[English] 日本語
 Yorodumi
Yorodumi- EMDB-11626: Structure of inner kinetochore CCAN-Cenp-A complex (Cenp-HIKTW) -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-11626 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of inner kinetochore CCAN-Cenp-A complex (Cenp-HIKTW) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 8.5 Å | |||||||||
 Authors Authors | Yan K / Yang J / Zhang Z / Barford D | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2019 Journal: Nature / Year: 2019Title: Structure of the inner kinetochore CCAN complex assembled onto a centromeric nucleosome. Authors: Kaige Yan / Jing Yang / Ziguo Zhang / Stephen H McLaughlin / Leifu Chang / Domenico Fasci / Ann E Ehrenhofer-Murray / Albert J R Heck / David Barford /     Abstract: In eukaryotes, accurate chromosome segregation in mitosis and meiosis maintains genome stability and prevents aneuploidy. Kinetochores are large protein complexes that, by assembling onto specialized ...In eukaryotes, accurate chromosome segregation in mitosis and meiosis maintains genome stability and prevents aneuploidy. Kinetochores are large protein complexes that, by assembling onto specialized Cenp-A nucleosomes, function to connect centromeric chromatin to microtubules of the mitotic spindle. Whereas the centromeres of vertebrate chromosomes comprise millions of DNA base pairs and attach to multiple microtubules, the simple point centromeres of budding yeast are connected to individual microtubules. All 16 budding yeast chromosomes assemble complete kinetochores using a single Cenp-A nucleosome (Cenp-A), each of which is perfectly centred on its cognate centromere. The inner and outer kinetochore modules are responsible for interacting with centromeric chromatin and microtubules, respectively. Here we describe the cryo-electron microscopy structure of the Saccharomyces cerevisiae inner kinetochore module, the constitutive centromere associated network (CCAN) complex, assembled onto a Cenp-A nucleosome (CCAN-Cenp-A). The structure explains the interdependency of the constituent subcomplexes of CCAN and shows how the Y-shaped opening of CCAN accommodates Cenp-A to enable specific CCAN subunits to contact the nucleosomal DNA and histone subunits. Interactions with the unwrapped DNA duplex at the two termini of Cenp-A are mediated predominantly by a DNA-binding groove in the Cenp-L-Cenp-N subcomplex. Disruption of these interactions impairs assembly of CCAN onto Cenp-A. Our data indicate a mechanism of Cenp-A nucleosome recognition by CCAN and how CCAN acts as a platform for assembly of the outer kinetochore to link centromeres to the mitotic spindle for chromosome segregation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_11626.map.gz emd_11626.map.gz | 49.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-11626-v30.xml emd-11626-v30.xml emd-11626.xml emd-11626.xml | 9.1 KB 9.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_11626.png emd_11626.png | 36.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-11626 http://ftp.pdbj.org/pub/emdb/structures/EMD-11626 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11626 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11626 | HTTPS FTP |
-Validation report
| Summary document |  emd_11626_validation.pdf.gz emd_11626_validation.pdf.gz | 224.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_11626_full_validation.pdf.gz emd_11626_full_validation.pdf.gz | 223.2 KB | Display | |
| Data in XML |  emd_11626_validation.xml.gz emd_11626_validation.xml.gz | 6.8 KB | Display | |
| Data in CIF |  emd_11626_validation.cif.gz emd_11626_validation.cif.gz | 7.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11626 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11626 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11626 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11626 | HTTPS FTP |
-Related structure data
| Related structure data |  0095C  0096C  0097C  6gypC  6gysC  6gyuC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_11626.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_11626.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.09 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
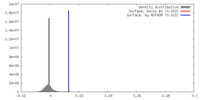
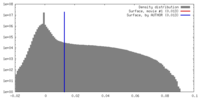
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : CCAN-Cenp-A Complex (Cenp-HIK-TW sub-module)
| Entire | Name: CCAN-Cenp-A Complex (Cenp-HIK-TW sub-module) |
|---|---|
| Components |
|
-Supramolecule #1: CCAN-Cenp-A Complex (Cenp-HIK-TW sub-module)
| Supramolecule | Name: CCAN-Cenp-A Complex (Cenp-HIK-TW sub-module) / type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  unidentified baculovirus unidentified baculovirus |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average electron dose: 32.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 8.5 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 33852 |
|---|---|
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)