+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8u6y | ||||||
|---|---|---|---|---|---|---|---|
| Title | Preholo-Proteasome from Beta 3 D205 deletion | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  HYDROLASE / HYDROLASE /  Proteasome / Proteasome /  core particle core particle | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of proteasome core complex assembly / proteasome core complex assembly / nuclear outer membrane-endoplasmic reticulum membrane network / Cross-presentation of soluble exogenous antigens (endosomes) / TNFR2 non-canonical NF-kB pathway / proteasomal ubiquitin-independent protein catabolic process / proteasome storage granule / endopeptidase activator activity /  proteasome assembly / proteasome assembly /  proteasome endopeptidase complex ...regulation of proteasome core complex assembly / proteasome core complex assembly / nuclear outer membrane-endoplasmic reticulum membrane network / Cross-presentation of soluble exogenous antigens (endosomes) / TNFR2 non-canonical NF-kB pathway / proteasomal ubiquitin-independent protein catabolic process / proteasome storage granule / endopeptidase activator activity / proteasome endopeptidase complex ...regulation of proteasome core complex assembly / proteasome core complex assembly / nuclear outer membrane-endoplasmic reticulum membrane network / Cross-presentation of soluble exogenous antigens (endosomes) / TNFR2 non-canonical NF-kB pathway / proteasomal ubiquitin-independent protein catabolic process / proteasome storage granule / endopeptidase activator activity /  proteasome assembly / proteasome assembly /  proteasome endopeptidase complex / Ub-specific processing proteases / proteasome core complex, beta-subunit complex / proteasome core complex, alpha-subunit complex / threonine-type endopeptidase activity / Neutrophil degranulation / proteasome endopeptidase complex / Ub-specific processing proteases / proteasome core complex, beta-subunit complex / proteasome core complex, alpha-subunit complex / threonine-type endopeptidase activity / Neutrophil degranulation /  proteasome complex / proteasomal protein catabolic process / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process / proteasome complex / proteasomal protein catabolic process / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process /  endopeptidase activity / endopeptidase activity /  mRNA binding / DNA damage response / endoplasmic reticulum membrane / mRNA binding / DNA damage response / endoplasmic reticulum membrane /  mitochondrion / mitochondrion /  nucleus / nucleus /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Saccharomyces cerevisiae S288C (yeast) Saccharomyces cerevisiae S288C (yeast) | ||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.8 Å cryo EM / Resolution: 2.8 Å | ||||||
 Authors Authors | Walsh Jr., R.M. / Rawson, S. / Velez, B. / Blickling, M. / Razi, A. / Hanna, J. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Mechanism of autocatalytic activation during proteasome assembly. Authors: Benjamin Velez / Richard M Walsh / Shaun Rawson / Aida Razi / Lea Adams / Erignacio Fermin Perez / Fenglong Jiao / Marie Blickling / Tamayanthi Rajakumar / Darlene Fung / Lan Huang / John Hanna /  Abstract: Many large molecular machines are too elaborate to assemble spontaneously and are built through ordered pathways orchestrated by dedicated chaperones. During assembly of the core particle (CP) of the ...Many large molecular machines are too elaborate to assemble spontaneously and are built through ordered pathways orchestrated by dedicated chaperones. During assembly of the core particle (CP) of the proteasome, where protein degradation occurs, its six active sites are simultaneously activated via cleavage of N-terminal propeptides. Such activation is autocatalytic and coupled to fusion of two half-CP intermediates, which protects cells by preventing activation until enclosure of the active sites within the CP interior. Here we uncover key mechanistic aspects of autocatalytic activation, which proceeds through alignment of the β5 and β2 catalytic triad residues, respectively, with these triads being misaligned before fusion. This mechanism contrasts with most other zymogens, in which catalytic centers are preformed. Our data also clarify the mechanism by which individual subunits can be added in a precise, temporally ordered manner. This work informs two decades-old mysteries in the proteasome field, with broader implications for protease biology and multisubunit complex assembly. