+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1mr0 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
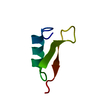
| Title | SOLUTION NMR STRUCTURE OF AGRP(87-120; C105A) | |||||||||
 Components Components | AGOUTI RELATED PROTEIN | |||||||||
 Keywords Keywords |  SIGNALING PROTEIN / RATIONAL PROTEIN DESIGN / ICK / SIGNALING PROTEIN / RATIONAL PROTEIN DESIGN / ICK /  INHIBITOR CYSTINE KNOT / AGRP / AGOUTI-RELATED PROTEIN / MELANOCORTIN INHIBITOR CYSTINE KNOT / AGRP / AGOUTI-RELATED PROTEIN / MELANOCORTIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationlong-day photoperiodism / type 1 melanocortin receptor binding / positive regulation of feeding behavior / adult feeding behavior /  regulation of feeding behavior / neuropeptide hormone activity / regulation of feeding behavior / neuropeptide hormone activity /  feeding behavior / feeding behavior /  eating behavior / neuropeptide signaling pathway / maternal process involved in female pregnancy ...long-day photoperiodism / type 1 melanocortin receptor binding / positive regulation of feeding behavior / adult feeding behavior / eating behavior / neuropeptide signaling pathway / maternal process involved in female pregnancy ...long-day photoperiodism / type 1 melanocortin receptor binding / positive regulation of feeding behavior / adult feeding behavior /  regulation of feeding behavior / neuropeptide hormone activity / regulation of feeding behavior / neuropeptide hormone activity /  feeding behavior / feeding behavior /  eating behavior / neuropeptide signaling pathway / maternal process involved in female pregnancy / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / hormone-mediated signaling pathway / response to insulin / Golgi lumen / eating behavior / neuropeptide signaling pathway / maternal process involved in female pregnancy / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / hormone-mediated signaling pathway / response to insulin / Golgi lumen /  circadian rhythm / circadian rhythm /  signaling receptor binding / neuronal cell body / signaling receptor binding / neuronal cell body /  extracellular space extracellular spaceSimilarity search - Function | |||||||||
| Method |  SOLUTION NMR / torsion angle dynamics SOLUTION NMR / torsion angle dynamics | |||||||||
 Authors Authors | Jackson, P.J. / Mcnulty, J.C. / Yang, Y.K. / Thompson, D.A. / Chai, B. / Gantz, I. / Barsh, G.S. / Millhauser, G.M. | |||||||||
 Citation Citation |  Journal: Biochemistry / Year: 2002 Journal: Biochemistry / Year: 2002Title: Design, pharmacology, and NMR structure of a minimized cystine knot with agouti-related protein activity. Authors: Jackson, P.J. / McNulty, J.C. / Yang, Y.K. / Thompson, D.A. / Chai, B. / Gantz, I. / Barsh, G.S. / Millhauser, G.L. #1:  Journal: Biochemistry / Year: 2001 Journal: Biochemistry / Year: 2001Title: High Resolution NMR Structure of the Chemically-Synthesized Melanocortin Receptor Binding Domain of Agrp(87-132) of the Agouti-Related Protein. Authors: Mcnulty, J.C. / Thompson, D.A. / Bolin, K.A. / Wilken, J. / Barsh, G.S. / Millhauser, G.M. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1mr0.cif.gz 1mr0.cif.gz | 391.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1mr0.ent.gz pdb1mr0.ent.gz | 340.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1mr0.json.gz 1mr0.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mr/1mr0 https://data.pdbj.org/pub/pdb/validation_reports/mr/1mr0 ftp://data.pdbj.org/pub/pdb/validation_reports/mr/1mr0 ftp://data.pdbj.org/pub/pdb/validation_reports/mr/1mr0 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 3912.590 Da / Num. of mol.: 1 / Fragment: RESIDUES 87-120 / Mutation: C105A / Source method: obtained synthetically Details: THE PROTEIN WAS CHEMICALLY SYNTHESIZED. THE SEQUENCE OF THE PROTEIN IS NATURALLY FOUND IN HOMO SAPIENS. THE SYNTHETIC METHOD IS STANDARD SOLID-PHASE SYNTHESIS FOLLOWED BY AQUEOUS OXIDATIVE FOLDING. References: UniProt: O00253 |
|---|
-Experimental details
-Experiment
| Experiment | Method:  SOLUTION NMR SOLUTION NMR | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||||||
| NMR details | Text: THE NOESY, TOCSY AND DQF-COSY EXPERIMENTS WERE PERFORMED AT 800 MHZ; THE HX STUDY WAS CONDUCTED AT 500 MHZ. |
- Sample preparation
Sample preparation
| Details |
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample conditions |
| |||||||||||||||
Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Radiation wavelength | Relative weight: 1 | |||||||||||||||
| NMR spectrometer |
|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: torsion angle dynamics / Software ordinal: 1 Details: STRUCTURE CALCULATIONS WERE ASSISTED WITH AUTOMATED ASSIGNMENT. FINAL STRUCTURES WERE CALCULATED USING 602 NOE-BASED UPPER LIMIT RESTRAINTS AND 23 ALPHA-TO- AMIDE 3-BOND J-COUPLING CONSTANTS. | ||||||||||||||||||||||||
| NMR representative | Selection criteria: lowest target function | ||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: target function / Conformers calculated total number: 1000 / Conformers submitted total number: 40 |
 Movie
Movie Controller
Controller













 PDBj
PDBj







