+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5gmv | ||||||
|---|---|---|---|---|---|---|---|
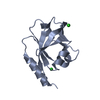
| Title | LC3B-FUNDC1 complex | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  PROTEIN BINDING / PROTEIN BINDING /  LC3B / FUNDC1 / specific recognition / LC3B / FUNDC1 / specific recognition /  phosphorylation / selective mitophagy phosphorylation / selective mitophagy | ||||||
| Function / homology |  Function and homology information Function and homology informationSARS-CoV-2 modulates autophagy /  ceramide binding / autophagy of mitochondrion / cellular response to nitrogen starvation / ceramide binding / autophagy of mitochondrion / cellular response to nitrogen starvation /  phosphatidylethanolamine binding / Translation of Replicase and Assembly of the Replication Transcription Complex / TBC/RABGAPs / phosphatidylethanolamine binding / Translation of Replicase and Assembly of the Replication Transcription Complex / TBC/RABGAPs /  Macroautophagy / Receptor Mediated Mitophagy / Macroautophagy / Receptor Mediated Mitophagy /  axoneme ...SARS-CoV-2 modulates autophagy / axoneme ...SARS-CoV-2 modulates autophagy /  ceramide binding / autophagy of mitochondrion / cellular response to nitrogen starvation / ceramide binding / autophagy of mitochondrion / cellular response to nitrogen starvation /  phosphatidylethanolamine binding / Translation of Replicase and Assembly of the Replication Transcription Complex / TBC/RABGAPs / phosphatidylethanolamine binding / Translation of Replicase and Assembly of the Replication Transcription Complex / TBC/RABGAPs /  Macroautophagy / Receptor Mediated Mitophagy / Macroautophagy / Receptor Mediated Mitophagy /  axoneme / autophagosome membrane / organelle membrane / autophagosome maturation / axoneme / autophagosome membrane / organelle membrane / autophagosome maturation /  mitophagy / mitophagy /  autophagosome assembly / autophagosome assembly /  autophagosome / autophagosome /  endomembrane system / PINK1-PRKN Mediated Mitophagy / Pexophagy / cellular response to starvation / endomembrane system / PINK1-PRKN Mediated Mitophagy / Pexophagy / cellular response to starvation /  mitochondrial membrane / mitochondrial membrane /  macroautophagy / macroautophagy /  autophagy / KEAP1-NFE2L2 pathway / Translation of Replicase and Assembly of the Replication Transcription Complex / cytoplasmic vesicle / autophagy / KEAP1-NFE2L2 pathway / Translation of Replicase and Assembly of the Replication Transcription Complex / cytoplasmic vesicle /  microtubule binding / microtubule binding /  microtubule / mitochondrial outer membrane / response to hypoxia / intracellular membrane-bounded organelle / microtubule / mitochondrial outer membrane / response to hypoxia / intracellular membrane-bounded organelle /  ubiquitin protein ligase binding / ubiquitin protein ligase binding /  mitochondrion / mitochondrion /  cytosol cytosolSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.25 Å MOLECULAR REPLACEMENT / Resolution: 2.25 Å | ||||||
 Authors Authors | Lv, M. / Wang, C. / Li, F. | ||||||
 Citation Citation |  Journal: Protein Cell / Year: 2017 Journal: Protein Cell / Year: 2017Title: Structural insights into the recognition of phosphorylated FUNDC1 by LC3B in mitophagy Authors: Lv, M. / Wang, C. / Li, F. / Peng, J. / Wen, B. / Gong, Q. / Shi, Y. / Tang, Y. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5gmv.cif.gz 5gmv.cif.gz | 67.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5gmv.ent.gz pdb5gmv.ent.gz | 49.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5gmv.json.gz 5gmv.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/gm/5gmv https://data.pdbj.org/pub/pdb/validation_reports/gm/5gmv ftp://data.pdbj.org/pub/pdb/validation_reports/gm/5gmv ftp://data.pdbj.org/pub/pdb/validation_reports/gm/5gmv | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3vtuS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 14710.027 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: MAP1LC3B, MAP1ALC3 / Production host: Homo sapiens (human) / Gene: MAP1LC3B, MAP1ALC3 / Production host:   Escherichia coli K-12 (bacteria) / Strain (production host): K-12 / References: UniProt: Q9GZQ8 Escherichia coli K-12 (bacteria) / Strain (production host): K-12 / References: UniProt: Q9GZQ8#2: Protein/peptide |  / FUNDC1 peptide / FUNDC1 peptideMass: 1032.981 Da / Num. of mol.: 2 / Source method: obtained synthetically / Source: (synth.)   Homo sapiens (human) / References: UniProt: Q8IVP5 Homo sapiens (human) / References: UniProt: Q8IVP5#3: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.19 Å3/Da / Density % sol: 43.93 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / Details: 30% PEG MME 2000, 0.1mM sodium cacodylate (pH 6.0) |
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRF SSRF  / Beamline: BL17U / Wavelength: 0.979 Å / Beamline: BL17U / Wavelength: 0.979 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Dec 10, 2015 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength : 0.979 Å / Relative weight: 1 : 0.979 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.25→43.425 Å / Num. obs: 12052 / % possible obs: 96.8 % / Redundancy: 3.3 % / Rsym value: 0.076 / Net I/av σ(I): 4.728 / Net I/σ(I): 7.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell |
|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 3VTU Resolution: 2.25→43.42 Å / Cor.coef. Fo:Fc: 0.931 / Cor.coef. Fo:Fc free: 0.889 / SU B: 8.956 / SU ML: 0.22 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.452 / ESU R Free: 0.279 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS U VALUES : REFINED INDIVIDUALLY
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 84.08 Å2 / Biso mean: 33.026 Å2 / Biso min: 14.22 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.25→43.42 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.25→2.308 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller












 PDBj
PDBj









