[English] 日本語
 Yorodumi
Yorodumi- PDB-7yp1: Cryo-EM structure of EBV gHgL-gp42 in complex with mAb 10E4 (loca... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7yp1 | ||||||
|---|---|---|---|---|---|---|---|
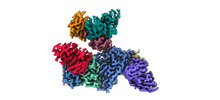
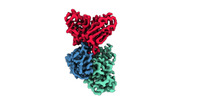
| Title | Cryo-EM structure of EBV gHgL-gp42 in complex with mAb 10E4 (localized refinement) | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  VIRAL PROTEIN / EBV / VIRAL PROTEIN / EBV /  Cryo-EM / Cryo-EM /  Glycoprotein / gHgL-gp42 complex / Glycoprotein / gHgL-gp42 complex /  Antibody Antibody | ||||||
| Biological species |   Human gammaherpesvirus 4 (Epstein-Barr virus) Human gammaherpesvirus 4 (Epstein-Barr virus)  Oryctolagus cuniculus (rabbit) Oryctolagus cuniculus (rabbit) | ||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.54 Å cryo EM / Resolution: 3.54 Å | ||||||
 Authors Authors | Liu, L. / Sun, H. / Jiang, Y. / Hong, J. / Zheng, Q. / Li, S. / Chen, Y. / Xia, N. | ||||||
| Funding support | 1items
| ||||||
 Citation Citation |  Journal: Cell Rep Med / Year: 2023 Journal: Cell Rep Med / Year: 2023Title: Non-overlapping epitopes on the gHgL-gp42 complex for the rational design of a triple-antibody cocktail against EBV infection. Authors: Junping Hong / Ling Zhong / Liqin Liu / Qian Wu / Wanlin Zhang / Kaiyun Chen / Dongmei Wei / Hui Sun / Xiang Zhou / Xinyu Zhang / Yin-Feng Kang / Yang Huang / Junyu Chen / Guosong Wang / Yan ...Authors: Junping Hong / Ling Zhong / Liqin Liu / Qian Wu / Wanlin Zhang / Kaiyun Chen / Dongmei Wei / Hui Sun / Xiang Zhou / Xinyu Zhang / Yin-Feng Kang / Yang Huang / Junyu Chen / Guosong Wang / Yan Zhou / Yanhong Chen / Qi-Sheng Feng / Hai Yu / Shaowei Li / Mu-Sheng Zeng / Yi-Xin Zeng / Miao Xu / Qingbing Zheng / Yixin Chen / Xiao Zhang / Ningshao Xia /   Abstract: Epstein-Barr virus (EBV) is closely associated with cancer, multiple sclerosis, and post-acute coronavirus disease 2019 (COVID-19) sequelae. There are currently no approved therapeutics or vaccines ...Epstein-Barr virus (EBV) is closely associated with cancer, multiple sclerosis, and post-acute coronavirus disease 2019 (COVID-19) sequelae. There are currently no approved therapeutics or vaccines against EBV. It is noteworthy that combining multiple EBV glycoproteins can elicit potent neutralizing antibodies (nAbs) against viral infection, suggesting possible synergistic effects. Here, we characterize three nAbs (anti-gp42 5E3, anti-gHgL 6H2, and anti-gHgL 10E4) targeting different glycoproteins of the gHgL-gp42 complex. Two antibody cocktails synergistically neutralize infection in B cells (5E3+6H2+10E4) and epithelial cells (6H2+10E4) in vitro. Moreover, 5E3 alone and the 5E3+6H2+10E4 cocktail confer potent in vivo protection against lethal EBV challenge in humanized mice. The cryo-EM structure of a heptatomic gHgL-gp42 immune complex reveals non-overlapping epitopes of 5E3, 6H2, and 10E4 on the gHgL-gp42 complex. Structural and functional analyses highlight different neutralization mechanisms for each of the three nAbs. In summary, our results provide insight for the rational design of therapeutics or vaccines against EBV infection. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7yp1.cif.gz 7yp1.cif.gz | 95.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7yp1.ent.gz pdb7yp1.ent.gz | 72.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7yp1.json.gz 7yp1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/yp/7yp1 https://data.pdbj.org/pub/pdb/validation_reports/yp/7yp1 ftp://data.pdbj.org/pub/pdb/validation_reports/yp/7yp1 ftp://data.pdbj.org/pub/pdb/validation_reports/yp/7yp1 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 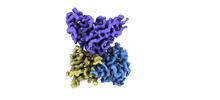 33992MC  7yoyC  7yp2C M: map data used to model this data C: citing same article ( |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 22774.117 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Human gammaherpesvirus 4 (Epstein-Barr virus) Human gammaherpesvirus 4 (Epstein-Barr virus)Cell line (production host): HEK293 / Production host:   Homo sapiens (human) Homo sapiens (human) |
|---|---|
| #2: Protein | Mass: 8623.719 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Human gammaherpesvirus 4 (Epstein-Barr virus) Human gammaherpesvirus 4 (Epstein-Barr virus)Cell line (production host): HEK293 / Production host:   Homo sapiens (human) Homo sapiens (human) |
| #3: Protein | Mass: 12273.763 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Oryctolagus cuniculus (rabbit) / Cell line (production host): HEK293 / Production host: Oryctolagus cuniculus (rabbit) / Cell line (production host): HEK293 / Production host:   Homo sapiens (human) Homo sapiens (human) |
| #4: Antibody | Mass: 11917.251 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Oryctolagus cuniculus (rabbit) / Cell line (production host): HEK293 / Production host: Oryctolagus cuniculus (rabbit) / Cell line (production host): HEK293 / Production host:   Homo sapiens (human) Homo sapiens (human) |
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Source (natural) |
| ||||||||||||||||||||||||
| Source (recombinant) |
| ||||||||||||||||||||||||
| Buffer solution | pH: 7.4 | ||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES | ||||||||||||||||||||||||
Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Tecnai F30 / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TECNAI F30 |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 3000 nm / Nominal defocus min: 1000 nm Bright-field microscopy / Nominal defocus max: 3000 nm / Nominal defocus min: 1000 nm |
| Image recording | Electron dose: 60 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.14_3259: / Classification: refinement |
|---|---|
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
3D reconstruction | Resolution: 3.54 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 402382 / Symmetry type: POINT |
 Movie
Movie Controller
Controller



 PDBj
PDBj

