[English] 日本語
 Yorodumi
Yorodumi- PDB-5hrx: Crystal structure of the fifth bromodomain of human PB1 in comple... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5hrx | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the fifth bromodomain of human PB1 in complex with 1-butylisochromeno[3,4-c]pyrazol-5(2H)-one) compound | ||||||
 Components Components | Protein polybromo-1 | ||||||
 Keywords Keywords |  TRANSCRIPTION / TRANSCRIPTION /  PBRM1 / BRG1-associated factor 180 / PBRM1 / BRG1-associated factor 180 /  chromatin remodeling chromatin remodeling | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of G0 to G1 transition / regulation of nucleotide-excision repair / RSC-type complex / regulation of mitotic metaphase/anaphase transition /  SWI/SNF complex / positive regulation of double-strand break repair / positive regulation of T cell differentiation / nuclear chromosome / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / regulation of G1/S transition of mitotic cell cycle ...regulation of G0 to G1 transition / regulation of nucleotide-excision repair / RSC-type complex / regulation of mitotic metaphase/anaphase transition / SWI/SNF complex / positive regulation of double-strand break repair / positive regulation of T cell differentiation / nuclear chromosome / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / regulation of G1/S transition of mitotic cell cycle ...regulation of G0 to G1 transition / regulation of nucleotide-excision repair / RSC-type complex / regulation of mitotic metaphase/anaphase transition /  SWI/SNF complex / positive regulation of double-strand break repair / positive regulation of T cell differentiation / nuclear chromosome / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / regulation of G1/S transition of mitotic cell cycle / positive regulation of myoblast differentiation / transcription elongation by RNA polymerase II / positive regulation of cell differentiation / SWI/SNF complex / positive regulation of double-strand break repair / positive regulation of T cell differentiation / nuclear chromosome / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / regulation of G1/S transition of mitotic cell cycle / positive regulation of myoblast differentiation / transcription elongation by RNA polymerase II / positive regulation of cell differentiation /  kinetochore / RMTs methylate histone arginines / kinetochore / RMTs methylate histone arginines /  nuclear matrix / mitotic cell cycle / nuclear matrix / mitotic cell cycle /  chromatin remodeling / negative regulation of cell population proliferation / chromatin remodeling / negative regulation of cell population proliferation /  chromatin binding / chromatin binding /  chromatin / regulation of transcription by RNA polymerase II / chromatin / regulation of transcription by RNA polymerase II /  DNA binding / DNA binding /  nucleoplasm / nucleoplasm /  nucleus nucleusSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.73 Å MOLECULAR REPLACEMENT / Resolution: 1.73 Å | ||||||
 Authors Authors | Tallant, C. / Myrianthopoulos, V. / Gaboriaud-Kolar, N. / Newman, J.A. / Picaud, S. / von Delft, F. / Arrowsmith, C.H. / Edwards, A.M. / Bountra, C. / Mikros, E. / Knapp, S. | ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 2016 Journal: J.Med.Chem. / Year: 2016Title: Discovery and Optimization of a Selective Ligand for the Switch/Sucrose Nonfermenting-Related Bromodomains of Polybromo Protein-1 by the Use of Virtual Screening and Hydration Analysis. Authors: Myrianthopoulos, V. / Gaboriaud-Kolar, N. / Tallant, C. / Hall, M.L. / Grigoriou, S. / Brownlee, P.M. / Fedorov, O. / Rogers, C. / Heidenreich, D. / Wanior, M. / Drosos, N. / Mexia, N. / ...Authors: Myrianthopoulos, V. / Gaboriaud-Kolar, N. / Tallant, C. / Hall, M.L. / Grigoriou, S. / Brownlee, P.M. / Fedorov, O. / Rogers, C. / Heidenreich, D. / Wanior, M. / Drosos, N. / Mexia, N. / Savitsky, P. / Bagratuni, T. / Kastritis, E. / Terpos, E. / Filippakopoulos, P. / Muller, S. / Skaltsounis, A.L. / Downs, J.A. / Knapp, S. / Mikros, E. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5hrx.cif.gz 5hrx.cif.gz | 105.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5hrx.ent.gz pdb5hrx.ent.gz | 81.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5hrx.json.gz 5hrx.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hr/5hrx https://data.pdbj.org/pub/pdb/validation_reports/hr/5hrx ftp://data.pdbj.org/pub/pdb/validation_reports/hr/5hrx ftp://data.pdbj.org/pub/pdb/validation_reports/hr/5hrx | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5hrvC  5hrwC  5ii1C  5ii2C  5iidC  3g0jS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 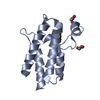
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 14648.000 Da / Num. of mol.: 2 / Fragment: UNP Residues 613-734 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: PBRM1, BAF180, PB1 / Production host: Homo sapiens (human) / Gene: PBRM1, BAF180, PB1 / Production host:   Escherichia coli (E. coli) / References: UniProt: Q86U86 Escherichia coli (E. coli) / References: UniProt: Q86U86#2: Chemical |  Ethylene glycol Ethylene glycol#3: Chemical | #4: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.18 Å3/Da / Density % sol: 43.51 % / Description: Plate crystals |
|---|---|
Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 7.5 Details: 0.1M lithium sulfate monohydrate, 0.1 M Tris pH 8.5, 30% PEG4K PH range: 7.5-8.5 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I04-1 / Wavelength: 0.9207 Å / Beamline: I04-1 / Wavelength: 0.9207 Å |
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: Mar 2, 2014 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.9207 Å / Relative weight: 1 : 0.9207 Å / Relative weight: 1 |
| Reflection | Resolution: 1.73→29.21 Å / Num. all: 26823 / Num. obs: 26823 / % possible obs: 97.8 % / Redundancy: 5.6 % / Rmerge(I) obs: 0.051 / Rsym value: 0.026 / Net I/σ(I): 17.3 |
| Reflection shell | Resolution: 1.73→1.76 Å / Redundancy: 5 % / Rmerge(I) obs: 0.893 / Mean I/σ(I) obs: 1.8 / % possible all: 96.3 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 3G0J Resolution: 1.73→29.21 Å / Cor.coef. Fo:Fc: 0.946 / Cor.coef. Fo:Fc free: 0.931 / SU B: 5.419 / SU ML: 0.089 / Cross valid method: THROUGHOUT / ESU R: 0.125 / ESU R Free: 0.124 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 32.679 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 1.73→29.21 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj








