[English] 日本語
 Yorodumi
Yorodumi- PDB-7nwn: A carbohydrate binding module family 9 (CBM9) from Caldicellulsir... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7nwn | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
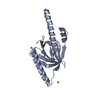
| Title | A carbohydrate binding module family 9 (CBM9) from Caldicellulsiruptor kristjanssonii | |||||||||||||||
 Components Components | Beta-xylanase Xylanase Xylanase | |||||||||||||||
 Keywords Keywords | SUGAR BINDING PROTEIN / CARBOHYDRATE BINDING MODULE | |||||||||||||||
| Function / homology |  Function and homology information Function and homology information endo-1,4-beta-xylanase activity / endo-1,4-beta-xylanase activity /  endo-1,4-beta-xylanase / polysaccharide catabolic process / endo-1,4-beta-xylanase / polysaccharide catabolic process /  carbohydrate binding carbohydrate bindingSimilarity search - Function | |||||||||||||||
| Biological species | Caldicellulosiruptor kristjanssonii | |||||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.97 Å MOLECULAR REPLACEMENT / Resolution: 1.97 Å | |||||||||||||||
 Authors Authors | Krska, D. / Mazurkewich, S. / Navarro Poulsen, J. / Larsbrink, J. / Lo Leggio, L. | |||||||||||||||
| Funding support |  Sweden, Sweden,  Denmark, 4items Denmark, 4items
| |||||||||||||||
 Citation Citation |  Journal: Biochemistry / Year: 2021 Journal: Biochemistry / Year: 2021Title: Structural and Functional Analysis of a Multimodular Hyperthermostable Xylanase-Glucuronoyl Esterase from Caldicellulosiruptor kristjansonii . Authors: Krska, D. / Mazurkewich, S. / Brown, H.A. / Theibich, Y. / Poulsen, J.N. / Morris, A.L. / Koropatkin, N.M. / Lo Leggio, L. / Larsbrink, J. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7nwn.cif.gz 7nwn.cif.gz | 64.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7nwn.ent.gz pdb7nwn.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  7nwn.json.gz 7nwn.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/nw/7nwn https://data.pdbj.org/pub/pdb/validation_reports/nw/7nwn ftp://data.pdbj.org/pub/pdb/validation_reports/nw/7nwn ftp://data.pdbj.org/pub/pdb/validation_reports/nw/7nwn | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7nn3C  7nwoC  7nwpC  7nwqC  1i8uS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
-Protein , 1 types, 1 molecules AAA
| #1: Protein |  Xylanase XylanaseMass: 24645.232 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Caldicellulosiruptor kristjanssonii (strain ATCC 700853 / DSM 12137 / I77R1B) (bacteria) Caldicellulosiruptor kristjanssonii (strain ATCC 700853 / DSM 12137 / I77R1B) (bacteria)Strain: ATCC 700853 / DSM 12137 / I77R1B / Gene: Calkr_2245 / Production host:   Escherichia coli BL21 (bacteria) / References: UniProt: E4S6E9, Escherichia coli BL21 (bacteria) / References: UniProt: E4S6E9,  endo-1,4-beta-xylanase endo-1,4-beta-xylanase |
|---|
-Non-polymers , 5 types, 159 molecules 








| #2: Chemical | ChemComp-PEG /  Diethylene glycol Diethylene glycol#3: Chemical | ChemComp-EDO /  Ethylene glycol Ethylene glycol#4: Chemical | #5: Chemical | ChemComp-P6G / |  Polyethylene glycol Polyethylene glycol#6: Water | ChemComp-HOH / |  Water Water |
|---|
-Details
| Has ligand of interest | N |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4.35 Å3/Da / Density % sol: 71.74 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop Details: 0.1 M phosphate-citrate and 40% polyethylene glycol 300 PH range: 4.2 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  MAX IV MAX IV  / Beamline: BioMAX / Wavelength: 1.00003 Å / Beamline: BioMAX / Wavelength: 1.00003 Å |
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Apr 19, 2019 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.00003 Å / Relative weight: 1 : 1.00003 Å / Relative weight: 1 |
| Reflection | Resolution: 1.97→46.191 Å / Num. obs: 31213 / % possible obs: 100 % / Redundancy: 79.9 % / CC1/2: 1 / Net I/σ(I): 26.81 |
| Reflection shell | Resolution: 1.97→2.11 Å / Num. unique obs: 5694 / CC1/2: 0.76 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1I8U Resolution: 1.97→46.191 Å / Cor.coef. Fo:Fc: 0.969 / Cor.coef. Fo:Fc free: 0.946 / WRfactor Rfree: 0.211 / WRfactor Rwork: 0.162 / SU B: 2.956 / SU ML: 0.08 / Average fsc free: 0.9379 / Average fsc overall: 0.949 / Average fsc work: 0.9496 / Cross valid method: FREE R-VALUE / ESU R: 0.104 / ESU R Free: 0.115 / Details: Hydrogens have not been used
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK BULK SOLVENT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 46.697 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.97→46.191 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller









 PDBj
PDBj




