[English] 日本語
 Yorodumi
Yorodumi- PDB-2wv8: Complex of human dihydroorotate dehydrogenase with the inhibitor ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2wv8 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Complex of human dihydroorotate dehydrogenase with the inhibitor 221290 | ||||||
 Components Components | DIHYDROOROTATE DEHYDROGENASE, MITOCHONDRIAL | ||||||
 Keywords Keywords |  OXIDOREDUCTASE / OXIDOREDUCTASE /  FLAVOPROTEIN / FLAVOPROTEIN /  TRANSMEMBRANE / MITOCHONDRION INNER MEMBRANE / TRANSIT PEPTIDE / TRANSMEMBRANE / MITOCHONDRION INNER MEMBRANE / TRANSIT PEPTIDE /  ENZYME INHIBITION / ENZYME INHIBITION /  PYRIMIDINE BIOSYNTHESIS / PYRIMIDINE BIOSYNTHESIS /  STRUCTURE-BASED DRUG DESIGN / INFLAMATION STRUCTURE-BASED DRUG DESIGN / INFLAMATION | ||||||
| Function / homology |  Function and homology information Function and homology informationpyrimidine ribonucleotide biosynthetic process /  dihydroorotate dehydrogenase (quinone) activity / dihydroorotate dehydrogenase (quinone) activity /  dihydroorotate dehydrogenase (quinone) / dihydroorotate dehydrogenase (quinone) /  dihydroorotate dehydrogenase activity / dihydroorotate dehydrogenase activity /  dihydroorotase activity / dihydroorotase activity /  Pyrimidine biosynthesis / UDP biosynthetic process / 'de novo' UMP biosynthetic process / 'de novo' pyrimidine nucleobase biosynthetic process / Pyrimidine biosynthesis / UDP biosynthetic process / 'de novo' UMP biosynthetic process / 'de novo' pyrimidine nucleobase biosynthetic process /  mitochondrial inner membrane ...pyrimidine ribonucleotide biosynthetic process / mitochondrial inner membrane ...pyrimidine ribonucleotide biosynthetic process /  dihydroorotate dehydrogenase (quinone) activity / dihydroorotate dehydrogenase (quinone) activity /  dihydroorotate dehydrogenase (quinone) / dihydroorotate dehydrogenase (quinone) /  dihydroorotate dehydrogenase activity / dihydroorotate dehydrogenase activity /  dihydroorotase activity / dihydroorotase activity /  Pyrimidine biosynthesis / UDP biosynthetic process / 'de novo' UMP biosynthetic process / 'de novo' pyrimidine nucleobase biosynthetic process / Pyrimidine biosynthesis / UDP biosynthetic process / 'de novo' UMP biosynthetic process / 'de novo' pyrimidine nucleobase biosynthetic process /  mitochondrial inner membrane / mitochondrial inner membrane /  mitochondrion / mitochondrion /  nucleoplasm / nucleoplasm /  cytosol cytosolSimilarity search - Function | ||||||
| Biological species |   HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.9 Å MOLECULAR REPLACEMENT / Resolution: 1.9 Å | ||||||
 Authors Authors | Walse, B. / Svensson, B. / Fritzson, I. / Dahlberg, L. / Wellmar, U. / Al-Karadaghi, S. | ||||||
 Citation Citation |  Journal: Chemmedchem / Year: 2010 Journal: Chemmedchem / Year: 2010Title: Inhibition of Human Dhodh by 4-Hydroxycoumarins, Fenamic Acids, and N-(Alkylcarbonyl)Anthranilic Acids Identified by Structure-Guided Fragment Selection. Authors: Fritzson, I. / Svensson, B. / Al-Karadaghi, S. / Walse, B. / Wellmar, U. / Nilsson, U.J. / Da Graca Thrige, D. / Jonsson, S. | ||||||
| History |
| ||||||
| Remark 700 | SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AB" IN EACH CHAIN ON SHEET RECORDS BELOW ... SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AB" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 8-STRANDED BARREL THIS IS REPRESENTED BY A 9-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2wv8.cif.gz 2wv8.cif.gz | 95.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2wv8.ent.gz pdb2wv8.ent.gz | 71 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2wv8.json.gz 2wv8.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/wv/2wv8 https://data.pdbj.org/pub/pdb/validation_reports/wv/2wv8 ftp://data.pdbj.org/pub/pdb/validation_reports/wv/2wv8 ftp://data.pdbj.org/pub/pdb/validation_reports/wv/2wv8 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein |  / DHODEHASE / DIHYDROOROTATE OXIDASE / DHODEHASE / DHODEHASE / DIHYDROOROTATE OXIDASE / DHODEHASEMass: 39654.266 Da / Num. of mol.: 1 / Fragment: RESIDUES 31-395 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   HOMO SAPIENS (human) / Production host: HOMO SAPIENS (human) / Production host:   ESCHERICHIA COLI (E. coli) / Strain (production host): BL21 ESCHERICHIA COLI (E. coli) / Strain (production host): BL21References: UniProt: Q02127,  dihydroorotate dehydrogenase (quinone) dihydroorotate dehydrogenase (quinone) |
|---|
-Non-polymers , 7 types, 354 molecules 
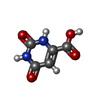










| #2: Chemical | ChemComp-FMN /  Flavin mononucleotide Flavin mononucleotide |
|---|---|
| #3: Chemical | ChemComp-ORO /  Orotic acid Orotic acid |
| #4: Chemical | ChemComp-VGN / |
| #5: Chemical | ChemComp-DDQ / |
| #6: Chemical | ChemComp-ACT /  Acetate Acetate |
| #7: Chemical | ChemComp-SO4 /  Sulfate Sulfate |
| #8: Water | ChemComp-HOH /  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.23 Å3/Da / Density % sol: 61.57 % / Description: NONE |
|---|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  MAX II MAX II  / Type: / Type:  MAX II MAX II  / Wavelength: 1.017 / Wavelength: 1.017 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.017 Å / Relative weight: 1 : 1.017 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→20 Å / Num. obs: 44920 / % possible obs: 96.4 % / Observed criterion σ(I): 2 / Redundancy: 4.6 % / Biso Wilson estimate: 12 Å2 / Rmerge(I) obs: 0.06 / Net I/σ(I): 17 |
| Reflection shell | Resolution: 1.9→2 Å / Redundancy: 4.5 % / Rmerge(I) obs: 0.26 / Mean I/σ(I) obs: 6 / % possible all: 97.1 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 1.9→19.38 Å / Rfactor Rfree error: 0.004 / Data cutoff high absF: 2791550.09 / Data cutoff low absF: 0 / Cross valid method: THROUGHOUT / σ(F): 0 MOLECULAR REPLACEMENT / Resolution: 1.9→19.38 Å / Rfactor Rfree error: 0.004 / Data cutoff high absF: 2791550.09 / Data cutoff low absF: 0 / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 57.7489 Å2 / ksol: 0.401457 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 20 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.9→19.38 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.9→2.02 Å / Rfactor Rfree error: 0.012 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
|
 Movie
Movie Controller
Controller


















 PDBj
PDBj