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8u6y.cif.gz 8u6y.cif.gz | 2.6 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8u6y.ent.gz pdb8u6y.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  8u6y.json.gz 8u6y.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/u6/8u6y https://data.pdbj.org/pub/pdb/validation_reports/u6/8u6y ftp://data.pdbj.org/pub/pdb/validation_reports/u6/8u6y ftp://data.pdbj.org/pub/pdb/validation_reports/u6/8u6y | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 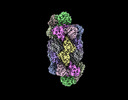 41963MC  8u7uC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Proteasome subunit alpha type- ... , 7 types, 14 molecules ARBSCTDUEVFWGX
| #1: Protein |  / Macropain subunit C7-alpha / Multicatalytic endopeptidase complex C7 / Proteasome component C7- ...Macropain subunit C7-alpha / Multicatalytic endopeptidase complex C7 / Proteasome component C7-alpha / Proteasome component Y8 / Proteinase YSCE subunit 7 / SCL1 suppressor protein / Macropain subunit C7-alpha / Multicatalytic endopeptidase complex C7 / Proteasome component C7- ...Macropain subunit C7-alpha / Multicatalytic endopeptidase complex C7 / Proteasome component C7-alpha / Proteasome component Y8 / Proteinase YSCE subunit 7 / SCL1 suppressor proteinMass: 28033.830 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P21243,  proteasome endopeptidase complex proteasome endopeptidase complex#2: Protein |  / Macropain subunit Y7 / Multicatalytic endopeptidase complex subunit Y7 / Proteasome component Y7 / ...Macropain subunit Y7 / Multicatalytic endopeptidase complex subunit Y7 / Proteasome component Y7 / Proteinase YSCE subunit 7 / Macropain subunit Y7 / Multicatalytic endopeptidase complex subunit Y7 / Proteasome component Y7 / ...Macropain subunit Y7 / Multicatalytic endopeptidase complex subunit Y7 / Proteasome component Y7 / Proteinase YSCE subunit 7Mass: 27191.828 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P23639,  proteasome endopeptidase complex proteasome endopeptidase complex#3: Protein |  / Macropain subunit Y13 / Multicatalytic endopeptidase complex subunit Y13 / Proteasome component Y13 ...Macropain subunit Y13 / Multicatalytic endopeptidase complex subunit Y13 / Proteasome component Y13 / Proteinase YSCE subunit 13 / Macropain subunit Y13 / Multicatalytic endopeptidase complex subunit Y13 / Proteasome component Y13 ...Macropain subunit Y13 / Multicatalytic endopeptidase complex subunit Y13 / Proteasome component Y13 / Proteinase YSCE subunit 13Mass: 27181.609 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: P23638 Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: P23638#4: Protein |  / Macropain subunit PRE6 / Multicatalytic endopeptidase complex subunit PRE6 / Proteasome component ...Macropain subunit PRE6 / Multicatalytic endopeptidase complex subunit PRE6 / Proteasome component PRE6 / Proteinase YSCE subunit PRE6 / Macropain subunit PRE6 / Multicatalytic endopeptidase complex subunit PRE6 / Proteasome component ...Macropain subunit PRE6 / Multicatalytic endopeptidase complex subunit PRE6 / Proteasome component PRE6 / Proteinase YSCE subunit PRE6Mass: 28478.111 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P40303,  proteasome endopeptidase complex proteasome endopeptidase complex#5: Protein |  / Macropain subunit PUP2 / Multicatalytic endopeptidase complex subunit PUP2 / Proteasome component ...Macropain subunit PUP2 / Multicatalytic endopeptidase complex subunit PUP2 / Proteasome component PUP2 / Proteinase YSCE subunit PUP2 / Macropain subunit PUP2 / Multicatalytic endopeptidase complex subunit PUP2 / Proteasome component ...Macropain subunit PUP2 / Multicatalytic endopeptidase complex subunit PUP2 / Proteasome component PUP2 / Proteinase YSCE subunit PUP2Mass: 28649.086 