[English] 日本語
 Yorodumi
Yorodumi- PDB-1cvu: CRYSTAL STRUCTURE OF ARACHIDONIC ACID BOUND TO THE CYCLOOXYGENASE... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1cvu | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURE OF ARACHIDONIC ACID BOUND TO THE CYCLOOXYGENASE ACTIVE SITE OF COX-2 | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | OXIDOREDUCTASE/peptide /  COX-2 / COX-2 /  CYCLOOXYGENASE / CYCLOOXYGENASE /  PROSTAGLANDIN / PROSTAGLANDIN /  ARACHIDONATE / ARACHIDONATE /  ENDOPEROXIDE / OXIDOREDUCTASE-peptide complex ENDOPEROXIDE / OXIDOREDUCTASE-peptide complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationBiosynthesis of DHA-derived SPMs / Biosynthesis of EPA-derived SPMs / Biosynthesis of DPAn-3 SPMs / Biosynthesis of electrophilic ω-3 PUFA oxo-derivatives / Synthesis of 15-eicosatetraenoic acid derivatives / cellular response to non-ionic osmotic stress / positive regulation of platelet-derived growth factor production / cellular response to homocysteine /  hair cycle / Nicotinamide salvaging ...Biosynthesis of DHA-derived SPMs / Biosynthesis of EPA-derived SPMs / Biosynthesis of DPAn-3 SPMs / Biosynthesis of electrophilic ω-3 PUFA oxo-derivatives / Synthesis of 15-eicosatetraenoic acid derivatives / cellular response to non-ionic osmotic stress / positive regulation of platelet-derived growth factor production / cellular response to homocysteine / hair cycle / Nicotinamide salvaging ...Biosynthesis of DHA-derived SPMs / Biosynthesis of EPA-derived SPMs / Biosynthesis of DPAn-3 SPMs / Biosynthesis of electrophilic ω-3 PUFA oxo-derivatives / Synthesis of 15-eicosatetraenoic acid derivatives / cellular response to non-ionic osmotic stress / positive regulation of platelet-derived growth factor production / cellular response to homocysteine /  hair cycle / Nicotinamide salvaging / Synthesis of Prostaglandins (PG) and Thromboxanes (TX) / positive regulation of fibroblast growth factor production / hair cycle / Nicotinamide salvaging / Synthesis of Prostaglandins (PG) and Thromboxanes (TX) / positive regulation of fibroblast growth factor production /  prostaglandin-endoperoxide synthase / prostaglandin-endoperoxide synthase /  prostaglandin-endoperoxide synthase activity / negative regulation of synaptic transmission, dopaminergic / cellular response to lead ion / response to nematode / positive regulation of transforming growth factor beta production / negative regulation of intrinsic apoptotic signaling pathway in response to osmotic stress / positive regulation of prostaglandin biosynthetic process / regulation of neuroinflammatory response / positive regulation of synaptic plasticity / oxidoreductase activity, acting on single donors with incorporation of molecular oxygen, incorporation of two atoms of oxygen / response to fatty acid / positive regulation of smooth muscle contraction / cyclooxygenase pathway / response to fructose / positive regulation of fever generation / prostaglandin secretion / response to vitamin D / cellular response to fluid shear stress / nuclear outer membrane / response to angiotensin / response to manganese ion / nuclear inner membrane / prostaglandin biosynthetic process / negative regulation of smooth muscle contraction / cellular response to ATP / positive regulation of cell migration involved in sprouting angiogenesis / maintenance of blood-brain barrier / prostaglandin-endoperoxide synthase activity / negative regulation of synaptic transmission, dopaminergic / cellular response to lead ion / response to nematode / positive regulation of transforming growth factor beta production / negative regulation of intrinsic apoptotic signaling pathway in response to osmotic stress / positive regulation of prostaglandin biosynthetic process / regulation of neuroinflammatory response / positive regulation of synaptic plasticity / oxidoreductase activity, acting on single donors with incorporation of molecular oxygen, incorporation of two atoms of oxygen / response to fatty acid / positive regulation of smooth muscle contraction / cyclooxygenase pathway / response to fructose / positive regulation of fever generation / prostaglandin secretion / response