[English] 日本語
 Yorodumi
Yorodumi- PDB-1slx: RAT ANIONIC N143H, E151H TRYPSIN COMPLEXED TO A86H ECOTIN; ZINC-BOUND -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1slx | ||||||
|---|---|---|---|---|---|---|---|
| Title | RAT ANIONIC N143H, E151H TRYPSIN COMPLEXED TO A86H ECOTIN; ZINC-BOUND | ||||||
 Components Components |
| ||||||
 Keywords Keywords | COMPLEX (SERINE PROTEASE/INHIBITOR) /  SERINE PROTEASE / SERINE PROTEASE /  INHIBITOR / INHIBITOR /  COMPLEX / METAL BINDING SITES / COMPLEX / METAL BINDING SITES /  PROTEIN ENGINEERING / PROTEASE-SUBSTRATE INTERACTIONS / PROTEIN ENGINEERING / PROTEASE-SUBSTRATE INTERACTIONS /  METALLOPROTEINS / COMPLEX (SERINE PROTEASE-INHIBITOR) COMPLEX METALLOPROTEINS / COMPLEX (SERINE PROTEASE-INHIBITOR) COMPLEX | ||||||
| Function / homology |  Function and homology information Function and homology information Antimicrobial peptides / Alpha-defensins / Activation of Matrix Metalloproteinases / Collagen degradation / Neutrophil degranulation / collagen catabolic process / Antimicrobial peptides / Alpha-defensins / Activation of Matrix Metalloproteinases / Collagen degradation / Neutrophil degranulation / collagen catabolic process /  trypsin / trypsin /  digestion / response to nutrient / serine-type endopeptidase inhibitor activity ... digestion / response to nutrient / serine-type endopeptidase inhibitor activity ... Antimicrobial peptides / Alpha-defensins / Activation of Matrix Metalloproteinases / Collagen degradation / Neutrophil degranulation / collagen catabolic process / Antimicrobial peptides / Alpha-defensins / Activation of Matrix Metalloproteinases / Collagen degradation / Neutrophil degranulation / collagen catabolic process /  trypsin / trypsin /  digestion / response to nutrient / serine-type endopeptidase inhibitor activity / outer membrane-bounded periplasmic space / serine-type endopeptidase activity / digestion / response to nutrient / serine-type endopeptidase inhibitor activity / outer membrane-bounded periplasmic space / serine-type endopeptidase activity /  calcium ion binding / protein homodimerization activity / calcium ion binding / protein homodimerization activity /  proteolysis / proteolysis /  extracellular space / extracellular region extracellular space / extracellular regionSimilarity search - Function | ||||||
| Biological species |   Escherichia coli (E. coli) Escherichia coli (E. coli)  Rattus norvegicus (Norway rat) Rattus norvegicus (Norway rat) | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2.2 Å X-RAY DIFFRACTION / Resolution: 2.2 Å | ||||||
 Authors Authors | Brinen, L.S. / Fletterick, R.J. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1996 Journal: Biochemistry / Year: 1996Title: X-ray structures of a designed binding site in trypsin show metal-dependent geometry. Authors: Brinen, L.S. / Willett, W.S. / Craik, C.S. / Fletterick, R.J. #1:  Journal: Embo J. / Year: 1994 Journal: Embo J. / Year: 1994Title: Macromolecular Chelation as an Improved Mechanism of Protease Inhibition: Structure of the Ecotin-Trypsin Complex Authors: Mcgrath, M.E. / Erpel, T. / Bystroff, C. / Fletterick, R.J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1slx.cif.gz 1slx.cif.gz | 79.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1slx.ent.gz pdb1slx.ent.gz | 62.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1slx.json.gz 1slx.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sl/1slx https://data.pdbj.org/pub/pdb/validation_reports/sl/1slx ftp://data.pdbj.org/pub/pdb/validation_reports/sl/1slx ftp://data.pdbj.org/pub/pdb/validation_reports/sl/1slx | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 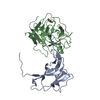
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
-Protein , 2 types, 2 molecules AB
| #1: Protein |  / TRYPSIN INHIBITOR / TRYPSIN INHIBITORMass: 16187.577 Da / Num. of mol.: 1 / Mutation: CHAIN A, A86H Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli (E. coli) / Gene: RAT ANIONIC TRYPSIN / Plasmid: PTACTAC / Gene (production host): ECOTIN / Production host: Escherichia coli (E. coli) / Gene: RAT ANIONIC TRYPSIN / Plasmid: PTACTAC / Gene (production host): ECOTIN / Production host:   Escherichia coli (E. coli) / References: UniProt: P23827 Escherichia coli (E. coli) / References: UniProt: P23827 |
|---|---|
| #2: Protein | Mass: 23847.914 Da / Num. of mol.: 1 / Mutation: CHAIN B, N143H, E151H Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Rattus norvegicus (Norway rat) / Gene: RAT ANIONIC TRYPSIN / Plasmid: PST / Gene (production host): RAT ANIONIC TRYPSIN / Production host: Rattus norvegicus (Norway rat) / Gene: RAT ANIONIC TRYPSIN / Plasmid: PST / Gene (production host): RAT ANIONIC TRYPSIN / Production host:   Saccharomyces cerevisiae (brewer's yeast) / References: UniProt: P00763, Saccharomyces cerevisiae (brewer's yeast) / References: UniProt: P00763,  trypsin trypsin |
-Non-polymers , 4 types, 66 molecules 






| #3: Chemical | ChemComp-CA / |
|---|---|
| #4: Chemical | ChemComp-ACT /  Acetate Acetate |
| #5: Chemical | ChemComp-ZN / |
| #6: Water | ChemComp-HOH /  Water Water |
-Details
| Compound details | SITE DIRECTED MUTAGENESIS WAS PERFORMED IN ORDER TO DESIGN A METAL BINDING SITE AT THE INTERFACE OF ...SITE DIRECTED MUTAGENESI |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.47 Å3/Da / Density % sol: 50 % | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Crystal grow | *PLUS Temperature: 18 ℃ / pH: 8 / Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 298 K |
|---|---|
| Diffraction source | Wavelength: 1.5418 |
| Detector | Type: RIGAKU / Detector: IMAGE PLATE / Date: Aug 8, 1994 |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.5418 Å / Relative weight: 1 : 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.2→50 Å / Num. obs: 46301 / % possible obs: 90.9 % / Observed criterion σ(I): 1.5 / Redundancy: 3 % / Rmerge(I) obs: 0.065 |
| Reflection | *PLUS Num. obs: 10021 / Num. measured all: 46301 |
| Reflection shell | *PLUS % possible obs: 87.4 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 2.2→6 Å / σ(F): 1.5
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 27.64 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.24 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.2→6 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.1 / Classification: refinement X-PLOR / Version: 3.1 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller


















 PDBj
PDBj









