[English] 日本語
 Yorodumi
Yorodumi- PDB-4dch: Insights into Glucokinase Activation Mechanism: Observation of Mu... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4dch | ||||||
|---|---|---|---|---|---|---|---|
| Title | Insights into Glucokinase Activation Mechanism: Observation of Multiple Distinct Protein Conformations | ||||||
 Components Components | Glucokinase | ||||||
 Keywords Keywords |  TRANSFERASE / GK Beta Cell / TRANSFERASE / GK Beta Cell /  small molecule / open conformation / small molecule / open conformation /  Kinase Kinase | ||||||
| Function / homology |  Function and homology information Function and homology informationDefective GCK causes maturity-onset diabetes of the young 2 (MODY2) /  mannokinase activity / glucose sensor activity / regulation of potassium ion transport / mannokinase activity / glucose sensor activity / regulation of potassium ion transport /  hexokinase / hexokinase /  fructokinase activity / carbohydrate phosphorylation / fructokinase activity / carbohydrate phosphorylation /  glucokinase activity / glucose catabolic process / glucose 6-phosphate metabolic process ...Defective GCK causes maturity-onset diabetes of the young 2 (MODY2) / glucokinase activity / glucose catabolic process / glucose 6-phosphate metabolic process ...Defective GCK causes maturity-onset diabetes of the young 2 (MODY2) /  mannokinase activity / glucose sensor activity / regulation of potassium ion transport / mannokinase activity / glucose sensor activity / regulation of potassium ion transport /  hexokinase / hexokinase /  fructokinase activity / carbohydrate phosphorylation / fructokinase activity / carbohydrate phosphorylation /  glucokinase activity / glucose catabolic process / glucose 6-phosphate metabolic process / NADP metabolic process / Regulation of Glucokinase by Glucokinase Regulatory Protein / Defective TPR may confer susceptibility towards thyroid papillary carcinoma (TPC) / glucokinase activity / glucose catabolic process / glucose 6-phosphate metabolic process / NADP metabolic process / Regulation of Glucokinase by Glucokinase Regulatory Protein / Defective TPR may confer susceptibility towards thyroid papillary carcinoma (TPC) /  D-glucose binding / cellular response to leptin stimulus / calcium ion import / canonical glycolysis / D-glucose binding / cellular response to leptin stimulus / calcium ion import / canonical glycolysis /  Glycolysis / regulation of glycolytic process / intracellular glucose homeostasis / Glycolysis / regulation of glycolytic process / intracellular glucose homeostasis /  Regulation of gene expression in beta cells / regulation of insulin secretion / positive regulation of glycogen biosynthetic process / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / negative regulation of gluconeogenesis / response to glucose / glycolytic process / positive regulation of insulin secretion / cellular response to insulin stimulus / glucose metabolic process / Regulation of gene expression in beta cells / regulation of insulin secretion / positive regulation of glycogen biosynthetic process / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / negative regulation of gluconeogenesis / response to glucose / glycolytic process / positive regulation of insulin secretion / cellular response to insulin stimulus / glucose metabolic process /  glucose homeostasis / glucose homeostasis /  mitochondrion / mitochondrion /  nucleoplasm / nucleoplasm /  ATP binding / ATP binding /  cytosol cytosolSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 1.79 Å molecular replacement / Resolution: 1.79 Å | ||||||
 Authors Authors | Greasley, S.E. / Hickey, M. / Feng, J. / Garcia, E. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2012 Journal: J.Biol.Chem. / Year: 2012Title: Insights into Mechanism of Glucokinase Activation: OBSERVATION OF MULTIPLE DISTINCT PROTEIN CONFORMATIONS. Authors: Liu, S. / Ammirati, M.J. / Song, X. / Knafels, J.D. / Zhang, J. / Greasley, S.E. / Pfefferkorn, J.A. / Qiu, X. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4dch.cif.gz 4dch.cif.gz | 105.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4dch.ent.gz pdb4dch.ent.gz | 79.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4dch.json.gz 4dch.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dc/4dch https://data.pdbj.org/pub/pdb/validation_reports/dc/4dch ftp://data.pdbj.org/pub/pdb/validation_reports/dc/4dch ftp://data.pdbj.org/pub/pdb/validation_reports/dc/4dch | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein |  / Hexokinase type IV / HK IV / Hexokinase-4 / HK4 / Hexokinase-D / Hexokinase type IV / HK IV / Hexokinase-4 / HK4 / Hexokinase-DMass: 53329.562 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: GCK / Production host: Homo sapiens (human) / Gene: GCK / Production host:   Escherichia coli (E. coli) / References: UniProt: P35557, Escherichia coli (E. coli) / References: UniProt: P35557,  glucokinase glucokinase | ||
|---|---|---|---|
| #2: Sugar | ChemComp-GLC /  Glucose Glucose | ||
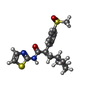
| #3: Chemical | ChemComp-4DC / ( | ||
| #4: Chemical |  Iodide Iodide#5: Water | ChemComp-HOH / |  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.84 Å3/Da / Density % sol: 56.64 % |
|---|---|
Crystal grow | Temperature: 286 K / Method: vapor diffusion, hanging drop / pH: 5.4 Details: 20% PEG 3350, 0.1A citrate pH 5.4, 0.2M ammonium iodide, 50mM glucose, VAPOR DIFFUSION, HANGING DROP, temperature 286K |
-Data collection
| Diffraction | Mean temperature: 93 K | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALS ALS  / Beamline: 5.0.1 / Wavelength: 1 Å / Beamline: 5.0.1 / Wavelength: 1 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: ADSC QUANTUM 315 / Detector: CCD / Date: Jul 29, 2005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Monochromator: Si(220) Asymmetric cut single crystal / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength : 1 Å / Relative weight: 1 : 1 Å / Relative weight: 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 1.8→50 Å / Num. all: 54275 / Num. obs: 54275 / % possible obs: 97.2 % / Observed criterion σ(F): 1 / Observed criterion σ(I): 1 / Redundancy: 3.4 % / Rmerge(I) obs: 0.068 / Χ2: 0.978 / Net I/σ(I): 11.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell |
|
-Phasing
Phasing | Method:  molecular replacement molecular replacement | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phasing MR | Rfactor: 0.516 / Cor.coef. Fo:Fc: 0.32
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 1.79→50 Å / Cor.coef. Fo:Fc: 0.954 / Cor.coef. Fo:Fc free: 0.937 / WRfactor Rfree: 0.2365 / WRfactor Rwork: 0.2058 / Occupancy max: 1 / Occupancy min: 0.5 / FOM work R set: 0.8509 / SU B: 2.417 / SU ML: 0.077 / SU R Cruickshank DPI: 0.1169 / SU Rfree: 0.1149 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.117 / ESU R Free: 0.115 / Stereochemistry target values: MAXIMUM LIKELIHOOD MOLECULAR REPLACEMENT / Resolution: 1.79→50 Å / Cor.coef. Fo:Fc: 0.954 / Cor.coef. Fo:Fc free: 0.937 / WRfactor Rfree: 0.2365 / WRfactor Rwork: 0.2058 / Occupancy max: 1 / Occupancy min: 0.5 / FOM work R set: 0.8509 / SU B: 2.417 / SU ML: 0.077 / SU R Cruickshank DPI: 0.1169 / SU Rfree: 0.1149 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.117 / ESU R Free: 0.115 / Stereochemistry target values: MAXIMUM LIKELIHOOD
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 58.97 Å2 / Biso mean: 29.7263 Å2 / Biso min: 8.72 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.79→50 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.792→1.839 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller
















 PDBj
PDBj