+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2kr4 | ||||||
|---|---|---|---|---|---|---|---|
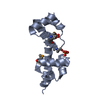
| Title | U-box domain of the E3 Ubiquitin Ligase E4B | ||||||
 Components Components | Ubiquitin conjugation factor E4 B | ||||||
 Keywords Keywords |  LIGASE / U-box / E4B / Ufd2 / RING / LIGASE / U-box / E4B / Ufd2 / RING /  Ubiquitin / Ubiquitin /  E3 Ligase / Ubl conjugation pathway E3 Ligase / Ubl conjugation pathway | ||||||
| Function / homology |  Function and homology information Function and homology informationgranzyme-mediated apoptotic signaling pathway / protein catabolic process => GO:0030163 / ventricular trabecula myocardium morphogenesis / ubiquitin-ubiquitin ligase activity / protein monoubiquitination / protein autoubiquitination /  ubiquitin ligase complex / ubiquitin ligase complex /  : / response to UV / response to endoplasmic reticulum stress ...granzyme-mediated apoptotic signaling pathway / protein catabolic process => GO:0030163 / ventricular trabecula myocardium morphogenesis / ubiquitin-ubiquitin ligase activity / protein monoubiquitination / protein autoubiquitination / : / response to UV / response to endoplasmic reticulum stress ...granzyme-mediated apoptotic signaling pathway / protein catabolic process => GO:0030163 / ventricular trabecula myocardium morphogenesis / ubiquitin-ubiquitin ligase activity / protein monoubiquitination / protein autoubiquitination /  ubiquitin ligase complex / ubiquitin ligase complex /  : / response to UV / response to endoplasmic reticulum stress / RING-type E3 ubiquitin transferase / protein polyubiquitination / neuron projection development / : / response to UV / response to endoplasmic reticulum stress / RING-type E3 ubiquitin transferase / protein polyubiquitination / neuron projection development /  ubiquitin protein ligase activity / unfolded protein binding / ubiquitin protein ligase activity / unfolded protein binding /  protein folding / protein folding /  ATPase binding / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process / protein ubiquitination / ATPase binding / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process / protein ubiquitination /  enzyme binding / enzyme binding /  ATP binding / ATP binding /  nucleus / nucleus /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Mus musculus (house mouse) Mus musculus (house mouse) | ||||||
| Method |  SOLUTION NMR / SOLUTION NMR /  simulated annealing, simulated annealing,  molecular dynamics molecular dynamics | ||||||
| Model details | closest to the average, model 1 | ||||||
 Authors Authors | Nordquist, K. / Soss, S. / Chazin, W. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 2010 Journal: Biochemistry / Year: 2010Title: Structural and functional characterization of the monomeric U-box domain from E4B. Authors: Nordquist, K.A. / Dimitrova, Y.N. / Brzovic, P.S. / Ridenour, W.B. / Munro, K.A. / Soss, S.E. / Caprioli, R.M. / Klevit, R.E. / Chazin, W.J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2kr4.cif.gz 2kr4.cif.gz | 519.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2kr4.ent.gz pdb2kr4.ent.gz | 438.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2kr4.json.gz 2kr4.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/kr/2kr4 https://data.pdbj.org/pub/pdb/validation_reports/kr/2kr4 ftp://data.pdbj.org/pub/pdb/validation_reports/kr/2kr4 ftp://data.pdbj.org/pub/pdb/validation_reports/kr/2kr4 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 9778.996 Da / Num. of mol.: 1 / Fragment: U-Box Domain (UNP residues 1092-1173) Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Mus musculus (house mouse) / Description: 6xHIS-SUMO tag with H3C protease cleavage site / Gene: Ube4b, Ufd2 / Production host: Mus musculus (house mouse) / Description: 6xHIS-SUMO tag with H3C protease cleavage site / Gene: Ube4b, Ufd2 / Production host:   Escherichia coli (E. coli) / References: UniProt: Q9ES00 Escherichia coli (E. coli) / References: UniProt: Q9ES00 |
|---|
-Experimental details
-Experiment
| Experiment | Method:  SOLUTION NMR SOLUTION NMR | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample |
| ||||||||||||
| Sample conditions | Ionic strength: 25 / pH: 6.5 / Pressure: ambient atm / Temperature: 298 K |
-NMR measurement
| NMR spectrometer |
|
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method:  simulated annealing, simulated annealing,  molecular dynamics / Software ordinal: 1 molecular dynamics / Software ordinal: 1 Details: Computed through AMBER9 in water, Computed through AMBER9 in water. Single residues in some models have phi-psi angle combinations that are outside the norm. These are found only for ...Details: Computed through AMBER9 in water, Computed through AMBER9 in water. Single residues in some models have phi-psi angle combinations that are outside the norm. These are found only for residues that lack experimental constraints and which are therefore poorly defined in the structure. | ||||||||||||||||||||||||
| NMR representative | Selection criteria: closest to the average | ||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the lowest energy Conformers calculated total number: 100 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller












 PDBj
PDBj

