[English] 日本語
 Yorodumi
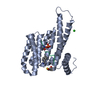
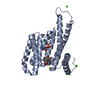
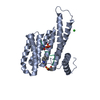
Yorodumi- PDB-7bjb: 14-3-3 sigma with RelA/p65 binding site pS45 and covalently bound... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7bjb | ||||||
|---|---|---|---|---|---|---|---|
| Title | 14-3-3 sigma with RelA/p65 binding site pS45 and covalently bound TCF521-044 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | PEPTIDE BINDING PROTEIN /  benzaldehyde / covalent fragment / p65 / 1433 / benzaldehyde / covalent fragment / p65 / 1433 /  RelA RelA | ||||||
| Function / homology |  Function and homology information Function and homology informationacetaldehyde metabolic process / prolactin signaling pathway / DEx/H-box helicases activate type I IFN and inflammatory cytokines production / NF-kappaB p50/p65 complex / IkBA variant leads to EDA-ID / positive regulation of Schwann cell differentiation / cellular response to peptidoglycan / Regulated proteolysis of p75NTR /  ankyrin repeat binding / RIP-mediated NFkB activation via ZBP1 ...acetaldehyde metabolic process / prolactin signaling pathway / DEx/H-box helicases activate type I IFN and inflammatory cytokines production / NF-kappaB p50/p65 complex / IkBA variant leads to EDA-ID / positive regulation of Schwann cell differentiation / cellular response to peptidoglycan / Regulated proteolysis of p75NTR / ankyrin repeat binding / RIP-mediated NFkB activation via ZBP1 ...acetaldehyde metabolic process / prolactin signaling pathway / DEx/H-box helicases activate type I IFN and inflammatory cytokines production / NF-kappaB p50/p65 complex / IkBA variant leads to EDA-ID / positive regulation of Schwann cell differentiation / cellular response to peptidoglycan / Regulated proteolysis of p75NTR /  ankyrin repeat binding / RIP-mediated NFkB activation via ZBP1 / SUMOylation of immune response proteins / CLEC7A/inflammasome pathway / negative regulation of protein sumoylation / defense response to tumor cell / postsynapse to nucleus signaling pathway / nucleotide-binding oligomerization domain containing 2 signaling pathway / cellular response to interleukin-6 / Interleukin-1 processing / ankyrin repeat binding / RIP-mediated NFkB activation via ZBP1 / SUMOylation of immune response proteins / CLEC7A/inflammasome pathway / negative regulation of protein sumoylation / defense response to tumor cell / postsynapse to nucleus signaling pathway / nucleotide-binding oligomerization domain containing 2 signaling pathway / cellular response to interleukin-6 / Interleukin-1 processing /  actinin binding / negative regulation of non-canonical NF-kappaB signal transduction / cellular response to angiotensin / actinin binding / negative regulation of non-canonical NF-kappaB signal transduction / cellular response to angiotensin /  NF-kappaB complex / response to UV-B / interleukin-1-mediated signaling pathway / positive regulation of leukocyte adhesion to vascular endothelial cell / Regulation of NFE2L2 gene expression / vascular endothelial growth factor signaling pathway / toll-like receptor 4 signaling pathway / cellular response to hepatocyte growth factor stimulus / positive regulation of amyloid-beta formation / positive regulation of miRNA metabolic process / regulation of epidermal cell division / response to cobalamin / NF-kappaB complex / response to UV-B / interleukin-1-mediated signaling pathway / positive regulation of leukocyte adhesion to vascular endothelial cell / Regulation of NFE2L2 gene expression / vascular endothelial growth factor signaling pathway / toll-like receptor 4 signaling pathway / cellular response to hepatocyte growth factor stimulus / positive regulation of amyloid-beta formation / positive regulation of miRNA metabolic process / regulation of epidermal cell division / response to cobalamin /  protein kinase C inhibitor activity / positive regulation of T cell receptor signaling pathway / positive regulation of epidermal cell differentiation / keratinocyte development / protein kinase C inhibitor activity / positive regulation of T cell receptor signaling pathway / positive regulation of epidermal cell differentiation / keratinocyte development /  keratinization / keratinization /  phosphate ion binding / non-canonical NF-kappaB signal transduction / cellular response to lipoteichoic acid / TRAF6 mediated NF-kB activation / response to muramyl dipeptide / The NLRP3 inflammasome / Regulation of localization of FOXO transcription factors / general transcription initiation factor binding / keratinocyte proliferation / Transcriptional Regulation by VENTX / phosphate ion binding / non-canonical NF-kappaB signal transduction / cellular response to lipoteichoic acid / TRAF6 mediated NF-kB activation / response to muramyl dipeptide / The NLRP3 inflammasome / Regulation of localization of FOXO transcription factors / general transcription initiation factor binding / keratinocyte proliferation / Transcriptional Regulation by VENTX /  NF-kappaB binding / hair follicle development / phosphoserine residue binding / neuropeptide signaling pathway / positive regulation of vascular endothelial growth factor production / Activation of BAD and translocation to mitochondria / negative regulation of keratinocyte proliferation / RNA polymerase II core promoter sequence-specific DNA binding / canonical NF-kappaB signal transduction / establishment of skin barrier / cellular response to interleukin-1 / response to amino acid / cellular defense response / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex / NF-kappaB binding / hair follicle development / phosphoserine residue binding / neuropeptide signaling pathway / positive regulation of vascular endothelial growth factor production / Activation of BAD and translocation to mitochondria / negative regulation of keratinocyte proliferation / RNA polymerase II core promoter sequence-specific DNA binding / canonical NF-kappaB signal transduction / establishment of skin barrier / cellular response to interleukin-1 / response to amino acid / cellular defense response / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex /  Purinergic signaling in leishmaniasis infection / negative regulation of stem cell proliferation / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / RHO GTPases activate PKNs / response to cAMP / tumor necrosis factor-mediated signaling pathway / protein kinase A signaling / response to muscle stretch / protein export from nucleus / negative regulation of innate immune response / positive regulation of interleukin-12 production / negative regulation of insulin receptor signaling pathway / protein sequestering activity / NF-kB is activated and signals survival / CD209 (DC-SIGN) signaling / response to interleukin-1 / negative regulation of angiogenesis / TP53 Regulates Transcription of Genes Involved in G2 Cell Cycle Arrest / release of cytochrome c from mitochondria / : / positive regulation of protein export from nucleus / negative regulation of miRNA transcription / liver development / positive regulation of interleukin-1 beta production / response to progesterone / response to cytokine / stem cell proliferation / Translocation of SLC2A4 (GLUT4) to the plasma membrane / response to ischemia / positive regulation of interleukin-8 production / Dectin-1 mediated noncanonical NF-kB signaling / negative regulation of extrinsic apoptotic signaling pathway / TP53 Regulates Metabolic Genes / Activation of NF-kappaB in B cells / animal organ morphogenesis / RNA polymerase II transcription regulatory region sequence-specific DNA binding / Purinergic signaling in leishmaniasis infection / negative regulation of stem cell proliferation / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / RHO GTPases activate PKNs / response to cAMP / tumor necrosis factor-mediated signaling pathway / protein kinase A signaling / response to muscle stretch / protein export from nucleus / negative regulation of innate immune response / positive regulation of interleukin-12 production / negative regulation of insulin receptor signaling pathway / protein sequestering activity / NF-kB is activated and signals survival / CD209 (DC-SIGN) signaling / response to interleukin-1 / negative regulation of angiogenesis / TP53 Regulates Transcription of Genes Involved in G2 Cell Cycle Arrest / release of cytochrome c from mitochondria / : / positive regulation of protein export from nucleus / negative regulation of miRNA transcription / liver development / positive regulation of interleukin-1 beta production / response to progesterone / response to cytokine / stem cell proliferation / Translocation of SLC2A4 (GLUT4) to the plasma membrane / response to ischemia / positive regulation of interleukin-8 production / Dectin-1 mediated noncanonical NF-kB signaling / negative regulation of extrinsic apoptotic signaling pathway / TP53 Regulates Metabolic Genes / Activation of NF-kappaB in B cells / animal organ morphogenesis / RNA polymerase II transcription regulatory region sequence-specific DNA binding /  peptide binding peptide bindingSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 1.8 Å molecular replacement / Resolution: 1.8 Å | ||||||
