSRLGDSQ LVKLNVDSNE QGSYVVAMET FTNLGPIVDM CVVDLERQGQ GQLV TCSGA FKEGSLRIIR NGIGIHEHAS IDLPGIKGLW PLRSDPNRET DDTLVLSFVG QTRVLMLNGE EVEETELMGF VDDQQ TFFC GNVAHQQLIQ ITSASVRLVS QEPKALVSEW KEPQAKNISV ASCNSSQVVV AVGRALYYLQ IHPQELRQIS HTEMEH EVA CLDITPLGDS NGLSPLCAIG LWTDISARIL KLPSFELLHK EMLGGEIIPR SILMTTFESS HYLLCALGDG ALFYFGL NI ETGLLSDRKK VTLGTQPTVL RTFRSLSTTN VFACSDRPTV IYSSNHKLVF SNVNLKEVNY MCPLNSDGYP DSLALANN S TLTIGTIDEI QKLHIRTVPL YESPRKICYQ EVSQCFGVLS SRIEVQDTSG GTTALRPSAS TQALSSSVSS SKLFSSSTA PHETSFGEEV EVHNLLIIDQ HTFEVLHAHQ FLQNEYALSL VSCKLGKDPN TYFIVGTAMV YPEEAEPKQG RIVVFQYSDG KLQTVAEKE VKGAVYSMVE FNGKLLASIN STVRLYEWTT EKELRTECNH YNNIMALYLK TKGDFILVGD LMRSVLLLAY K PMEGNFEE IARDFNPNWM SAVEILDDDN FLGAENAFNL FVCQKDSAAT TDEERQHLQE VGLFHLGEFV NVFCHGSLVM QN LGETSTP TQGSVLFGTV NGMIGLVTSL SESWYNLLLD MQNRLNKVIK SVGKIEHSFW RSFHTERKTE PATGFIDGDL IES FLDISR PKMQEVVANL QYDDGSGMKR EATADDLIKV VEELTRIH UniProtKB: DNA damage-binding protein 1 |
-Macromolecule #2: DDB1- and CUL4-associated factor 12
| Macromolecule | Name: DDB1- and CUL4-associated factor 12 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 53.373191 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MDWSHPQFEK SAVDENLYFQ GGGRMARKVV SRKRKAPASP GAGSDAQGPQ FGWDHSLHKR KRLPPVKRSL VYYLKNREVR LQNETSYSR VLHGYAAQQL PSLLKEREFH LGTLNKVFAS QWLNHRQVVC GTKCNTLFVV DVQTSQITKI PILKDREPGG V TQQGCGIH ...String: MDWSHPQFEK SAVDENLYFQ GGGRMARKVV SRKRKAPASP GAGSDAQGPQ FGWDHSLHKR KRLPPVKRSL VYYLKNREVR LQNETSYSR VLHGYAAQQL PSLLKEREFH LGTLNKVFAS QWLNHRQVVC GTKCNTLFVV DVQTSQITKI PILKDREPGG V TQQGCGIH AIELNPSRTL LATGGDNPNS LAIYRLPTLD PVCVGDDGHK DWIFSIAWIS DTMAVSGSRD GSMGLWEVTD DV LTKSDAR HNVSRVPVYA HITHKALKDI PKEDTNPDNC KVRALAFNNK NKELGAVSLD GYFHLWKAEN TLSKLLSTKL PYC RENVCL AYGSEWSVYA VGSQAHVSFL DPRQPSYNVK SVCSRERGSG IRSVSFYEHI ITVGTGQGSL LFYDIRAQRF LEER LSACY GSKPRLAGEN LKLTTGKGWL NHDETWRNYF SDIDFFPNAV YTHCYDSSGT KLFVAGGPLP SGLHGNYAGL WS UniProtKB: DDB1- and CUL4-associated factor 12 |
-Macromolecule #3: T-complex protein 1 subunit epsilon
| Macromolecule | Name: T-complex protein 1 subunit epsilon / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 62.436809 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGSSHHHHHH SAVDENLYFQ GGGRMASMGT LAFDEYGRPF LIIKDQDRKS RLMGLEALKS HIMAAKAVAN TMRTSLGPNG LDKMMVDKD GDVTVTNDGA TILSMMDVDH QIAKLMVELS KSQDDEIGDG TTGVVVLAGA LLEEAEQLLD RGIHPIRIAD G YEQAARVA ...String: MGSSHHHHHH SAVDENLYFQ GGGRMASMGT LAFDEYGRPF LIIKDQDRKS RLMGLEALKS HIMAAKAVAN TMRTSLGPNG LDKMMVDKD GDVTVTNDGA TILSMMDVDH QIAKLMVELS KSQDDEIGDG TTGVVVLAGA LLEEAEQLLD RGIHPIRIAD G YEQAARVA IEHLDKISDS VLVDIKDTEP LIQTAKTTLG SKVVNSCHRQ MAEIAVNAVL TVADMERRDV DFELIKVEGK VG GRLEDTK LIKGVIVDKD FSHPQMPKKV EDAKIAILTC PFEPPKPKTK HKLDVTSVED YKALQKYEKE KFEEMIQQIK ETG ANLAIC QWGFDDEANH LLLQNNLPAV RWVGGPEIEL IAIATGGRIV PRFSELTAEK LGFAGLVQEI SFGTTKDKML VIEQ CKNSR AVTIFIRGGN KMIIEEAKRS LHDALCVIRN LIRDNRVVYG GGAAEISCAL AVSQEADKCP TLEQYAMRAF ADALE VIPM ALSENSGMNP IQTMTEVRAR QVKEMNPALG IDCLHKGTND MKQQHVIETL IGKKQQISLA TQMVRMILKI DDIRKP GES EE UniProtKB: T-complex protein 1 subunit epsilon |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 51.8 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 2.83 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION / Number images used: 451315 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION |
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)