to vitamin D / cellular response to fluid shear stress / nuclear outer membrane / response to angiotensin / response to manganese ion / nuclear inner membrane / prostaglandin biosynthetic process / negative regulation of smooth muscle contraction / cellular response to ATP / positive regulation of cell migration involved in sprouting angiogenesis / maintenance of blood-brain barrier /  bone mineralization / negative regulation of calcium ion transport / bone mineralization / negative regulation of calcium ion transport /  decidualization / negative regulation of cell cycle / positive regulation of vascular endothelial growth factor production / response to tumor necrosis factor / brown fat cell differentiation / response to glucocorticoid / keratinocyte differentiation / positive regulation of vasoconstriction / decidualization / negative regulation of cell cycle / positive regulation of vascular endothelial growth factor production / response to tumor necrosis factor / brown fat cell differentiation / response to glucocorticoid / keratinocyte differentiation / positive regulation of vasoconstriction /  embryo implantation / positive regulation of brown fat cell differentiation / positive regulation of synaptic transmission, glutamatergic / embryo implantation / positive regulation of brown fat cell differentiation / positive regulation of synaptic transmission, glutamatergic /  learning / response to cytokine / learning / response to cytokine /  caveola / positive regulation of smooth muscle cell proliferation / caveola / positive regulation of smooth muscle cell proliferation /  peroxidase activity / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process / peroxidase activity / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process /  memory / memory /  regulation of blood pressure / positive regulation of protein import into nucleus / cellular response to mechanical stimulus / positive regulation of nitric oxide biosynthetic process / positive regulation of peptidyl-serine phosphorylation / response to estradiol / cellular response to heat / regulation of cell population proliferation / cellular response to hypoxia / regulation of blood pressure / positive regulation of protein import into nucleus / cellular response to mechanical stimulus / positive regulation of nitric oxide biosynthetic process / positive regulation of peptidyl-serine phosphorylation / response to estradiol / cellular response to heat / regulation of cell population proliferation / cellular response to hypoxia /  angiogenesis / response to oxidative stress / response to lipopolysaccharide / neuron projection / response to xenobiotic stimulus / positive regulation of apoptotic process / negative regulation of cell population proliferation / angiogenesis / response to oxidative stress / response to lipopolysaccharide / neuron projection / response to xenobiotic stimulus / positive regulation of apoptotic process / negative regulation of cell population proliferation /  heme binding / positive regulation of cell population proliferation / endoplasmic reticulum membrane / negative regulation of apoptotic process / heme binding / positive regulation of cell population proliferation / endoplasmic reticulum membrane / negative regulation of apoptotic process /  enzyme binding / protein homodimerization activity / protein-containing complex / enzyme binding / protein homodimerization activity / protein-containing complex /  metal ion binding / metal ion binding /  cytoplasm cytoplasmSimilarity search - Function | |||||||||
| Biological species |   Mus musculus (house mouse) Mus musculus (house mouse) | |||||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2.4 Å X-RAY DIFFRACTION / Resolution: 2.4 Å | |||||||||
 Authors Authors | Kiefer, J.R. / Pawlitz, J.L. / Moreland, K.T. / Stegeman, R.A. / Gierse, J.K. / Stevens, A.M. / Goodwin, D.C. / Rowlinson, S.W. / Marnett, L.J. / Stallings, W.C. / Kurumbail, R.G. | |||||||||
