[English] 日本語
 Yorodumi
Yorodumi- PDB-4mqs: Structure of active human M2 muscarinic acetylcholine receptor bo... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4mqs | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of active human M2 muscarinic acetylcholine receptor bound to the agonist iperoxo | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  SIGNALING PROTEIN / SIGNALING PROTEIN /  G protein-coupled receptor / G protein-coupled receptor /  Muscarinic acetylcholine receptor Muscarinic acetylcholine receptor | ||||||
| Function / homology |  Function and homology information Function and homology information Muscarinic acetylcholine receptors / symmetric synapse / phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway / G protein-coupled acetylcholine receptor activity / regulation of smooth muscle contraction / cholinergic synapse / adenylate cyclase-inhibiting G protein-coupled acetylcholine receptor signaling pathway / G protein-coupled serotonin receptor activity / arrestin family protein binding / Muscarinic acetylcholine receptors / symmetric synapse / phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway / G protein-coupled acetylcholine receptor activity / regulation of smooth muscle contraction / cholinergic synapse / adenylate cyclase-inhibiting G protein-coupled acetylcholine receptor signaling pathway / G protein-coupled serotonin receptor activity / arrestin family protein binding /  regulation of heart contraction ... regulation of heart contraction ... Muscarinic acetylcholine receptors / symmetric synapse / phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway / G protein-coupled acetylcholine receptor activity / regulation of smooth muscle contraction / cholinergic synapse / adenylate cyclase-inhibiting G protein-coupled acetylcholine receptor signaling pathway / G protein-coupled serotonin receptor activity / arrestin family protein binding / Muscarinic acetylcholine receptors / symmetric synapse / phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway / G protein-coupled acetylcholine receptor activity / regulation of smooth muscle contraction / cholinergic synapse / adenylate cyclase-inhibiting G protein-coupled acetylcholine receptor signaling pathway / G protein-coupled serotonin receptor activity / arrestin family protein binding /  regulation of heart contraction / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / asymmetric synapse / axon terminus / presynaptic modulation of chemical synaptic transmission / clathrin-coated endocytic vesicle membrane / response to virus / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G protein-coupled acetylcholine receptor signaling pathway / Cargo recognition for clathrin-mediated endocytosis / regulation of heart contraction / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / asymmetric synapse / axon terminus / presynaptic modulation of chemical synaptic transmission / clathrin-coated endocytic vesicle membrane / response to virus / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G protein-coupled acetylcholine receptor signaling pathway / Cargo recognition for clathrin-mediated endocytosis /  presynaptic membrane / presynaptic membrane /  Clathrin-mediated endocytosis / Clathrin-mediated endocytosis /  nervous system development / G alpha (i) signalling events / chemical synaptic transmission / nervous system development / G alpha (i) signalling events / chemical synaptic transmission /  postsynaptic membrane / G protein-coupled receptor signaling pathway / neuronal cell body / postsynaptic membrane / G protein-coupled receptor signaling pathway / neuronal cell body /  dendrite / dendrite /  synapse / glutamatergic synapse / synapse / glutamatergic synapse /  membrane / membrane /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human)  Lama glama (llama) Lama glama (llama) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.5 Å MOLECULAR REPLACEMENT / Resolution: 3.5 Å | ||||||