 Authors Authors | Wolter, M. / Ottmann, C. | ||||||
| Funding support | European Union, 1items
| ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 2021 Journal: J.Med.Chem. / Year: 2021Title: An Exploration of Chemical Properties Required for Cooperative Stabilization of the 14-3-3 Interaction with NF-kappa B-Utilizing a Reversible Covalent Tethering Approach. Authors: Wolter, M. / Valenti, D. / Cossar, P.J. / Hristeva, S. / Levy, L.M. / Genski, T. / Hoffmann, T. / Brunsveld, L. / Tzalis, D. / Ottmann, C. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7bjb.cif.gz 7bjb.cif.gz | 75.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7bjb.ent.gz pdb7bjb.ent.gz | 52.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7bjb.json.gz 7bjb.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bj/7bjb https://data.pdbj.org/pub/pdb/validation_reports/bj/7bjb ftp://data.pdbj.org/pub/pdb/validation_reports/bj/7bjb ftp://data.pdbj.org/pub/pdb/validation_reports/bj/7bjb | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7bi3C  7biqC  7biwC  7biyC  7bjfC  7bjlC  7bjwC  7bkhC  7nj9C  7njbC  7nk3C  7nk5C  7nlaC  7nleC  7nm1C  7nm3C  7nm9C  7nmhC  7nr7C  7nv4C  7nviC  7nwsC  7nxsC  7nxtC  7nxwC  7nxyC  7ny4C  7nyeC  7nyfC  7nygC  7nz6C  7nzgC  7nzkC  7nzvC  7o34C  7o3aC  7o3fC  7o3pC  7o3qC  7o3rC  7o3sC  7o57C  7o59C  7o5aC  7o5cC  7o5dC  7o5fC  7o5gC  7o5oC  7o5pC  7o5sC  7o5uC  7o5xC  7o6fC  7o6gC  7o6iC  7o6jC  7o6kC  7o6mC  7o6oC  6qhlS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
-Protein / Protein/peptide , 2 types, 2 molecules AP
| #1: Protein | Mass: 28226.518 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: SFN, HME1 / Production host: Homo sapiens (human) / Gene: SFN, HME1 / Production host:   Escherichia coli (E. coli) / References: UniProt: P31947 Escherichia coli (E. coli) / References: UniProt: P31947 |
|---|---|
| #2: Protein/peptide | Mass: 1412.429 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)   Homo sapiens (human) / References: UniProt: Q04206 Homo sapiens (human) / References: UniProt: Q04206 |
-Non-polymers , 6 types, 321 molecules 










| #3: Chemical | ChemComp-CL /  Chloride Chloride | ||||
|---|---|---|---|---|---|
| #4: Chemical | ChemComp-TZK / | ||||
| #5: Chemical | ChemComp-GOL /  Glycerol Glycerol | ||||
| #6: Chemical | | #7: Chemical | ChemComp-MG / | #8: Water | ChemComp-HOH / |  Water Water |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.44 Å3/Da / Density % sol: 49.66 % / Mosaicity: 0 ° |
|---|---|
Crystal grow | Temperature: 277 K / Method: vapor diffusion, hanging drop / pH: 7.1 Details: 0.095 M HEPES Na pH 7.1, 27% PEG400, 0.19M Calcium chloride, 5% Glycerol |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source: SEALED TUBE / Type: RIGAKU MICROMAX-003 / Wavelength: 1.54187 Å | ||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS 200K / Detector: PIXEL / Date: Dec 19, 2018 | ||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength : 1.54187 Å / Relative weight: 1 : 1.54187 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||
| Reflection | Resolution: 1.8→34.44 Å / Num. obs: 27297 / % possible obs: 99.9 % / Redundancy: 6.1 % / Biso Wilson estimate: 12.05 Å2 / CC1/2: 0.999 / Rmerge(I) obs: 0.048 / Rpim(I) all: 0.021 / Rrim(I) all: 0.052 / Net I/σ(I): 25.9 | ||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 6QHL Resolution: 1.8→34.44 Å / SU ML: 0.18 / Cross valid method: FREE R-VALUE / σ(F): 1.52 / Phase error: 21.33 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 71.62 Å2 / Biso mean: 16.6513 Å2 / Biso min: 4.02 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 1.8→34.44 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0
|
 Movie
Movie Controller
Controller



















 PDBj
PDBj





























