[English] 日本語
 Yorodumi
Yorodumi- PDB-4jdd: 14-3-3 protein interaction with Estrogen Receptor Alpha provides ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4jdd | ||||||
|---|---|---|---|---|---|---|---|
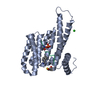
| Title | 14-3-3 protein interaction with Estrogen Receptor Alpha provides a novel drug target interface | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  SIGNALING PROTEIN/Peptide / SIGNALING PROTEIN/Peptide /  14-3-3 / 14-3-3 /  ADAPTER PROTEIN / ADAPTER PROTEIN /  PROTEIN-PROTEIN INTERACTION / PROTEIN-PROTEIN INTERACTION /  SIGNALING PROTEIN-Peptide complex SIGNALING PROTEIN-Peptide complex | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of epithelial cell apoptotic process / antral ovarian follicle growth / G protein-coupled estrogen receptor activity / regulation of branching involved in prostate gland morphogenesis / RUNX1 regulates transcription of genes involved in WNT signaling / RUNX1 regulates estrogen receptor mediated transcription /  regulation of toll-like receptor signaling pathway / regulation of epidermal cell division / prostate epithelial cord arborization involved in prostate glandular acinus morphogenesis / regulation of toll-like receptor signaling pathway / regulation of epidermal cell division / prostate epithelial cord arborization involved in prostate glandular acinus morphogenesis /  protein kinase C inhibitor activity ...regulation of epithelial cell apoptotic process / antral ovarian follicle growth / G protein-coupled estrogen receptor activity / regulation of branching involved in prostate gland morphogenesis / RUNX1 regulates transcription of genes involved in WNT signaling / RUNX1 regulates estrogen receptor mediated transcription / protein kinase C inhibitor activity ...regulation of epithelial cell apoptotic process / antral ovarian follicle growth / G protein-coupled estrogen receptor activity / regulation of branching involved in prostate gland morphogenesis / RUNX1 regulates transcription of genes involved in WNT signaling / RUNX1 regulates estrogen receptor mediated transcription /  regulation of toll-like receptor signaling pathway / regulation of epidermal cell division / prostate epithelial cord arborization involved in prostate glandular acinus morphogenesis / regulation of toll-like receptor signaling pathway / regulation of epidermal cell division / prostate epithelial cord arborization involved in prostate glandular acinus morphogenesis /  protein kinase C inhibitor activity / nuclear estrogen receptor activity / epithelial cell proliferation involved in mammary gland duct elongation / positive regulation of epidermal cell differentiation / keratinocyte development / protein kinase C inhibitor activity / nuclear estrogen receptor activity / epithelial cell proliferation involved in mammary gland duct elongation / positive regulation of epidermal cell differentiation / keratinocyte development /  keratinization / prostate epithelial cord elongation / epithelial cell development / negative regulation of smooth muscle cell apoptotic process / mammary gland branching involved in pregnancy / uterus development / vagina development / androgen metabolic process / TFIIB-class transcription factor binding / steroid hormone mediated signaling pathway / Regulation of localization of FOXO transcription factors / keratinocyte proliferation / phosphoserine residue binding / Activation of BAD and translocation to mitochondria / negative regulation of keratinocyte proliferation / mammary gland alveolus development / intracellular estrogen receptor signaling pathway / establishment of skin barrier / cellular response to estrogen stimulus / keratinization / prostate epithelial cord elongation / epithelial cell development / negative regulation of smooth muscle cell apoptotic process / mammary gland branching involved in pregnancy / uterus development / vagina development / androgen metabolic process / TFIIB-class transcription factor binding / steroid hormone mediated signaling pathway / Regulation of localization of FOXO transcription factors / keratinocyte proliferation / phosphoserine residue binding / Activation of BAD and translocation to mitochondria / negative regulation of keratinocyte proliferation / mammary gland alveolus development / intracellular estrogen receptor signaling pathway / establishment of skin barrier / cellular response to estrogen stimulus /  estrogen response element binding / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / : / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex / intracellular steroid hormone receptor signaling pathway / negative regulation of canonical NF-kappaB signal transduction / protein kinase A signaling / negative regulation of stem cell proliferation / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / Nuclear signaling by ERBB4 / RNA polymerase II preinitiation complex assembly / RHO GTPases activate PKNs / protein localization to chromatin / TBP-class protein binding / TFAP2 (AP-2) family regulates transcription of growth factors and their receptors / estrogen response element binding / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / : / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex / intracellular steroid hormone receptor signaling pathway / negative regulation of canonical NF-kappaB signal transduction / protein kinase A signaling / negative regulation of stem cell proliferation / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / Nuclear signaling by ERBB4 / RNA polymerase II preinitiation complex assembly / RHO GTPases activate PKNs / protein localization to chromatin / TBP-class protein binding / TFAP2 (AP-2) family regulates transcription of growth factors and their receptors /  steroid binding / protein export from nucleus / negative regulation of innate immune response / nitric-oxide synthase regulator activity / ESR-mediated signaling / protein sequestering activity / TP53 Regulates Transcription of Genes Involved in G2 Cell Cycle Arrest / release of cytochrome c from mitochondria / 14-3-3 protein binding / transcription corepressor binding / positive regulation of protein export from nucleus / negative regulation of miRNA transcription / stem cell proliferation / cellular response to estradiol stimulus / Translocation of SLC2A4 (GLUT4) to the plasma membrane / steroid binding / protein export from nucleus / negative regulation of innate immune response / nitric-oxide synthase regulator activity / ESR-mediated signaling / protein sequestering activity / TP53 Regulates Transcription of Genes Involved in G2 Cell Cycle Arrest / release of cytochrome c from mitochondria / 14-3-3 protein binding / transcription corepressor binding / positive regulation of protein export from nucleus / negative regulation of miRNA transcription / stem cell proliferation / cellular response to estradiol stimulus / Translocation of SLC2A4 (GLUT4) to the plasma membrane /  transcription coregulator binding / transcription coregulator binding /  stem cell differentiation / nuclear estrogen receptor binding / positive regulation of nitric-oxide synthase activity / TP53 Regulates Metabolic Genes / negative regulation of protein kinase activity / SUMOylation of intracellular receptors / stem cell differentiation / nuclear estrogen receptor binding / positive regulation of nitric-oxide synthase activity / TP53 Regulates Metabolic Genes / negative regulation of protein kinase activity / SUMOylation of intracellular receptors /  euchromatin / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process / negative regulation of DNA-binding transcription factor activity / euchromatin / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process / negative regulation of DNA-binding transcription factor activity /  transcription coactivator binding / transcription coactivator binding /  beta-catenin binding / Nuclear Receptor transcription pathway / response to estrogen / male gonad development / Constitutive Signaling by Aberrant PI3K in Cancer / Regulation of RUNX2 expression and activity / beta-catenin binding / Nuclear Receptor transcription pathway / response to estrogen / male gonad development / Constitutive Signaling by Aberrant PI3K in Cancer / Regulation of RUNX2 expression and activity /  nuclear receptor activity / positive regulation of nitric oxide biosynthetic process / intrinsic apoptotic signaling pathway in response to DNA damage / positive regulation of DNA-binding transcription factor activity / positive regulation of fibroblast proliferation / Ovarian tumor domain proteases / sequence-specific double-stranded DNA binding / response to estradiol / PIP3 activates AKT signaling / phospholipase C-activating G protein-coupled receptor signaling pathway / nuclear receptor activity / positive regulation of nitric oxide biosynthetic process / intrinsic apoptotic signaling pathway in response to DNA damage / positive regulation of DNA-binding transcription factor activity / positive regulation of fibroblast proliferation / Ovarian tumor domain proteases / sequence-specific double-stranded DNA binding / response to estradiol / PIP3 activates AKT signaling / phospholipase C-activating G protein-coupled receptor signaling pathway /  ATPase binding / positive regulation of cytosolic calcium ion concentration / ATPase binding / positive regulation of cytosolic calcium ion concentration /  regulation of inflammatory response / fibroblast proliferation / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / positive regulation of cell growth / DNA-binding transcription activator activity, RNA polymerase II-specific / Estrogen-dependent gene expression / regulation of inflammatory response / fibroblast proliferation / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / positive regulation of cell growth / DNA-binding transcription activator activity, RNA polymerase II-specific / Estrogen-dependent gene expression /  transcription regulator complex / Extra-nuclear estrogen signaling transcription regulator complex / Extra-nuclear estrogen signalingSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.1 Å molecular replacement / Resolution: 2.1 Å | ||||||
 Authors Authors | Bier, D. / Ottmann, C. | ||||||
 Citation Citation |  Journal: Proc. Natl. Acad. Sci. U.S.A. / Year: 2013 Journal: Proc. Natl. Acad. Sci. U.S.A. / Year: 2013Title: Interaction of 14-3-3 proteins with the estrogen receptor alpha F domain provides a drug target interface. Authors: De Vries-van Leeuwen, I.J. / da Costa Pereira, D. / Flach, K.D. / Piersma, S.R. / Haase, C. / Bier, D. / Yalcin, Z. / Michalides, R. / Feenstra, K.A. / Jimenez, C.R. / de Greef, T.F. / ...Authors: De Vries-van Leeuwen, I.J. / da Costa Pereira, D. / Flach, K.D. / Piersma, S.R. / Haase, C. / Bier, D. / Yalcin, Z. / Michalides, R. / Feenstra, K.A. / Jimenez, C.R. / de Greef, T.F. / Brunsveld, L. / Ottmann, C. / Zwart, W. / de Boer, A.H. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4jdd.cif.gz 4jdd.cif.gz | 59.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4jdd.ent.gz pdb4jdd.ent.gz | 46.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4jdd.json.gz 4jdd.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/jd/4jdd https://data.pdbj.org/pub/pdb/validation_reports/jd/4jdd ftp://data.pdbj.org/pub/pdb/validation_reports/jd/4jdd ftp://data.pdbj.org/pub/pdb/validation_reports/jd/4jdd | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 26542.914 Da / Num. of mol.: 1 / Fragment: UNP residues 1-231 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: HME1, NM_006142, SFN / Plasmid: PPROEX HTB / Production host: Homo sapiens (human) / Gene: HME1, NM_006142, SFN / Plasmid: PPROEX HTB / Production host:   Escherichia coli (E. coli) / Strain (production host): ROSETTA (DE3) / References: UniProt: P31947 Escherichia coli (E. coli) / Strain (production host): ROSETTA (DE3) / References: UniProt: P31947 |
|---|---|
| #2: Protein/peptide | Mass: 1158.109 Da / Num. of mol.: 1 / Fragment: UNP RESIDUES 585-595 / Source method: obtained synthetically / Source: (synth.)   Homo sapiens (human) / References: UniProt: P03372 Homo sapiens (human) / References: UniProt: P03372 |
| #3: Chemical | ChemComp-FSC /  Fusicoccin Fusicoccin |
| #4: Water | ChemComp-HOH /  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.58 Å3/Da / Density % sol: 52.35 % |
|---|---|
Crystal grow | Temperature: 277.15 K / Method: vapor diffusion, sitting drop / pH: 7.5 Details: 0.095M HEPES Na, 0.19M calcium chloride, 5% glycerol, 26.6% PEG400, pH 7.5, VAPOR DIFFUSION, SITTING DROP, temperature 277.15K |
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: BRUKER AXS MICROSTAR / Wavelength: 1.54 Å ROTATING ANODE / Type: BRUKER AXS MICROSTAR / Wavelength: 1.54 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: MAR scanner 345 mm plate / Detector: IMAGE PLATE / Date: Aug 20, 2012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength : 1.54 Å / Relative weight: 1 : 1.54 Å / Relative weight: 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.1→19.8 Å / Num. all: 17090 / Num. obs: 17090 / % possible obs: 99.8 % / Observed criterion σ(F): -3 / Observed criterion σ(I): -3 / Biso Wilson estimate: 23.566 Å2 / Rmerge(I) obs: 0.112 / Net I/σ(I): 17.79 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
Phasing | Method:  molecular replacement molecular replacement | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phasing MR | Model details: Phaser MODE: MR_AUTO
|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 2.1→19.8 Å / Cor.coef. Fo:Fc: 0.933 / Cor.coef. Fo:Fc free: 0.909 / WRfactor Rfree: 0.1977 / WRfactor Rwork: 0.1636 / Occupancy max: 1 / Occupancy min: 0 / FOM work R set: 0.8459 / SU B: 4.347 / SU ML: 0.119 / SU R Cruickshank DPI: 0.2073 / SU Rfree: 0.1758 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.207 / ESU R Free: 0.176 / Stereochemistry target values: MAXIMUM LIKELIHOOD MOLECULAR REPLACEMENT / Resolution: 2.1→19.8 Å / Cor.coef. Fo:Fc: 0.933 / Cor.coef. Fo:Fc free: 0.909 / WRfactor Rfree: 0.1977 / WRfactor Rwork: 0.1636 / Occupancy max: 1 / Occupancy min: 0 / FOM work R set: 0.8459 / SU B: 4.347 / SU ML: 0.119 / SU R Cruickshank DPI: 0.2073 / SU Rfree: 0.1758 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.207 / ESU R Free: 0.176 / Stereochemistry target values: MAXIMUM LIKELIHOODDetails: HYDROGENS HAVE BEEN USED IF PRESENT IN THE INPUT U VALUES: REFINED INDIVIDUALLY
| |||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 79.51 Å2 / Biso mean: 20.0007 Å2 / Biso min: 0.45 Å2
| |||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.1→19.8 Å
| |||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.1→2.154 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller


























 PDBj
PDBj