[English] 日本語
 Yorodumi
Yorodumi- PDB-2w3a: HUMAN DIHYDROFOLATE REDUCTASE COMPLEXED WITH NADPH AND TRIMETHOPRIM -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2w3a | ||||||
|---|---|---|---|---|---|---|---|
| Title | HUMAN DIHYDROFOLATE REDUCTASE COMPLEXED WITH NADPH AND TRIMETHOPRIM | ||||||
 Components Components | DIHYDROFOLATE REDUCTASE | ||||||
 Keywords Keywords |  OXIDOREDUCTASE / NONCLASSICAL ANTIFOLATES / ONE-CARBON METABOLISM / LIPOPHILIC ANTIFOLATES / OXIDOREDUCTASE / NONCLASSICAL ANTIFOLATES / ONE-CARBON METABOLISM / LIPOPHILIC ANTIFOLATES /  NADP / NADP /  REDUCTASE REDUCTASE | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of removal of superoxide radicals / tetrahydrobiopterin biosynthetic process / Metabolism of folate and pterines / tetrahydrofolate metabolic process / sequence-specific mRNA binding / response to methotrexate / axon regeneration /  folic acid binding / dihydrofolate metabolic process / G1/S-Specific Transcription ...regulation of removal of superoxide radicals / tetrahydrobiopterin biosynthetic process / Metabolism of folate and pterines / tetrahydrofolate metabolic process / sequence-specific mRNA binding / response to methotrexate / axon regeneration / folic acid binding / dihydrofolate metabolic process / G1/S-Specific Transcription ...regulation of removal of superoxide radicals / tetrahydrobiopterin biosynthetic process / Metabolism of folate and pterines / tetrahydrofolate metabolic process / sequence-specific mRNA binding / response to methotrexate / axon regeneration /  folic acid binding / dihydrofolate metabolic process / G1/S-Specific Transcription / glycine biosynthetic process / folic acid binding / dihydrofolate metabolic process / G1/S-Specific Transcription / glycine biosynthetic process /  dihydrofolate reductase / folic acid metabolic process / dihydrofolate reductase / folic acid metabolic process /  dihydrofolate reductase activity / dihydrofolate reductase activity /  NADPH binding / Tetrahydrobiopterin (BH4) synthesis, recycling, salvage and regulation / tetrahydrofolate biosynthetic process / mRNA regulatory element binding translation repressor activity / positive regulation of nitric-oxide synthase activity / one-carbon metabolic process / NADPH binding / Tetrahydrobiopterin (BH4) synthesis, recycling, salvage and regulation / tetrahydrofolate biosynthetic process / mRNA regulatory element binding translation repressor activity / positive regulation of nitric-oxide synthase activity / one-carbon metabolic process /  NADP binding / negative regulation of translation / NADP binding / negative regulation of translation /  mRNA binding / mRNA binding /  mitochondrion / mitochondrion /  cytosol cytosolSimilarity search - Function | ||||||
| Biological species |   HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.5 Å MOLECULAR REPLACEMENT / Resolution: 1.5 Å | ||||||
 Authors Authors | Leung, A.K.W. / Reynolds, R.C. / Borhani, D.W. | ||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: Structural Basis for Selective Inhibition of Mycobacterium Avium Dihydrofolate Reductase by a Lipophilic Antifolate Authors: Leung, A.K.W. / Ross, L.J. / Zywno-Van Ginkel, S. / Reynolds, R.C. / Seitz, L.E. / Pathak, V. / Barrow, W.W. / White, E.L. / Suling, W.J. / Piper, J.R. / Borhani, D.W. | ||||||
| History |
| ||||||
| Remark 700 | SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN ... SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN THE SHEET RECORDS BELOW, TWO SHEETS ARE DEFINED. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2w3a.cif.gz 2w3a.cif.gz | 185.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2w3a.ent.gz pdb2w3a.ent.gz | 149.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2w3a.json.gz 2w3a.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/w3/2w3a https://data.pdbj.org/pub/pdb/validation_reports/w3/2w3a ftp://data.pdbj.org/pub/pdb/validation_reports/w3/2w3a ftp://data.pdbj.org/pub/pdb/validation_reports/w3/2w3a | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Noncrystallographic symmetry (NCS) | NCS oper: (Code: given Matrix: (-0.061704, 0.997265, -0.040674), Vector  : : |
- Components
Components
-Protein , 1 types, 2 molecules AB
| #1: Protein |  Mass: 21480.723 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   HOMO SAPIENS (human) / Plasmid: PDFR / Production host: HOMO SAPIENS (human) / Plasmid: PDFR / Production host:   ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: P00374, ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: P00374,  dihydrofolate reductase dihydrofolate reductase |
|---|
-Non-polymers , 5 types, 415 molecules 


