+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8tie | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
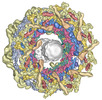
| Title | Double nuclear outer ring of Nup84-complexes from the yeast NPC | |||||||||||||||
 Components Components |
| |||||||||||||||
 Keywords Keywords |  TRANSPORT PROTEIN / TRANSPORT PROTEIN /  nuclear pore complex / nucleocytoplasmic transport / nuclear pore complex / nucleocytoplasmic transport /  nucleoporin / nucleoporin /  membrane protein / membrane protein /  translocase translocase | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationmRNA export from nucleus in response to heat stress / Seh1-associated complex / positive regulation of ER to Golgi vesicle-mediated transport / protein exit from endoplasmic reticulum / COPII-coated vesicle budding / COPII-mediated vesicle transport / nuclear pore central transport channel / nuclear pore localization / telomere tethering at nuclear periphery / regulation of TORC1 signaling ...mRNA export from nucleus in response to heat stress / Seh1-associated complex / positive regulation of ER to Golgi vesicle-mediated transport / protein exit from endoplasmic reticulum / COPII-coated vesicle budding / COPII-mediated vesicle transport / nuclear pore central transport channel / nuclear pore localization / telomere tethering at nuclear periphery / regulation of TORC1 signaling / nuclear pore outer ring / post-transcriptional tethering of RNA polymerase II gene DNA at nuclear periphery / COPII vesicle coat / positive regulation of protein exit from endoplasmic reticulum / structural constituent of nuclear pore / silent mating-type cassette heterochromatin formation / nucleocytoplasmic transport / vacuolar membrane / subtelomeric heterochromatin formation /  ribosomal large subunit export from nucleus / positive regulation of TOR signaling / mRNA transport / mRNA export from nucleus / ribosomal large subunit export from nucleus / positive regulation of TOR signaling / mRNA transport / mRNA export from nucleus /  nuclear pore / nuclear pore /  : / positive regulation of TORC1 signaling / protein export from nucleus / cell periphery / protein import into nucleus / double-strand break repair / : / positive regulation of TORC1 signaling / protein export from nucleus / cell periphery / protein import into nucleus / double-strand break repair /  nuclear envelope / nuclear envelope /  nuclear membrane / nuclear membrane /  chromosome, telomeric region / endoplasmic reticulum membrane / structural molecule activity / positive regulation of DNA-templated transcription / negative regulation of transcription by RNA polymerase II / chromosome, telomeric region / endoplasmic reticulum membrane / structural molecule activity / positive regulation of DNA-templated transcription / negative regulation of transcription by RNA polymerase II /  endoplasmic reticulum / positive regulation of transcription by RNA polymerase II / identical protein binding endoplasmic reticulum / positive regulation of transcription by RNA polymerase II / identical protein bindingSimilarity search - Function | |||||||||||||||
| Biological species |   Saccharomyces cerevisiae (brewer's yeast) Saccharomyces cerevisiae (brewer's yeast) | |||||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 8.1 Å cryo EM / Resolution: 8.1 Å | |||||||||||||||
 Authors Authors | Akey, C.W. / Echeverria, I. / Ouch, C. / Fernandez-Martinez, J. / Rout, M.P. | |||||||||||||||
| Funding support |  United States, 4items United States, 4items
| |||||||||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2023 Journal: Mol Cell / Year: 2023Title: Implications of a multiscale structure of the yeast nuclear pore complex. Authors: Christopher W Akey / Ignacia Echeverria / Christna Ouch / Ilona Nudelman / Yi Shi / Junjie Wang / Brian T Chait / Andrej Sali / Javier Fernandez-Martinez / Michael P Rout /   Abstract: Nuclear pore complexes (NPCs) direct the nucleocytoplasmic transport of macromolecules. Here, we provide a composite multiscale structure of the yeast NPC, based on improved 3D density maps from ...Nuclear pore complexes (NPCs) direct the nucleocytoplasmic transport of macromolecules. Here, we provide a composite multiscale structure of the yeast NPC, based on improved 3D density maps from cryogenic electron microscopy and AlphaFold2 models. Key features of the inner and outer rings were integrated into a comprehensive model. We resolved flexible connectors that tie together the core scaffold, along with equatorial transmembrane complexes and a lumenal ring that anchor this channel within the pore membrane. The organization of the nuclear double outer ring reveals an architecture that may be shared with ancestral NPCs. Additional connections between the core scaffold and the central transporter suggest that under certain conditions, a degree of local organization is present at the periphery of the transport machinery. These connectors may couple conformational changes in the scaffold to the central transporter to modulate transport. Collectively, this analysis provides insights into assembly, transport, and NPC evolution. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8tie.cif.gz 8tie.cif.gz | 1.6 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8tie.ent.gz pdb8tie.ent.gz | 1.3 MB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8tie.json.gz 8tie.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ti/8tie https://data.pdbj.org/pub/pdb/validation_reports/ti/8tie ftp://data.pdbj.org/pub/pdb/validation_reports/ti/8tie ftp://data.pdbj.org/pub/pdb/validation_reports/ti/8tie | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  41285MC  8t9lC  8tj5C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 7 types, 14 molecules albmcndoepfqgr
| #1: Protein | Mass: 120560.328 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Details: long arm of Nup84 Y-complex / Source: (natural)   Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / Plasmid details: nucleus / Strain: MATa ade2-1 ura3-1 his3-11 / References: UniProt: P35729 Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / Plasmid details: nucleus / Strain: MATa ade2-1 ura3-1 his3-11 / References: UniProt: P35729#2: Protein | Mass: 84972.438 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Details: short arm of Nup84 Y-complex / Source: (natural)   Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: P46673 Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: P46673#3: Protein | Mass: 145810.578 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Details: stem junction of Nup84 Y-complex / Source: (natural)   Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: A0A8H4C085 Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: A0A8H4C085#4: Protein |  Protein targeting Protein targetingMass: 33082.965 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Details: cofactor of Nup145C on stem of Nup84 Y-complex / Source: (natural)   Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: Q04491 Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: Q04491#5: Protein | Mass: 34652.078 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Details: cofactor Nup85 / Source: (natural)   Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303#6: Protein | Mass: 83718.867 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Details: tail of Nup84 Y-complex / Source: (natural)   Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: P52891 Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: P52891#7: Protein | Mass: 133452.672 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Details: tail Nup84 Y-complex / Source: (natural)   Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: A0A6V8RYD2 Saccharomyces cerevisiae (brewer's yeast) / Cell line: W303 / References: UniProt: A0A6V8RYD2 |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight |
| |||||||||||||||||||||
| Source (natural) |
| |||||||||||||||||||||
| Buffer solution | pH: 7.5 Details: 20mM HEPES,50mM Potassium acetate,20mM NaCl,2mM MgCl2,1mM DTT | |||||||||||||||||||||
| Specimen | Conc.: 0.1 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES / Details: One step affinity purified : YES / Details: One step affinity purified | |||||||||||||||||||||
Vitrification | Instrument: FEI VITROBOT MARK III / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 283 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS Details: Preliminary grid screening done manually with individual images of low magnification montages of candidate meshes. |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Calibrated magnification: 37651 X / Nominal defocus max: 3500 nm / Nominal defocus min: 1500 nm / Cs Bright-field microscopy / Calibrated magnification: 37651 X / Nominal defocus max: 3500 nm / Nominal defocus min: 1500 nm / Cs : 2.7 mm / C2 aperture diameter: 100 µm / Alignment procedure: COMA FREE : 2.7 mm / C2 aperture diameter: 100 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 40 e/Å2 / Detector mode: SUPER-RESOLUTION / Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Num. of grids imaged: 1 / Num. of real images: 4015 / Details: 3218 images retained after triage |
| EM imaging optics | Energyfilter name : GIF Bioquantum / Energyfilter slit width: 20 eV : GIF Bioquantum / Energyfilter slit width: 20 eV |
| Image scans | Width: 3838 / Height: 3710 / Movie frames/image: 40 / Used frames/image: 2-40 |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
CTF correction | Details: CTF correction applied in RELION during the alignment and reconstruction Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 26049 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
3D reconstruction | Resolution: 8.1 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 29655 / Algorithm: FOURIER SPACE Details: multibody in RELION followed by refinements in cryosparc Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL / Target criteria: cross correlation Details: Molecular dynamics flexible fitting after docking Alphafold2 CSMs for Nups into the 3D map of double Y complex ring with Chimera | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Details: none / Source name: Other / Type: experimental model |
 Movie
Movie Controller
Controller











 PDBj
PDBj