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P32379,  proteasome endopeptidase complex proteasome endopeptidase complex#6: Protein |  / Macropain subunit PRE5 / Multicatalytic endopeptidase complex subunit PRE5 / Proteasome component ...Macropain subunit PRE5 / Multicatalytic endopeptidase complex subunit PRE5 / Proteasome component PRE5 / Proteinase YSCE subunit PRE5 / Macropain subunit PRE5 / Multicatalytic endopeptidase complex subunit PRE5 / Proteasome component ...Macropain subunit PRE5 / Multicatalytic endopeptidase complex subunit PRE5 / Proteasome component PRE5 / Proteinase YSCE subunit PRE5Mass: 25634.000 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P40302,  proteasome endopeptidase complex proteasome endopeptidase complex#7: Protein |  / Macropain subunit C1 / Multicatalytic endopeptidase complex subunit C1 / Proteasome component C1 / ...Macropain subunit C1 / Multicatalytic endopeptidase complex subunit C1 / Proteasome component C1 / Proteinase YSCE subunit 1 / Macropain subunit C1 / Multicatalytic endopeptidase complex subunit C1 / Proteasome component C1 / ...Macropain subunit C1 / Multicatalytic endopeptidase complex subunit C1 / Proteasome component C1 / Proteinase YSCE subunit 1Mass: 31575.068 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P21242,  proteasome endopeptidase complex proteasome endopeptidase complex |
|---|
-Protein , 3 types, 6 molecules HYOfPg
| #8: Protein | Mass: 16777.766 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: P38293 Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: P38293#15: Protein | Mass: 30718.074 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: Q05778 Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: Q05778#16: Protein | Mass: 30762.895 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: P36040 Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: P36040 |
|---|
-Proteasome subunit beta type- ... , 7 types, 14 molecules IZJaKbLcMdNeQh
| #9: Protein |  PSMB2 / Macropain subunit PUP1 / Multicatalytic endopeptidase complex subunit PUP1 / Proteasome component ...Macropain subunit PUP1 / Multicatalytic endopeptidase complex subunit PUP1 / Proteasome component PUP1 / Proteinase YSCE subunit PUP1 PSMB2 / Macropain subunit PUP1 / Multicatalytic endopeptidase complex subunit PUP1 / Proteasome component ...Macropain subunit PUP1 / Multicatalytic endopeptidase complex subunit PUP1 / Proteasome component PUP1 / Proteinase YSCE subunit PUP1Mass: 28299.889 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P25043,  proteasome endopeptidase complex proteasome endopeptidase complex#10: Protein |  PSMB3 / Macropain subunit PUP3 / Multicatalytic endopeptidase complex subunit PUP3 / Proteasome component PUP3 PSMB3 / Macropain subunit PUP3 / Multicatalytic endopeptidase complex subunit PUP3 / Proteasome component PUP3Mass: 22512.754 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: P25451 Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c / References: UniProt: P25451#11: Protein |  PSMB4 / Macropain subunit C11 / Multicatalytic endopeptidase complex subunit C11 / Proteasome component C11 ...Macropain subunit C11 / Multicatalytic endopeptidase complex subunit C11 / Proteasome component C11 / Proteinase YSCE subunit 11 PSMB4 / Macropain subunit C11 / Multicatalytic endopeptidase complex subunit C11 / Proteasome component C11 ...Macropain subunit C11 / Multicatalytic endopeptidase complex subunit C11 / Proteasome component C11 / Proteinase YSCE subunit 11Mass: 22545.676 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P22141,  proteasome endopeptidase complex proteasome endopeptidase complex#12: Protein |  PSMB5 / Macropain subunit PRE2 / Multicatalytic endopeptidase complex subunit PRE2 / Proteasome component ...Macropain subunit PRE2 / Multicatalytic endopeptidase complex subunit PRE2 / Proteasome component PRE2 / Proteinase YSCE subunit PRE2 PSMB5 / Macropain subunit PRE2 / Multicatalytic endopeptidase complex subunit PRE2 / Proteasome component ...Macropain subunit PRE2 / Multicatalytic endopeptidase complex subunit PRE2 / Proteasome component PRE2 / Proteinase YSCE subunit PRE2Mass: 31670.539 