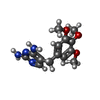





| #2: Chemical |  Glycerol Glycerol#3: Chemical |  Nicotinamide adenine dinucleotide phosphate Nicotinamide adenine dinucleotide phosphate#4: Chemical | ChemComp-SO4 / |  Sulfate Sulfate#5: Chemical |  Trimethoprim Trimethoprim#6: Water | ChemComp-HOH / |  Water Water |
|---|
-Details
| Nonpolymer details | GLYCEROL (GOL): 1,2,3-PROPANETRI |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.34 Å3/Da / Density % sol: 47.47 % / Description: NONE |
|---|---|
Crystal grow | Temperature: 277 K / Method: vapor diffusion, hanging drop / pH: 8.75 Details: HUMAN DHFR/FOLATE COMPLEX WAS MIXED WITH NADPH AND TRIMETHOPRIM (BOTH 2 MM FINAL). CRYSTALS WERE GROWN BY HANGING DROP VAPOR DIFFUSION AT 277 K BY MIXING EQUAL VOLUMES OF PROTEIN/NADPH/TMP ...Details: HUMAN DHFR/FOLATE COMPLEX WAS MIXED WITH NADPH AND TRIMETHOPRIM (BOTH 2 MM FINAL). CRYSTALS WERE GROWN BY HANGING DROP VAPOR DIFFUSION AT 277 K BY MIXING EQUAL VOLUMES OF PROTEIN/NADPH/TMP WITH RESERVOIR (24% PEG 4000, 200 MM LI2SO4, 100 MM TRIS.HCL, PH 8.75). TRUNCATED TRIANGULAR CRYSTALS APPEARED SLOWLY, IN ABOUT A MONTH. THE CRYSTAL WAS CRYOPROTECTED WITH 15% GLYCEROL AND FLASH-COOLED IN LIQUID N2. |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  CHESS CHESS  / Beamline: A1 / Wavelength: 0.908 / Beamline: A1 / Wavelength: 0.908 |
| Detector | Type: ADSC QUANTUM-1 / Detector: CCD / Date: Jul 14, 1997 / Details: MIRRORS |
| Radiation | Monochromator: SI / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.908 Å / Relative weight: 1 : 0.908 Å / Relative weight: 1 |
| Reflection | Resolution: 1.5→20 Å / Num. obs: 55140 / % possible obs: 85.7 % / Observed criterion σ(I): -10 / Redundancy: 3.3 % / Biso Wilson estimate: 16 Å2 / Rmerge(I) obs: 0.05 / Net I/σ(I): 8.3 |
| Reflection shell | Resolution: 1.5→1.54 Å / Redundancy: 1.1 % / Rmerge(I) obs: 0.19 / Mean I/σ(I) obs: 2.9 / % possible all: 28.3 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: UNPUBLISHED HUMAN DHFR FOLATE COMPLEX Resolution: 1.5→9.9 Å / Cor.coef. Fo:Fc: 0.945 / Cor.coef. Fo:Fc free: 0.931 / SU B: 2.788 / SU ML: 0.049 / Cross valid method: THROUGHOUT / ESU R: 0.125 / ESU R Free: 0.091 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: BABINET MODEL WITH MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 16.75 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.5→9.9 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller







































 PDBj
PDBj









