[English] 日本語
 Yorodumi
Yorodumi- PDB-2kiq: Solution structure of the FF Domain 2 of human transcription elon... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2kiq | ||||||
|---|---|---|---|---|---|---|---|
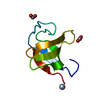
| Title | Solution structure of the FF Domain 2 of human transcription elongation factor CA150 | ||||||
 Components Components | Transcription elongation regulator 1 | ||||||
 Keywords Keywords |  TRANSCRIPTION REGULATOR / Human transcription elongation factor CA150 / RNA polymerase II C-terminal domain interacting protein / FF domain / TRANSCRIPTION REGULATOR / Human transcription elongation factor CA150 / RNA polymerase II C-terminal domain interacting protein / FF domain /  Residual dipolar coupling / Activator / Residual dipolar coupling / Activator /  Alternative splicing / Alternative splicing /  Coiled coil / Coiled coil /  Nucleus / Nucleus /  Phosphoprotein / Phosphoprotein /  Repressor / Repressor /  Transcription / Transcription /  Transcription regulation Transcription regulation | ||||||
| Function / homology |  Function and homology information Function and homology informationtranscription elongation factor activity / ubiquitin-like protein conjugating enzyme binding /  RNA polymerase binding / negative regulation of transcription elongation by RNA polymerase II / mRNA Splicing - Major Pathway / RNA polymerase binding / negative regulation of transcription elongation by RNA polymerase II / mRNA Splicing - Major Pathway /  RNA splicing / positive regulation of transcription elongation by RNA polymerase II / RNA splicing / positive regulation of transcription elongation by RNA polymerase II /  transcription coregulator activity / transcription coregulator activity /  mRNA processing / transcription corepressor activity ...transcription elongation factor activity / ubiquitin-like protein conjugating enzyme binding / mRNA processing / transcription corepressor activity ...transcription elongation factor activity / ubiquitin-like protein conjugating enzyme binding /  RNA polymerase binding / negative regulation of transcription elongation by RNA polymerase II / mRNA Splicing - Major Pathway / RNA polymerase binding / negative regulation of transcription elongation by RNA polymerase II / mRNA Splicing - Major Pathway /  RNA splicing / positive regulation of transcription elongation by RNA polymerase II / RNA splicing / positive regulation of transcription elongation by RNA polymerase II /  transcription coregulator activity / transcription coregulator activity /  mRNA processing / transcription corepressor activity / RNA polymerase II-specific DNA-binding transcription factor binding / mRNA processing / transcription corepressor activity / RNA polymerase II-specific DNA-binding transcription factor binding /  transcription coactivator activity / nuclear speck / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II / transcription coactivator activity / nuclear speck / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II /  RNA binding / RNA binding /  nucleoplasm / identical protein binding / nucleoplasm / identical protein binding /  nucleus nucleusSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  SOLUTION NMR / GLOBAL FOLD COMPUTATION BASED ON EXACT SOLUTIONS FROM RDCS, NOE ASSIGNMENT BASED ON HAUSDORFF-BASED PATTERN MATCHING, ENERGY-MINIMIZATION SOLUTION NMR / GLOBAL FOLD COMPUTATION BASED ON EXACT SOLUTIONS FROM RDCS, NOE ASSIGNMENT BASED ON HAUSDORFF-BASED PATTERN MATCHING, ENERGY-MINIMIZATION | ||||||
| Model details | lowest energy, model 1 | ||||||
 Authors Authors | Zeng, J. / Boyles, J. / Tripathy, C. / Yan, A. / Zhou, P. / Donald, B.R. | ||||||
 Citation Citation |  Journal: J.Biomol.Nmr / Year: 2009 Journal: J.Biomol.Nmr / Year: 2009Title: High-resolution protein structure determination starting with a global fold calculated from exact solutions to the RDC equations. Authors: Zeng, J. / Boyles, J. / Tripathy, C. / Wang, L. / Yan, A. / Zhou, P. / Donald, B.R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2kiq.cif.gz 2kiq.cif.gz | 362.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2kiq.ent.gz pdb2kiq.ent.gz | 304 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2kiq.json.gz 2kiq.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ki/2kiq https://data.pdbj.org/pub/pdb/validation_reports/ki/2kiq ftp://data.pdbj.org/pub/pdb/validation_reports/ki/2kiq ftp://data.pdbj.org/pub/pdb/validation_reports/ki/2kiq | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein |  / TATA box-binding protein-associated factor 2S / Transcription factor CA150 / TATA box-binding protein-associated factor 2S / Transcription factor CA150Mass: 7315.560 Da / Num. of mol.: 1 / Fragment: UNP residues 724-782 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: TCERG1, CA150, TAF2S / Plasmid: pET15b-modified / Production host: Homo sapiens (human) / Gene: TCERG1, CA150, TAF2S / Plasmid: pET15b-modified / Production host:   Escherichia coli (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: O14776 Escherichia coli (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: O14776 |
|---|
-Experimental details
-Experiment
| Experiment | Method:  SOLUTION NMR SOLUTION NMR | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample |
| ||||||||||||
| Sample conditions | Ionic strength: 25 / pH: 7.0 / Pressure: ambient / Temperature: 303 K |
-NMR measurement
| NMR spectrometer |
|
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: GLOBAL FOLD COMPUTATION BASED ON EXACT SOLUTIONS FROM RDCS, NOE ASSIGNMENT BASED ON HAUSDORFF-BASED PATTERN MATCHING, ENERGY-MINIMIZATION Software ordinal: 1 Details: 1. ORIENTATIONS AND CONFORMATIONS OF SECONDARY STRUCTURE ELEMENTS WERE CALCULATED USING THE RDC-EXACT MODULE, WHICH EXACTLY SOLVES A SYSTEM OF QUARTIC RDC EQUATIONS AND COMPUTES THE GLOBAL ...Details: 1. ORIENTATIONS AND CONFORMATIONS OF SECONDARY STRUCTURE ELEMENTS WERE CALCULATED USING THE RDC-EXACT MODULE, WHICH EXACTLY SOLVES A SYSTEM OF QUARTIC RDC EQUATIONS AND COMPUTES THE GLOBAL OPTIMAL SOLUTIONS OF BACKBONE DIHEDRAL ANGLES. 2. THE PACKER MODULE WAS USED TO DETERMINE THE TRANSLATIONS BETWEEN SECONDARY STRUCTURE ELEMENTS USING A SPARSE SET OF NOE DISTANCE RESTRAINTS. THE HANA MODULE WAS USED TO COMPUTE NOE ASSIGNMENTS BASED ON A HAUSDORFF-BASED PATTERN MATCHING TECHNIQUE. THE STRUCTURES WERE REFINED AGAINST RESIDUAL DIPOLAR COUPLINGS USING XPLOR-NIH AND A WATER-REFINEMENT PROTOCOL. | ||||||||||||||||||||||||||||
| NMR representative | Selection criteria: lowest energy | ||||||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the lowest energy and acceptable covalent geometry Conformers calculated total number: 50 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller










 PDBj
PDBj