 Authors Authors | Kruse, A.C. / Ring, A.M. / Manglik, A. / Hu, J. / Hu, K. / Eitel, K. / Huebner, H. / Pardon, E. / Valant, C. / Sexton, P.M. ...Kruse, A.C. / Ring, A.M. / Manglik, A. / Hu, J. / Hu, K. / Eitel, K. / Huebner, H. / Pardon, E. / Valant, C. / Sexton, P.M. / Christopoulos, A. / Felder, C.C. / Gmeiner, P. / Steyaert, J. / Weis, W.I. / Garcia, K.C. / Wess, J. / Kobilka, B.K. | ||||||
 Citation Citation |  Journal: Nature / Year: 2013 Journal: Nature / Year: 2013Title: Activation and allosteric modulation of a muscarinic acetylcholine receptor. Authors: Kruse, A.C. / Ring, A.M. / Manglik, A. / Hu, J. / Hu, K. / Eitel, K. / Hubner, H. / Pardon, E. / Valant, C. / Sexton, P.M. / Christopoulos, A. / Felder, C.C. / Gmeiner, P. / Steyaert, J. / ...Authors: Kruse, A.C. / Ring, A.M. / Manglik, A. / Hu, J. / Hu, K. / Eitel, K. / Hubner, H. / Pardon, E. / Valant, C. / Sexton, P.M. / Christopoulos, A. / Felder, C.C. / Gmeiner, P. / Steyaert, J. / Weis, W.I. / Garcia, K.C. / Wess, J. / Kobilka, B.K. | ||||||
| History |
| ||||||
| Remark 650 | HELIX DETERMINATION METHOD: AUTHOR DETERMINED |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4mqs.cif.gz 4mqs.cif.gz | 93.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4mqs.ent.gz pdb4mqs.ent.gz | 67.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4mqs.json.gz 4mqs.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mq/4mqs https://data.pdbj.org/pub/pdb/validation_reports/mq/4mqs ftp://data.pdbj.org/pub/pdb/validation_reports/mq/4mqs ftp://data.pdbj.org/pub/pdb/validation_reports/mq/4mqs | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4mqtC  3uonS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein |  Mass: 39951.734 Da / Num. of mol.: 1 / Fragment: UNP residues 1-232, 373-466 / Mutation: N0D, N1D, N4D, N7D, A373T, K374R Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: CHRM2 / Plasmid: PVL1392 / Production host: Homo sapiens (human) / Gene: CHRM2 / Plasmid: PVL1392 / Production host:   Spodoptera frugiperda (fall armyworm) / References: UniProt: P08172 Spodoptera frugiperda (fall armyworm) / References: UniProt: P08172 |
|---|---|
| #2: Antibody | Mass: 13428.396 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Lama glama (llama) / Plasmid: pMAL3CBN / Production host: Lama glama (llama) / Plasmid: pMAL3CBN / Production host:   Escherichia coli (E. coli) / Strain (production host): BL21(DE3) Escherichia coli (E. coli) / Strain (production host): BL21(DE3) |
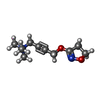
| #3: Chemical | ChemComp-IXO / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.76 Å3/Da / Density % sol: 67.31 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: lipidic cubic phase Details: Reconstituted in 10:1 monoolein:cholesterol mix. Precipitant solution: 10 - 20% PEG300, 100 mM HEPES, pH 7.2 - 7.9, 1.2% 1,2,3-heptanetriol, and 20 - 80 mM EDTA, pH 8.0, Lipidic cubic phase, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 77 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 23-ID-D / Wavelength: 1.033 Å / Beamline: 23-ID-D / Wavelength: 1.033 Å |
| Detector | Type: MARMOSAIC 300 mm CCD / Detector: CCD / Date: Feb 23, 2013 / Details: mirrors |
| Radiation | Monochromator: Double crystal cryo-cooled Si(111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.033 Å / Relative weight: 1 : 1.033 Å / Relative weight: 1 |
| Reflection | Resolution: 3.5→33 Å / Num. all: 10386 / Num. obs: 10386 / % possible obs: 95.9 % / Observed criterion σ(I): -3 / Redundancy: 4.8 % / Rmerge(I) obs: 0.188 |
| Reflection shell | Resolution: 3.5→3.6 Å / Redundancy: 2.5 % / Rmerge(I) obs: 0.744 / Num. unique all: 871 / % possible all: 83.1 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 3UON Resolution: 3.5→32.522 Å / SU ML: 0.57 / Cross valid method: THROUGHOUT / σ(F): 1.38 / Phase error: 35.28 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.5→32.522 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj