[English] 日本語
 Yorodumi
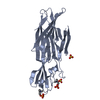
Yorodumi- PDB-3qlm: Crystal structure of porcine pancreatic phospholipase A2 in compl... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3qlm | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of porcine pancreatic phospholipase A2 in complex with n-hexadecanoic acid | ||||||
 Components Components | Phospholipase A2, major isoenzyme | ||||||
 Keywords Keywords |  HYDROLASE / HYDROLASE /  Pla2 / Pla2 /  Pancreatic enzyme / Pancreatic enzyme /  palmitic acid binding palmitic acid binding | ||||||
| Function / homology |  Function and homology information Function and homology informationAcyl chain remodelling of PC / Acyl chain remodelling of PS / Acyl chain remodelling of PE / Acyl chain remodelling of PI / Acyl chain remodelling of PG / Synthesis of PA / positive regulation of podocyte apoptotic process / regulation of glucose import / phosphatidylglycerol metabolic process / calcium-dependent phospholipase A2 activity ...Acyl chain remodelling of PC / Acyl chain remodelling of PS / Acyl chain remodelling of PE / Acyl chain remodelling of PI / Acyl chain remodelling of PG / Synthesis of PA / positive regulation of podocyte apoptotic process / regulation of glucose import / phosphatidylglycerol metabolic process / calcium-dependent phospholipase A2 activity / phosphatidylcholine metabolic process / leukotriene biosynthetic process / neutrophil mediated immunity /  phospholipase A2 / phospholipase A2 /  bile acid binding / bile acid binding /  phospholipase A2 activity / positive regulation of calcium ion transport into cytosol / phospholipid metabolic process / lipid catabolic process / phospholipase A2 activity / positive regulation of calcium ion transport into cytosol / phospholipid metabolic process / lipid catabolic process /  neutrophil chemotaxis / positive regulation of interleukin-8 production / neutrophil chemotaxis / positive regulation of interleukin-8 production /  phospholipid binding / positive regulation of MAP kinase activity / cellular response to insulin stimulus / fatty acid biosynthetic process / positive regulation of fibroblast proliferation / positive regulation of immune response / positive regulation of NF-kappaB transcription factor activity / intracellular signal transduction / phospholipid binding / positive regulation of MAP kinase activity / cellular response to insulin stimulus / fatty acid biosynthetic process / positive regulation of fibroblast proliferation / positive regulation of immune response / positive regulation of NF-kappaB transcription factor activity / intracellular signal transduction /  signaling receptor binding / signaling receptor binding /  calcium ion binding / positive regulation of cell population proliferation / calcium ion binding / positive regulation of cell population proliferation /  cell surface / positive regulation of transcription by RNA polymerase II / extracellular region cell surface / positive regulation of transcription by RNA polymerase II / extracellular regionSimilarity search - Function | ||||||
| Biological species |   Sus scrofa (pig) Sus scrofa (pig) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.5 Å MOLECULAR REPLACEMENT / Resolution: 2.5 Å | ||||||
 Authors Authors | Aparna, V. / Dileep, K.V. / Karthe, P. / Mandal, P.K. / Sadasivan, C. / Haridas, M. | ||||||
 Citation Citation |  Journal: Chem.Biol.Drug Des. / Year: 2012 Journal: Chem.Biol.Drug Des. / Year: 2012Title: Anti-inflammatory property of n-hexadecanoic acid: Structural evidence and Kinetic assessment. Authors: Aparna, V. / Dileep, K.V. / Mandal, P.K. / Karthe, P. / Sadasivan, C. / Haridas, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3qlm.cif.gz 3qlm.cif.gz | 40.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3qlm.ent.gz pdb3qlm.ent.gz | 27.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3qlm.json.gz 3qlm.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ql/3qlm https://data.pdbj.org/pub/pdb/validation_reports/ql/3qlm ftp://data.pdbj.org/pub/pdb/validation_reports/ql/3qlm ftp://data.pdbj.org/pub/pdb/validation_reports/ql/3qlm | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3o4mS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein |  / Group IB phospholipase A2 / Phosphatidylcholine 2-acylhydrolase 1B / Group IB phospholipase A2 / Phosphatidylcholine 2-acylhydrolase 1BMass: 14009.714 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Sus scrofa (pig) / Tissue: Pancreas Sus scrofa (pig) / Tissue: Pancreas / References: UniProt: P00592, / References: UniProt: P00592,  phospholipase A2 phospholipase A2 | ||||
|---|---|---|---|---|---|
| #2: Chemical | | #3: Chemical | ChemComp-PLM / |  Palmitic acid Palmitic acid#4: Water | ChemComp-HOH / |  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.44 Å3/Da / Density % sol: 64.27 % |
|---|---|
Crystal grow | Temperature: 294 K / Method: vapor diffusion, hanging drop / pH: 7.2 Details: 15% MPD, 0.05M Tris Maliate, 5mM Calcium Chloride, pH 7.2, VAPOR DIFFUSION, HANGING DROP, temperature 294K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: BRUKER AXS MICROSTAR / Wavelength: 1.542 Å ROTATING ANODE / Type: BRUKER AXS MICROSTAR / Wavelength: 1.542 Å |
| Detector | Type: MAR scanner 345 mm plate / Detector: IMAGE PLATE / Date: Feb 24, 2010 / Details: Mirrors |
| Radiation | Monochromator: Mirrors / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.542 Å / Relative weight: 1 : 1.542 Å / Relative weight: 1 |
| Reflection | Resolution: 2.5→45.44 Å / Num. all: 6866 / Num. obs: 6010 / % possible obs: 90.4 % / Observed criterion σ(F): 2.6 / Observed criterion σ(I): 2 / Rmerge(I) obs: 0.079 / Rsym value: 0.084 |
| Reflection shell | Resolution: 2.6→2.74 Å / % possible all: 90.4 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 3O4M Resolution: 2.5→15 Å / Cor.coef. Fo:Fc: 0.947 / Cor.coef. Fo:Fc free: 0.924 / SU B: 9.089 / SU ML: 0.197 / Cross valid method: THROUGHOUT / ESU R Free: 0.273 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 40.616 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.5→15 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.502→2.566 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller


















 PDBj
PDBj











