+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6zk6 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Protein Phosphatase 1 (PP1) T320E mutant | ||||||
 Components Components | Serine/threonine-protein phosphatase PP1-alpha catalytic subunit | ||||||
 Keywords Keywords |  HYDROLASE / protein phosphatase 1 regulation / HYDROLASE / protein phosphatase 1 regulation /  phosphorylation / phosphomimetic mutant phosphorylation / phosphomimetic mutant | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of glycogen catabolic process / PTW/PP1 phosphatase complex /  glycogen granule / regulation of glycogen biosynthetic process / glycogen granule / regulation of glycogen biosynthetic process /  protein phosphatase 1 binding / cadherin binding involved in cell-cell adhesion / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / regulation of canonical Wnt signaling pathway / regulation of translational initiation / myosin phosphatase activity ...regulation of glycogen catabolic process / PTW/PP1 phosphatase complex / protein phosphatase 1 binding / cadherin binding involved in cell-cell adhesion / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / regulation of canonical Wnt signaling pathway / regulation of translational initiation / myosin phosphatase activity ...regulation of glycogen catabolic process / PTW/PP1 phosphatase complex /  glycogen granule / regulation of glycogen biosynthetic process / glycogen granule / regulation of glycogen biosynthetic process /  protein phosphatase 1 binding / cadherin binding involved in cell-cell adhesion / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / regulation of canonical Wnt signaling pathway / regulation of translational initiation / myosin phosphatase activity / protein phosphatase 1 binding / cadherin binding involved in cell-cell adhesion / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / regulation of canonical Wnt signaling pathway / regulation of translational initiation / myosin phosphatase activity /  protein serine/threonine phosphatase activity / branching morphogenesis of an epithelial tube / glycogen metabolic process / protein-serine/threonine phosphatase / entrainment of circadian clock by photoperiod / Triglyceride catabolism / protein serine/threonine phosphatase activity / branching morphogenesis of an epithelial tube / glycogen metabolic process / protein-serine/threonine phosphatase / entrainment of circadian clock by photoperiod / Triglyceride catabolism /  phosphatase activity / phosphatase activity /  phosphoprotein phosphatase activity / DARPP-32 events / ribonucleoprotein complex binding / phosphoprotein phosphatase activity / DARPP-32 events / ribonucleoprotein complex binding /  dephosphorylation / protein dephosphorylation / Downregulation of TGF-beta receptor signaling / response to lead ion / dephosphorylation / protein dephosphorylation / Downregulation of TGF-beta receptor signaling / response to lead ion /  adherens junction / lung development / circadian regulation of gene expression / adherens junction / lung development / circadian regulation of gene expression /  regulation of circadian rhythm / regulation of circadian rhythm /  Circadian Clock / presynapse / Circadian Clock / presynapse /  perikaryon / perikaryon /  dendritic spine / dendritic spine /  cell cycle / cell cycle /  cell division / glutamatergic synapse / cell division / glutamatergic synapse /  nucleolus / extracellular exosome / nucleolus / extracellular exosome /  nucleoplasm / nucleoplasm /  metal ion binding / metal ion binding /  nucleus / nucleus /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.9 Å MOLECULAR REPLACEMENT / Resolution: 1.9 Å | ||||||
 Authors Authors | Salvi, F. / Barabas, O. / Koehn, M. | ||||||
| Funding support | European Union, 1items
| ||||||
 Citation Citation |  Journal: Chembiochem / Year: 2021 Journal: Chembiochem / Year: 2021Title: Towards Dissecting the Mechanism of Protein Phosphatase-1 Inhibition by Its C-Terminal Phosphorylation. Authors: Salvi, F. / Hoermann, B. / Del Pino Garcia, J. / Fontanillo, M. / Derua, R. / Beullens, M. / Bollen, M. / Barabas, O. / Kohn, M. | ||||||
| History |
|
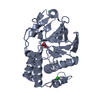
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6zk6.cif.gz 6zk6.cif.gz | 161.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6zk6.ent.gz pdb6zk6.ent.gz | 104.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6zk6.json.gz 6zk6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/zk/6zk6 https://data.pdbj.org/pub/pdb/validation_reports/zk/6zk6 ftp://data.pdbj.org/pub/pdb/validation_reports/zk/6zk6 ftp://data.pdbj.org/pub/pdb/validation_reports/zk/6zk6 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6g0jS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
| ||||||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 37691.113 Da / Num. of mol.: 1 / Mutation: T320E Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: PPP1CA, PPP1A / Production host: Homo sapiens (human) / Gene: PPP1CA, PPP1A / Production host:   Escherichia coli (E. coli) Escherichia coli (E. coli)References: UniProt: P62136, protein-serine/threonine phosphatase | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| #2: Chemical | | #3: Chemical |  Iron Iron#4: Chemical | ChemComp-PO4 / |  Phosphate Phosphate#5: Water | ChemComp-HOH / |  Water WaterHas ligand of interest | N | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.25 Å3/Da / Density % sol: 45.26 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 8 / Details: PEG 3350, TRIS , lithium chloride |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: MASSIF-3 / Wavelength: 0.9677 Å / Beamline: MASSIF-3 / Wavelength: 0.9677 Å |
| Detector | Type: DECTRIS EIGER X 4M / Detector: PIXEL / Date: Nov 30, 2016 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.9677 Å / Relative weight: 1 : 0.9677 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→36.22 Å / Num. obs: 26672 / % possible obs: 97.3 % / Redundancy: 3.4 % / Biso Wilson estimate: 34.47 Å2 / CC1/2: 0.999 / Rmerge(I) obs: 0.057 / Rpim(I) all: 0.033 / Rrim(I) all: 0.066 / Net I/σ(I): 11 |
| Reflection shell | Resolution: 1.9→1.94 Å / Rmerge(I) obs: 0.744 / Mean I/σ(I) obs: 1.5 / Num. unique obs: 1722 / CC1/2: 0.603 / Rpim(I) all: 0.433 / Rrim(I) all: 0.866 / % possible all: 98.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 6G0J Resolution: 1.9→36.22 Å / SU ML: 0.1812 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 20.2815 Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 43.57 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.9→36.22 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj