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P30656,  proteasome endopeptidase complex proteasome endopeptidase complex#13: Protein |  / Multicatalytic endopeptidase complex subunit C5 / Proteasome component C5 / Multicatalytic endopeptidase complex subunit C5 / Proteasome component C5Mass: 26905.076 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P23724,  proteasome endopeptidase complex proteasome endopeptidase complex#14: Protein |  PSMB1 / Macropain subunit PRE3 / Multicatalytic endopeptidase complex subunit PRE3 / Proteasome component ...Macropain subunit PRE3 / Multicatalytic endopeptidase complex subunit PRE3 / Proteasome component PRE3 / Proteinase YSCE subunit PRE3 PSMB1 / Macropain subunit PRE3 / Multicatalytic endopeptidase complex subunit PRE3 / Proteasome component ...Macropain subunit PRE3 / Multicatalytic endopeptidase complex subunit PRE3 / Proteasome component PRE3 / Proteinase YSCE subunit PRE3Mass: 23573.604 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P38624,  proteasome endopeptidase complex proteasome endopeptidase complex#17: Protein |  / Macropain subunit PRE4 / Multicatalytic endopeptidase complex subunit PRE4 / Proteasome component ...Macropain subunit PRE4 / Multicatalytic endopeptidase complex subunit PRE4 / Proteasome component PRE4 / Proteinase YSCE subunit PRE4 / Macropain subunit PRE4 / Multicatalytic endopeptidase complex subunit PRE4 / Proteasome component ...Macropain subunit PRE4 / Multicatalytic endopeptidase complex subunit PRE4 / Proteasome component PRE4 / Proteinase YSCE subunit PRE4Mass: 29471.289 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288cReferences: UniProt: P30657,  proteasome endopeptidase complex proteasome endopeptidase complex |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Preholo-proteasome from Beta 3 D205 deletion mutant / Type: COMPLEX Details: D205 is terminal most residue in the Beta 3 subunit. The deletion of this residue causes an accumulation of assembly intermediates including the Preholo-Proteasome Entity ID: all / Source: NATURAL | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.75 MDa / Experimental value: NO | ||||||||||||||||||||
| Source (natural) | Organism:   Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c Saccharomyces cerevisiae S288C (yeast) / Strain: ATCC 204508 / S288c | ||||||||||||||||||||
| Buffer solution | pH: 7.5 Details: Fluorinated Fos-Choline was added immediately prior to deposition on a grid for plunge freezing. | ||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||
| Specimen | Conc.: 4.48 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES | ||||||||||||||||||||
| Specimen support | Grid material: COPPER / Grid mesh size: 400 divisions/in. / Grid type: Quantifoil R1.2/1.3 | ||||||||||||||||||||
Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 295.15 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal magnification: 81000 X / Calibrated magnification: 47169 X / Nominal defocus max: 2200 nm / Nominal defocus min: 800 nm / Cs Bright-field microscopy / Nominal magnification: 81000 X / Calibrated magnification: 47169 X / Nominal defocus max: 2200 nm / Nominal defocus min: 800 nm / Cs : 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: COMA FREE : 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 3.995 sec. / Electron dose: 57 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 7461 |
| EM imaging optics | Energyfilter name : GIF Bioquantum / Energyfilter slit width: 25 eV : GIF Bioquantum / Energyfilter slit width: 25 eV |
- Processing
Processing
| EM software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 1146597 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry : C2 (2 fold cyclic : C2 (2 fold cyclic ) ) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
3D reconstruction | Resolution: 2.8 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 19020 / Symmetry type: POINT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: AB INITIO MODEL / Space: REAL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 8T08 Accession code: 8T08 / Source name: PDB / Type: experimental model |
 Movie
Movie Controller
Controller




 PDBj
PDBj













