[English] 日本語
 Yorodumi
Yorodumi- EMDB-12713: Structural basis for VIPP1 oligomerization and maintenance of thy... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12713 | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structural basis for VIPP1 oligomerization and maintenance of thylakoid membrane integrity | ||||||||||||||||||||||||
 Map data Map data | C17 refined map | ||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||
| Function / homology | phage shock / PspA/IM30 / PspA/IM30 family / plasma membrane / cytosol / Membrane-associated protein Vipp1 Function and homology information Function and homology information | ||||||||||||||||||||||||
| Biological species |  | ||||||||||||||||||||||||
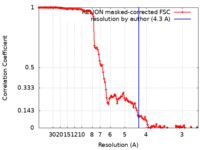
| Method | single particle reconstruction / cryo EM / Resolution: 4.3 Å | ||||||||||||||||||||||||
 Authors Authors | Gupta TK / Klumpe S / Gries K / Strauss M / Rudack T / Schuller JM / Schroda M / Engel BD | ||||||||||||||||||||||||
| Funding support |  Germany, Germany,  Japan, 7 items Japan, 7 items
| ||||||||||||||||||||||||
 Citation Citation |  Journal: Cell / Year: 2021 Journal: Cell / Year: 2021Title: Structural basis for VIPP1 oligomerization and maintenance of thylakoid membrane integrity. Authors: Tilak Kumar Gupta / Sven Klumpe / Karin Gries / Steffen Heinz / Wojciech Wietrzynski / Norikazu Ohnishi / Justus Niemeyer / Benjamin Spaniol / Miroslava Schaffer / Anna Rast / Matthias ...Authors: Tilak Kumar Gupta / Sven Klumpe / Karin Gries / Steffen Heinz / Wojciech Wietrzynski / Norikazu Ohnishi / Justus Niemeyer / Benjamin Spaniol / Miroslava Schaffer / Anna Rast / Matthias Ostermeier / Mike Strauss / Jürgen M Plitzko / Wolfgang Baumeister / Till Rudack / Wataru Sakamoto / Jörg Nickelsen / Jan M Schuller / Michael Schroda / Benjamin D Engel /    Abstract: Vesicle-inducing protein in plastids 1 (VIPP1) is essential for the biogenesis and maintenance of thylakoid membranes, which transform light into life. However, it is unknown how VIPP1 performs its ...Vesicle-inducing protein in plastids 1 (VIPP1) is essential for the biogenesis and maintenance of thylakoid membranes, which transform light into life. However, it is unknown how VIPP1 performs its vital membrane-remodeling functions. Here, we use cryo-electron microscopy to determine structures of cyanobacterial VIPP1 rings, revealing how VIPP1 monomers flex and interweave to form basket-like assemblies of different symmetries. Three VIPP1 monomers together coordinate a non-canonical nucleotide binding pocket on one end of the ring. Inside the ring's lumen, amphipathic helices from each monomer align to form large hydrophobic columns, enabling VIPP1 to bind and curve membranes. In vivo mutations in these hydrophobic surfaces cause extreme thylakoid swelling under high light, indicating an essential role of VIPP1 lipid binding in resisting stress-induced damage. Using cryo-correlative light and electron microscopy (cryo-CLEM), we observe oligomeric VIPP1 coats encapsulating membrane tubules within the Chlamydomonas chloroplast. Our work provides a structural foundation for understanding how VIPP1 directs thylakoid biogenesis and maintenance. | ||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12713.map.gz emd_12713.map.gz | 140.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12713-v30.xml emd-12713-v30.xml emd-12713.xml emd-12713.xml | 21.3 KB 21.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_12713_fsc.xml emd_12713_fsc.xml | 12.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_12713.png emd_12713.png | 50.8 KB | ||
| Masks |  emd_12713_msk_1.map emd_12713_msk_1.map | 178 MB |  Mask map Mask map | |
| Others |  emd_12713_additional_1.map.gz emd_12713_additional_1.map.gz emd_12713_half_map_1.map.gz emd_12713_half_map_1.map.gz emd_12713_half_map_2.map.gz emd_12713_half_map_2.map.gz | 165.7 MB 140.2 MB 140.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12713 http://ftp.pdbj.org/pub/emdb/structures/EMD-12713 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12713 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12713 | HTTPS FTP |
-Validation report
| Summary document |  emd_12713_validation.pdf.gz emd_12713_validation.pdf.gz | 459.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_12713_full_validation.pdf.gz emd_12713_full_validation.pdf.gz | 458.4 KB | Display | |
| Data in XML |  emd_12713_validation.xml.gz emd_12713_validation.xml.gz | 20.1 KB | Display | |
| Data in CIF |  emd_12713_validation.cif.gz emd_12713_validation.cif.gz | 26.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12713 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12713 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12713 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12713 | HTTPS FTP |
-Related structure data
| Related structure data |  7o40MC  7o3wC  7o3xC  7o3yC  7o3zC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_12713.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12713.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | C17 refined map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.35 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_12713_msk_1.map emd_12713_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: C17 map before refinement
| File | emd_12713_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | C17 map before refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: C17 half map 1
| File | emd_12713_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | C17 half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: C17 half map 2
| File | emd_12713_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | C17 half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : VIPP1 / IM30 complex
| Entire | Name: VIPP1 / IM30 complex |
|---|---|
| Components |
|
-Supramolecule #1: VIPP1 / IM30 complex
| Supramolecule | Name: VIPP1 / IM30 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
-Macromolecule #1: Protein sll0617
| Macromolecule | Name: Protein sll0617 / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Strain: PCC 6803 / Kazusa |
| Molecular weight | Theoretical: 28.822145 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: ALFDRLGRVV RANLNDLVSK AEDPEKVLEQ AVIDMQEDLV QLRQAVARTI AEEKRTEQRL NQDTQEAKKW EDRAKLALTN GEENLAREA LARKKSLTDT AAAYQTQLAQ QRTMSENLRR NLAALEAKIS EAKTKKNMLQ ARAKAAKANA ELQQTLGGLG T SSATSAFE ...String: ALFDRLGRVV RANLNDLVSK AEDPEKVLEQ AVIDMQEDLV QLRQAVARTI AEEKRTEQRL NQDTQEAKKW EDRAKLALTN GEENLAREA LARKKSLTDT AAAYQTQLAQ QRTMSENLRR NLAALEAKIS EAKTKKNMLQ ARAKAAKANA ELQQTLGGLG T SSATSAFE RMENKVLDME ATSQAAGELA GFGIENQFAQ LEASSGVEDE LAALKASMAG GALPGTSAAT PQLEAAPVDS SV PANNASQ DDAVIDQELD DLRRRLNNL |
-Macromolecule #2: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 2 / Number of copies: 1 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE-PROPANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 45.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Chain ID: A |
|---|---|
| Details | Initial model 4whe used for comparative modelling and Rosetta to predict the missing segments |
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT |
| Output model |  PDB-7o40: |
 Movie
Movie Controller
Controller

















 Z
Z Y
Y X
X