 Citation Citation |  Journal: Nature / Year: 2000 Journal: Nature / Year: 2000Title: Structural insights into the stereochemistry of the cyclooxygenase reaction. Authors: Kiefer, J.R. / Pawlitz, J.L. / Moreland, K.T. / Stegeman, R.A. / Hood, W.F. / Gierse, J.K. / Stevens, A.M. / Goodwin, D.C. / Rowlinson, S.W. / Marnett, L.J. / Stallings, W.C. / Kurumbail, R.G. #1:  Journal: Nature / Year: 1996 Journal: Nature / Year: 1996Title: Structural Basis for Selective Inhibition of by Anti-Inflammatory Agents Authors: Kurumbail, R.G. / Stevens, A.M. / McDonald, J.J. / STEGEMAN, R.A. / PAK, J.Y. / GILDEHAUS, D. / MIYASHIRO, J.M. / PENNING, T.D. / SEIBERT, K. / ISAKSON, P.C. / STALLINGS, W.C. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1cvu.cif.gz 1cvu.cif.gz | 254.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1cvu.ent.gz pdb1cvu.ent.gz | 209.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1cvu.json.gz 1cvu.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/cv/1cvu https://data.pdbj.org/pub/pdb/validation_reports/cv/1cvu ftp://data.pdbj.org/pub/pdb/validation_reports/cv/1cvu ftp://data.pdbj.org/pub/pdb/validation_reports/cv/1cvu | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein / Protein/peptide , 2 types, 3 molecules ABF
| #1: Protein |  Cyclooxygenase / EC 1.14.99.1 Cyclooxygenase / EC 1.14.99.1Mass: 63416.359 Da / Num. of mol.: 2 / Fragment: PROSTAGLANDIN H2 SYNTHASE-2 / Mutation: H207A (COX-1 NUMBERING) Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Mus musculus (house mouse) / Plasmid: PVL1393 / Production host: Mus musculus (house mouse) / Plasmid: PVL1393 / Production host:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm)References: UniProt: Q05769,  prostaglandin-endoperoxide synthase prostaglandin-endoperoxide synthase#2: Protein/peptide | | Mass: 907.000 Da / Num. of mol.: 1 / Source method: obtained synthetically / Details: SYNTHETIC |
|---|
-Sugars , 4 types, 11 molecules 


| #3: Polysaccharide |  / Mass: 424.401 Da / Num. of mol.: 2 / Source method: obtained synthetically / Mass: 424.401 Da / Num. of mol.: 2 / Source method: obtained synthetically#4: Polysaccharide |  / Mass: 748.682 Da / Num. of mol.: 2 / Mass: 748.682 Da / Num. of mol.: 2Source method: isolated from a genetically manipulated source #5: Sugar |  N-Acetylglucosamine N-Acetylglucosamine#6: Sugar | ChemComp-BOG /  Octyl glucoside Octyl glucoside |
|---|
-Non-polymers , 2 types, 773 molecules 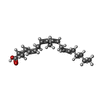


| #7: Chemical |  Arachidonic acid Arachidonic acid#8: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.96 Å3/Da / Density % sol: 58.46 % | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop / pH: 8 Details: MONOMETHYL PEG 550, MAGNESIUM SULFATE, ARACHIDNOIC ACID, EPPS, B-OG, pH 8.0, VAPOR DIFFUSION, HANGING DROP, temperature 291K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 18 ℃ / Details: Stevens, A.M., (1999) J. Cryst. Growth., 196, 350. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 130 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RU300 / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RU300 / Wavelength: 1.5418 |
| Detector | Type: RIGAKU RAXIS IV / Detector: IMAGE PLATE / Date: Oct 8, 1998 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.5418 Å / Relative weight: 1 : 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→35 Å / Num. all: 58608 / Num. obs: 212739 / % possible obs: 98.8 % / Observed criterion σ(F): 0 / Observed criterion σ(I): -3 / Redundancy: 3.6 % / Biso Wilson estimate: 26.8 Å2 / Rmerge(I) obs: 0.078 / Net I/σ(I): 9.6 |
| Reflection shell | Resolution: 2.4→2.49 Å / Redundancy: 2.5 % / Rmerge(I) obs: 0.241 / % possible all: 97.8 |
| Reflection | *PLUS Num. obs: 58608 / Num. measured all: 212739 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 2.4→35 Å / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: ENGH & HUBER Details: CONJUGATE GRADIENT, MOLECULAR DYNAMICS, INDIVIDUAL ATOM ISOTROPIC B, BULK SOLVENT CORRECTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.4→35 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.851 / Classification: refinement X-PLOR / Version: 3.851 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Highest resolution: 2.4 Å / Lowest resolution: 35 Å / σ(F): 0 / Rfactor obs: 0.206 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller

















 PDBj
PDBj






