[English] 日本語
 Yorodumi
Yorodumi- PDB-3jql: Crystal Structure of the Complex Formed Between Phospholipase A2 ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3jql | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of the Complex Formed Between Phospholipase A2 and a Hexapeptide Fragment of Amyloid Beta Peptide, Lys-Leu-Val-Phe-Phe-Ala at 1.2 A Resolution | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  HYDROLASE / HYDROLASE /  PHOSPHOLIPASE A2 / PHOSPHOLIPASE A2 /  HEXAPEPTIDE / HEXAPEPTIDE /  AMYLOID BETA / AMYLOID BETA /  Disulfide bond / Lipid degradation / Metal-binding / Disulfide bond / Lipid degradation / Metal-binding /  Secreted Secreted | ||||||
| Function / homology |  Function and homology information Function and homology information phospholipase A2 / phospholipase A2 /  phospholipase A2 activity / arachidonic acid secretion / phospholipid metabolic process / lipid catabolic process / phospholipase A2 activity / arachidonic acid secretion / phospholipid metabolic process / lipid catabolic process /  calcium ion binding / extracellular region calcium ion binding / extracellular regionSimilarity search - Function | ||||||
| Biological species |   Naja sagittifera (cobra) Naja sagittifera (cobra) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.2 Å MOLECULAR REPLACEMENT / Resolution: 1.2 Å | ||||||
 Authors Authors | Mirza, Z. / Vikram, G. / Singh, N. / Sinha, M. / Sharma, S. / Srinivasan, A. / Kaur, P. / Singh, T.P. | ||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: Crystal Structure of the Complex Formed Between Phospholipase A2 and a Hexapeptide Fragment of Amyloid Beta Peptide, Lys-Leu-Val-Phe-Phe-Ala at 1.2 A Resolution Authors: Mirza, Z. / Vikram, G. / Singh, N. / Sinha, M. / Sharma, S. / Srinivasan, A. / Kaur, P. / Singh, T.P. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3jql.cif.gz 3jql.cif.gz | 66.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3jql.ent.gz pdb3jql.ent.gz | 48 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3jql.json.gz 3jql.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/jq/3jql https://data.pdbj.org/pub/pdb/validation_reports/jq/3jql ftp://data.pdbj.org/pub/pdb/validation_reports/jq/3jql ftp://data.pdbj.org/pub/pdb/validation_reports/jq/3jql | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1mf4S S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 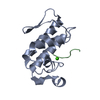
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 13128.515 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Naja sagittifera (cobra) / References: UniProt: P60045 Naja sagittifera (cobra) / References: UniProt: P60045 |
|---|---|
| #2: Protein/peptide |  Amyloid beta Amyloid betaMass: 724.909 Da / Num. of mol.: 1 / Source method: obtained synthetically / Details: Peptide synthesis |
| #3: Chemical | ChemComp-CA / |
| #4: Water | ChemComp-HOH /  Water Water |
| Sequence details | IN CHAIN A, RESIDUE NUMBER 16 IS SIMPLY SKIPPED. THE AUTHORS BELIEVE THAT THE SEQRES IS CORRECT AND ...IN CHAIN A, RESIDUE NUMBER 16 IS SIMPLY SKIPPED. THE AUTHORS BELIEVE THAT THE SEQRES IS CORRECT AND THESE MUTATIONS ARE PRESENT IN THE DEPOSITED SEQUENCE. |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.04 Å3/Da / Density % sol: 39.83 % |
|---|---|
Crystal grow | Temperature: 290 K / Method: vapor diffusion, hanging drop / pH: 6 Details: calcium chloride, sodium phosphate, PH6.0, VAPOR DIFFUSION, HANGING DROP, temperature 290K |
-Data collection
| Diffraction | Mean temperature: 300 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RU300 / Wavelength: 1.5418 Å ROTATING ANODE / Type: RIGAKU RU300 / Wavelength: 1.5418 Å |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Mar 30, 2009 / Details: MIRROR |
| Radiation | Monochromator: GRAPHITE / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.5418 Å / Relative weight: 1 : 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.2→42 Å / Num. all: 34924 / Num. obs: 34924 / % possible obs: 95.9 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Rsym value: 0.036 / Net I/σ(I): 7.9 |
| Reflection shell | Resolution: 1.2→1.22 Å / Mean I/σ(I) obs: 2.5 / Rsym value: 0.122 / % possible all: 94.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1MF4 Resolution: 1.2→42 Å / Cor.coef. Fo:Fc: 0.962 / Cor.coef. Fo:Fc free: 0.961 / SU B: 1.294 / SU ML: 0.027 / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 0 / ESU R: 0.052 / ESU R Free: 0.047 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 23.326 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.2→42 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.199→1.23 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller












 PDBj
PDBj




