+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6ya6 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Minimal construct of Cdc7-Dbf4 bound to XL413 | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  CELL CYCLE / CELL CYCLE /  kinase / kinase /  cdc7 / cdc7 /  dbf4 / bivalent substrate / dbf4 / bivalent substrate /  transferase transferase | ||||||
| Function / homology |  Function and homology information Function and homology informationDbf4-dependent protein kinase complex / regulation of cell cycle phase transition / positive regulation of nuclear cell cycle DNA replication / mitotic DNA damage checkpoint signaling / cell cycle phase transition / double-strand break repair via break-induced replication /  intercellular bridge / Transcriptional Regulation by E2F6 / Activation of the pre-replicative complex / intercellular bridge / Transcriptional Regulation by E2F6 / Activation of the pre-replicative complex /  enzyme activator activity ...Dbf4-dependent protein kinase complex / regulation of cell cycle phase transition / positive regulation of nuclear cell cycle DNA replication / mitotic DNA damage checkpoint signaling / cell cycle phase transition / double-strand break repair via break-induced replication / enzyme activator activity ...Dbf4-dependent protein kinase complex / regulation of cell cycle phase transition / positive regulation of nuclear cell cycle DNA replication / mitotic DNA damage checkpoint signaling / cell cycle phase transition / double-strand break repair via break-induced replication /  intercellular bridge / Transcriptional Regulation by E2F6 / Activation of the pre-replicative complex / intercellular bridge / Transcriptional Regulation by E2F6 / Activation of the pre-replicative complex /  enzyme activator activity / Activation of ATR in response to replication stress / positive regulation of G2/M transition of mitotic cell cycle / protein serine/threonine kinase activator activity / G1/S transition of mitotic cell cycle / enzyme activator activity / Activation of ATR in response to replication stress / positive regulation of G2/M transition of mitotic cell cycle / protein serine/threonine kinase activator activity / G1/S transition of mitotic cell cycle /  mitotic spindle / mitotic spindle /  kinase activity / kinase activity /  DNA replication / DNA replication /  nucleic acid binding / nucleic acid binding /  nuclear body / nuclear body /  non-specific serine/threonine protein kinase / non-specific serine/threonine protein kinase /  protein kinase activity / protein kinase activity /  phosphorylation / phosphorylation /  cell division / protein serine kinase activity / protein serine/threonine kinase activity / positive regulation of cell population proliferation / cell division / protein serine kinase activity / protein serine/threonine kinase activity / positive regulation of cell population proliferation /  signal transduction / zinc ion binding / signal transduction / zinc ion binding /  nucleoplasm / nucleoplasm /  ATP binding / ATP binding /  metal ion binding / metal ion binding /  nucleus / nucleus /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.44 Å MOLECULAR REPLACEMENT / Resolution: 1.44 Å | ||||||
 Authors Authors | Dick, S.D. / Cherepanov, P. | ||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| ||||||
 Citation Citation |  Journal: Structure / Year: 2020 Journal: Structure / Year: 2020Title: Structural Basis for the Activation and Target Site Specificity of CDC7 Kinase. Authors: Dick, S.D. / Federico, S. / Hughes, S.M. / Pye, V.E. / O'Reilly, N. / Cherepanov, P. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6ya6.cif.gz 6ya6.cif.gz | 243.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6ya6.ent.gz pdb6ya6.ent.gz | 157.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6ya6.json.gz 6ya6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ya/6ya6 https://data.pdbj.org/pub/pdb/validation_reports/ya/6ya6 ftp://data.pdbj.org/pub/pdb/validation_reports/ya/6ya6 ftp://data.pdbj.org/pub/pdb/validation_reports/ya/6ya6 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6ya7C  6ya8C  5mh9 S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
|
- Components
Components
-Protein , 2 types, 2 molecules AB
| #1: Protein | Mass: 40230.449 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: CDC7, CDC7L1 / Production host: Homo sapiens (human) / Gene: CDC7, CDC7L1 / Production host:   Escherichia coli (E. coli) Escherichia coli (E. coli)References: UniProt: O00311,  non-specific serine/threonine protein kinase non-specific serine/threonine protein kinase |
|---|---|
| #2: Protein | Mass: 17003.393 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: DBF4, ASK, DBF4A, ZDBF1 / Production host: Homo sapiens (human) / Gene: DBF4, ASK, DBF4A, ZDBF1 / Production host:   Escherichia coli (E. coli) / References: UniProt: Q9UBU7 Escherichia coli (E. coli) / References: UniProt: Q9UBU7 |
-Non-polymers , 7 types, 471 molecules 


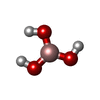









| #3: Chemical | ChemComp-0SX / | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| #4: Chemical | | #5: Chemical | ChemComp-PEG / |  Diethylene glycol Diethylene glycol#6: Chemical | ChemComp-BO3 / |  Boric acid Boric acid#7: Chemical | ChemComp-CL / |  Chloride Chloride#8: Chemical | ChemComp-IMD / |  Imidazole Imidazole#9: Water | ChemComp-HOH / |  Water Water |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.15 Å3/Da / Density % sol: 42.72 % |
|---|---|
Crystal grow | Temperature: 291.15 K / Method: vapor diffusion, hanging drop / pH: 7 / Details: 0.1M PCTP pH7, 18% PEG 1500, 8% MPD |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: BM14 / Wavelength: 1.0332 Å / Beamline: BM14 / Wavelength: 1.0332 Å |
| Detector | Type: MARMOSAIC 225 mm CCD / Detector: CCD / Date: Nov 21, 2013 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.0332 Å / Relative weight: 1 : 1.0332 Å / Relative weight: 1 |
| Reflection | Resolution: 1.44→59.61 Å / Num. obs: 82613 / % possible obs: 100 % / Redundancy: 9.1 % / Biso Wilson estimate: 13.58 Å2 / CC1/2: 1 / Rmerge(I) obs: 0.049 / Net I/σ(I): 26.4 |
| Reflection shell | Resolution: 1.44→1.491 Å / Rmerge(I) obs: 0.584 / Num. unique obs: 7764 / CC1/2: 0.77 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5MH9  5mh9 Resolution: 1.44→42.85 Å / SU ML: 0.1286 / Cross valid method: FREE R-VALUE / σ(F): 1.36 / Phase error: 17.0071
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 20.95 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.44→42.85 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj














