[English] 日本語
 Yorodumi
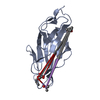
Yorodumi- PDB-4gpm: Crystal Structure of Engineered Protein. Northeast Structural Gen... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4gpm | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of Engineered Protein. Northeast Structural Genomics Consortium Target OR264. | ||||||
 Components Components | Engineered Protein OR264 Engineering Engineering | ||||||
 Keywords Keywords |  DE NOVO PROTEIN / DE NOVO PROTEIN /  Structural Genomics / PSI-Biology / Northeast Structural Genomics Consortium / NESG Structural Genomics / PSI-Biology / Northeast Structural Genomics Consortium / NESG | ||||||
| Function / homology | Ankyrin repeat-containing domain / Serine Threonine Protein Phosphatase 5, Tetratricopeptide repeat /  Alpha Horseshoe / Mainly Alpha Alpha Horseshoe / Mainly Alpha Function and homology information Function and homology information | ||||||
| Biological species | synthetic construct (others) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.002 Å MOLECULAR REPLACEMENT / Resolution: 2.002 Å | ||||||
 Authors Authors | Vorobiev, S. / Su, M. / Parmeggiani, F. / Seetharaman, J. / Huang, P.-S. / Maglaqui, M. / Xiao, R. / Lee, D. / Everett, J.K. / Acton, T.B. ...Vorobiev, S. / Su, M. / Parmeggiani, F. / Seetharaman, J. / Huang, P.-S. / Maglaqui, M. / Xiao, R. / Lee, D. / Everett, J.K. / Acton, T.B. / Baker, D. / Montelione, G.T. / Tong, L. / Hunt, J.F. / Northeast Structural Genomics Consortium (NESG) | ||||||
 Citation Citation |  Journal: NAT.CHEM. / Year: 2017 Journal: NAT.CHEM. / Year: 2017Title: Computational design of self-assembling cyclic protein homo-oligomers. Authors: Fallas, J.A. / Ueda, G. / Sheffler, W. / Nguyen, V. / McNamara, D.E. / Sankaran, B. / Pereira, J.H. / Parmeggiani, F. / Brunette, T.J. / Cascio, D. / Yeates, T.R. / Zwart, P. / Baker, D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4gpm.cif.gz 4gpm.cif.gz | 127.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4gpm.ent.gz pdb4gpm.ent.gz | 100.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4gpm.json.gz 4gpm.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/gp/4gpm https://data.pdbj.org/pub/pdb/validation_reports/gp/4gpm ftp://data.pdbj.org/pub/pdb/validation_reports/gp/4gpm ftp://data.pdbj.org/pub/pdb/validation_reports/gp/4gpm | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4gmrC  4hb5C  4hxtC  5hryC  5hrzC  5hs0C  5k7vC  5kbaC  5kwdC  2xeeS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data | |
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Details | monomer,19.62 kD,98.7% |
- Components
Components
| #1: Protein |  Engineering EngineeringMass: 18352.297 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.) synthetic construct (others) / Plasmid: pET21, OR264-21.1 / Production host:   Escherichia coli (E. coli) / Strain (production host): BL21(DE3)+ Magic Escherichia coli (E. coli) / Strain (production host): BL21(DE3)+ Magic#2: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.97 Å3/Da / Density % sol: 37.41 % |
|---|---|
Crystal grow | Temperature: 291 K / Method: microbatch crystallization under oil / pH: 9 Details: 40% PEG 1000, 0.1M lithium chloride, 0.1M TAPS, pH 9.0 , Microbatch crystallization under oil, temperature 291K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X4C / Wavelength: 0.97915 Å / Beamline: X4C / Wavelength: 0.97915 Å |
| Detector | Type: MAR CCD 165 mm / Detector: CCD / Date: Aug 9, 2012 |
| Radiation | Monochromator: Si 111 CHANNEL / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.97915 Å / Relative weight: 1 : 0.97915 Å / Relative weight: 1 |
| Reflection | Resolution: 1.99→50 Å / Num. all: 37872 / Num. obs: 35751 / % possible obs: 94.4 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 3.2 % / Biso Wilson estimate: 23.73 Å2 / Rmerge(I) obs: 0.1 / Net I/σ(I): 15.4 |
| Reflection shell | Resolution: 1.99→2.06 Å / Redundancy: 2.7 % / Rmerge(I) obs: 0.331 / Mean I/σ(I) obs: 3 / Num. unique all: 3796 / % possible all: 80.3 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 2XEE, chain A Resolution: 2.002→30.139 Å / Occupancy max: 1 / Occupancy min: 1 / SU ML: 0.56 / Cross valid method: THROUGHOUT / σ(F): 0.65 / Phase error: 19.8 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.86 Å / VDW probe radii: 1.1 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 40.481 Å2 / ksol: 0.401 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 80.64 Å2 / Biso mean: 27.95 Å2 / Biso min: 9.19 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.002→30.139 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 13
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | S33: 0 Å ° / Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj

