[English] 日本語
 Yorodumi
Yorodumi- PDB-8ox7: Cryo-EM structure of ATP8B1-CDC50A in E2P autoinhibited "closed" ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8ox7 | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of ATP8B1-CDC50A in E2P autoinhibited "closed" conformation | |||||||||||||||||||||||||||
 Components Components |
| |||||||||||||||||||||||||||
 Keywords Keywords |  MEMBRANE PROTEIN / lipid transporter autoinhibition P-type ATPase P4-ATPase CDC50A MEMBRANE PROTEIN / lipid transporter autoinhibition P-type ATPase P4-ATPase CDC50A | |||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationvestibulocochlear nerve formation / regulation of plasma membrane organization /  regulation of microvillus assembly / positive regulation of phospholipid translocation / phosphatidylcholine flippase activity / aminophospholipid flippase activity / aminophospholipid transport / phosphatidylserine flippase activity / protein localization to endosome / inner ear receptor cell development ...vestibulocochlear nerve formation / regulation of plasma membrane organization / regulation of microvillus assembly / positive regulation of phospholipid translocation / phosphatidylcholine flippase activity / aminophospholipid flippase activity / aminophospholipid transport / phosphatidylserine flippase activity / protein localization to endosome / inner ear receptor cell development ...vestibulocochlear nerve formation / regulation of plasma membrane organization /  regulation of microvillus assembly / positive regulation of phospholipid translocation / phosphatidylcholine flippase activity / aminophospholipid flippase activity / aminophospholipid transport / phosphatidylserine flippase activity / protein localization to endosome / inner ear receptor cell development / regulation of microvillus assembly / positive regulation of phospholipid translocation / phosphatidylcholine flippase activity / aminophospholipid flippase activity / aminophospholipid transport / phosphatidylserine flippase activity / protein localization to endosome / inner ear receptor cell development /  phospholipid-translocating ATPase complex / ATPase-coupled intramembrane lipid transporter activity / phosphatidylserine floppase activity / positive regulation of protein exit from endoplasmic reticulum / xenobiotic transmembrane transport / phospholipid-translocating ATPase complex / ATPase-coupled intramembrane lipid transporter activity / phosphatidylserine floppase activity / positive regulation of protein exit from endoplasmic reticulum / xenobiotic transmembrane transport /  cardiolipin binding / phosphatidylcholine floppase activity / stereocilium / apical protein localization / bile acid metabolic process / bile acid and bile salt transport / P-type phospholipid transporter / phospholipid translocation / transport vesicle membrane / azurophil granule membrane / Golgi organization / Ion transport by P-type ATPases / specific granule membrane / monoatomic ion transmembrane transport / regulation of chloride transport / sensory perception of sound / cardiolipin binding / phosphatidylcholine floppase activity / stereocilium / apical protein localization / bile acid metabolic process / bile acid and bile salt transport / P-type phospholipid transporter / phospholipid translocation / transport vesicle membrane / azurophil granule membrane / Golgi organization / Ion transport by P-type ATPases / specific granule membrane / monoatomic ion transmembrane transport / regulation of chloride transport / sensory perception of sound /  trans-Golgi network / positive regulation of neuron projection development / late endosome membrane / early endosome membrane / trans-Golgi network / positive regulation of neuron projection development / late endosome membrane / early endosome membrane /  nuclear body / apical plasma membrane / negative regulation of DNA-templated transcription / Neutrophil degranulation / structural molecule activity / nuclear body / apical plasma membrane / negative regulation of DNA-templated transcription / Neutrophil degranulation / structural molecule activity /  Golgi apparatus / magnesium ion binding / Golgi apparatus / magnesium ion binding /  endoplasmic reticulum / endoplasmic reticulum /  ATP hydrolysis activity / ATP hydrolysis activity /  nucleoplasm / nucleoplasm /  ATP binding / ATP binding /  membrane / membrane /  plasma membrane / plasma membrane /  cytosol cytosolSimilarity search - Function | |||||||||||||||||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.56 Å cryo EM / Resolution: 2.56 Å | |||||||||||||||||||||||||||
 Authors Authors | Dieudonne, T. / Kummerer, F. / Juknaviciute Laursen, M. / Stock, C. / Kock Flygaard, R. / Khalid, S. / Lenoir, G. / Lyons, J.A. / Lindorff-Larsen, K. / Nissen, P. | |||||||||||||||||||||||||||
| Funding support | European Union,  Denmark, Denmark,  France, France,  United Kingdom, 8items United Kingdom, 8items
| |||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Activation and substrate specificity of the human P4-ATPase ATP8B1. Authors: Thibaud Dieudonné / Felix Kümmerer / Michelle Juknaviciute Laursen / Charlott Stock / Rasmus Kock Flygaard / Syma Khalid / Guillaume Lenoir / Joseph A Lyons / Kresten Lindorff-Larsen / Poul Nissen /    Abstract: Asymmetric distribution of phospholipids in eukaryotic membranes is essential for cell integrity, signaling pathways, and vesicular trafficking. P4-ATPases, also known as flippases, participate in ...Asymmetric distribution of phospholipids in eukaryotic membranes is essential for cell integrity, signaling pathways, and vesicular trafficking. P4-ATPases, also known as flippases, participate in creating and maintaining this asymmetry through active transport of phospholipids from the exoplasmic to the cytosolic leaflet. Here, we present a total of nine cryo-electron microscopy structures of the human flippase ATP8B1-CDC50A complex at 2.4 to 3.1 Å overall resolution, along with functional and computational studies, addressing the autophosphorylation steps from ATP, substrate recognition and occlusion, as well as a phosphoinositide binding site. We find that the P4-ATPase transport site is occupied by water upon phosphorylation from ATP. Additionally, we identify two different autoinhibited states, a closed and an outward-open conformation. Furthermore, we identify and characterize the PI(3,4,5)P binding site of ATP8B1 in an electropositive pocket between transmembrane segments 5, 7, 8, and 10. Our study also highlights the structural basis of a broad lipid specificity of ATP8B1 and adds phosphatidylinositol as a transport substrate for ATP8B1. We report a critical role of the sn-2 ester bond of glycerophospholipids in substrate recognition by ATP8B1 through conserved S403. These findings provide fundamental insights into ATP8B1 catalytic cycle and regulation, and substrate recognition in P4-ATPases. | |||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8ox7.cif.gz 8ox7.cif.gz | 277.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8ox7.ent.gz pdb8ox7.ent.gz | 216.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8ox7.json.gz 8ox7.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ox/8ox7 https://data.pdbj.org/pub/pdb/validation_reports/ox/8ox7 ftp://data.pdbj.org/pub/pdb/validation_reports/ox/8ox7 ftp://data.pdbj.org/pub/pdb/validation_reports/ox/8ox7 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  17259MC  8ox4C  8ox5C  8ox6C  8ox8C  8ox9C  8oxaC  8oxbC  8oxcC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 2 types, 2 molecules BA
| #1: Protein | Mass: 41085.984 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: TMEM30A, C6orf67, CDC50A / Production host: Homo sapiens (human) / Gene: TMEM30A, C6orf67, CDC50A / Production host:   Saccharomyces cerevisiae (brewer's yeast) / References: UniProt: Q9NV96 Saccharomyces cerevisiae (brewer's yeast) / References: UniProt: Q9NV96 |
|---|---|
| #2: Protein |  / ATPase class I type 8B member 1 / Familial intrahepatic cholestasis type 1 / P4-ATPase flippase ...ATPase class I type 8B member 1 / Familial intrahepatic cholestasis type 1 / P4-ATPase flippase complex alpha subunit ATP8B1 / ATPase class I type 8B member 1 / Familial intrahepatic cholestasis type 1 / P4-ATPase flippase ...ATPase class I type 8B member 1 / Familial intrahepatic cholestasis type 1 / P4-ATPase flippase complex alpha subunit ATP8B1Mass: 143976.781 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: ATP8B1, ATPIC, FIC1, PFIC / Production host: Homo sapiens (human) / Gene: ATP8B1, ATPIC, FIC1, PFIC / Production host:   Saccharomyces cerevisiae (brewer's yeast) Saccharomyces cerevisiae (brewer's yeast)References: UniProt: O43520, P-type phospholipid transporter |
-Sugars , 2 types, 3 molecules 
| #3: Polysaccharide | beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta- ...beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose / Mass: 586.542 Da / Num. of mol.: 1 / Mass: 586.542 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source |
|---|---|
| #4: Sugar |  N-Acetylglucosamine N-Acetylglucosamine |
-Non-polymers , 3 types, 3 molecules 



| #5: Chemical | ChemComp-MG / |
|---|---|
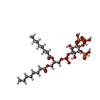
| #6: Chemical | ChemComp-IP9 / ( |
| #7: Water | ChemComp-HOH /  Water Water |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: ATP8B1-CDC50A complex / Type: COMPLEX / Entity ID: #1-#2 / Source: RECOMBINANT |
|---|---|
| Molecular weight | Value: 0.185 MDa / Experimental value: NO |
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:   Saccharomyces cerevisiae (brewer's yeast) Saccharomyces cerevisiae (brewer's yeast) |
| Buffer solution | pH: 7 |
| Specimen | Conc.: 3 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YESDetails: 50 mM MOPS-Tris pH 7, 100 mM KCl, 1 mM DTT, 5 mM MgCl2 supplemented with 0.03 mg.mL-1 LMNG, 0.0015 mg.mL-1 PI(3,4,5)P3 and 2 mM ATP |
Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 1900 nm / Nominal defocus min: 700 nm / Cs Bright-field microscopy / Nominal defocus max: 1900 nm / Nominal defocus min: 700 nm / Cs : 2.7 mm : 2.7 mm |
| Image recording | Electron dose: 60 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
| EM imaging optics | Energyfilter name : GIF Bioquantum / Energyfilter slit width: 20 eV : GIF Bioquantum / Energyfilter slit width: 20 eV |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||
3D reconstruction | Resolution: 2.56 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 573549 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL | ||||||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 7PY4 Accession code: 7PY4 / Source name: PDB / Type: experimental model | ||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj









