+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
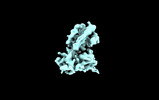
| Title | Structure of EP141-C3aR-Go complex | |||||||||||||||
 Map data Map data | ||||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords |  GPCR / GPCR /  SIGNALING PROTEIN SIGNALING PROTEIN | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationcomplement component C3a receptor activity / complement component C5a receptor activity / mu-type opioid receptor binding / complement receptor mediated signaling pathway /  corticotropin-releasing hormone receptor 1 binding / positive regulation of neutrophil chemotaxis / corticotropin-releasing hormone receptor 1 binding / positive regulation of neutrophil chemotaxis /  blood circulation / azurophil granule membrane / dopamine receptor signaling pathway / positive regulation of macrophage chemotaxis ...complement component C3a receptor activity / complement component C5a receptor activity / mu-type opioid receptor binding / complement receptor mediated signaling pathway / blood circulation / azurophil granule membrane / dopamine receptor signaling pathway / positive regulation of macrophage chemotaxis ...complement component C3a receptor activity / complement component C5a receptor activity / mu-type opioid receptor binding / complement receptor mediated signaling pathway /  corticotropin-releasing hormone receptor 1 binding / positive regulation of neutrophil chemotaxis / corticotropin-releasing hormone receptor 1 binding / positive regulation of neutrophil chemotaxis /  blood circulation / azurophil granule membrane / dopamine receptor signaling pathway / positive regulation of macrophage chemotaxis / positive regulation of vascular endothelial growth factor production / G protein-coupled serotonin receptor binding / specific granule membrane / blood circulation / azurophil granule membrane / dopamine receptor signaling pathway / positive regulation of macrophage chemotaxis / positive regulation of vascular endothelial growth factor production / G protein-coupled serotonin receptor binding / specific granule membrane /  Purinergic signaling in leishmaniasis infection / Peptide ligand-binding receptors / Purinergic signaling in leishmaniasis infection / Peptide ligand-binding receptors /  muscle contraction / muscle contraction /  Regulation of Complement cascade / G protein-coupled receptor activity / calcium-mediated signaling / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Sensory perception of sweet, bitter, and umami (glutamate) taste / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / adenylate cyclase-activating dopamine receptor signaling pathway / ADP signalling through P2Y purinoceptor 1 / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / sensory perception of taste / GPER1 signaling / G-protein beta-subunit binding / Regulation of Complement cascade / G protein-coupled receptor activity / calcium-mediated signaling / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Sensory perception of sweet, bitter, and umami (glutamate) taste / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / adenylate cyclase-activating dopamine receptor signaling pathway / ADP signalling through P2Y purinoceptor 1 / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / sensory perception of taste / GPER1 signaling / G-protein beta-subunit binding /  heterotrimeric G-protein complex / Inactivation, recovery and regulation of the phototransduction cascade / positive regulation of angiogenesis / heterotrimeric G-protein complex / Inactivation, recovery and regulation of the phototransduction cascade / positive regulation of angiogenesis /  extracellular vesicle / G alpha (12/13) signalling events / extracellular vesicle / G alpha (12/13) signalling events /  chemotaxis / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / retina development in camera-type eye / chemotaxis / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / retina development in camera-type eye /  GTPase binding / Ca2+ pathway / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / G alpha (i) signalling events / fibroblast proliferation / G alpha (s) signalling events / G alpha (q) signalling events / Ras protein signal transduction / cell population proliferation / Extra-nuclear estrogen signaling / GTPase binding / Ca2+ pathway / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / G alpha (i) signalling events / fibroblast proliferation / G alpha (s) signalling events / G alpha (q) signalling events / Ras protein signal transduction / cell population proliferation / Extra-nuclear estrogen signaling /  inflammatory response / G protein-coupled receptor signaling pathway / lysosomal membrane / inflammatory response / G protein-coupled receptor signaling pathway / lysosomal membrane /  GTPase activity / GTPase activity /  synapse / Neutrophil degranulation / protein-containing complex binding / GTP binding / synapse / Neutrophil degranulation / protein-containing complex binding / GTP binding /  signal transduction / extracellular exosome / signal transduction / extracellular exosome /  membrane / membrane /  metal ion binding / metal ion binding /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | |||||||||||||||
| Biological species |   Homo sapiens (human) / Homo sapiens (human) /   Mus musculus (house mouse) Mus musculus (house mouse) | |||||||||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.1 Å cryo EM / Resolution: 3.1 Å | |||||||||||||||
 Authors Authors | Yadav MK / Yadav R / Maharana J / Sarma P / Banerjee R / Shukla AK / Gati C | |||||||||||||||
| Funding support |  India, India,  United Kingdom, 4 items United Kingdom, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Cell / Year: 2023 Journal: Cell / Year: 2023Title: Molecular basis of anaphylatoxin binding, activation, and signaling bias at complement receptors. Authors: Manish K Yadav / Jagannath Maharana / Ravi Yadav / Shirsha Saha / Parishmita Sarma / Chahat Soni / Vinay Singh / Sayantan Saha / Manisankar Ganguly / Xaria X Li / Samanwita Mohapatra / Sudha ...Authors: Manish K Yadav / Jagannath Maharana / Ravi Yadav / Shirsha Saha / Parishmita Sarma / Chahat Soni / Vinay Singh / Sayantan Saha / Manisankar Ganguly / Xaria X Li / Samanwita Mohapatra / Sudha Mishra / Htet A Khant / Mohamed Chami / Trent M Woodruff / Ramanuj Banerjee / Arun K Shukla / Cornelius Gati /     Abstract: The complement system is a critical part of our innate immune response, and the terminal products of this cascade, anaphylatoxins C3a and C5a, exert their physiological and pathophysiological ...The complement system is a critical part of our innate immune response, and the terminal products of this cascade, anaphylatoxins C3a and C5a, exert their physiological and pathophysiological responses primarily via two GPCRs, C3aR and C5aR1. However, the molecular mechanism of ligand recognition, activation, and signaling bias of these receptors remains mostly elusive. Here, we present nine cryo-EM structures of C3aR and C5aR1 activated by their natural and synthetic agonists, which reveal distinct binding pocket topologies of complement anaphylatoxins and provide key insights into receptor activation and transducer coupling. We also uncover the structural basis of a naturally occurring mechanism to dampen the inflammatory response of C5a via proteolytic cleavage of the terminal arginine and the G-protein signaling bias elicited by a peptide agonist of C3aR identified here. In summary, our study elucidates the innerworkings of the complement anaphylatoxin receptors and should facilitate structure-guided drug discovery to target these receptors in a spectrum of disorders. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_36001.map.gz emd_36001.map.gz | 85.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36001-v30.xml emd-36001-v30.xml emd-36001.xml emd-36001.xml | 21.7 KB 21.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_36001_fsc.xml emd_36001_fsc.xml | 9.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_36001.png emd_36001.png | 104.1 KB | ||
| Filedesc metadata |  emd-36001.cif.gz emd-36001.cif.gz | 6.9 KB | ||
| Others |  emd_36001_half_map_1.map.gz emd_36001_half_map_1.map.gz emd_36001_half_map_2.map.gz emd_36001_half_map_2.map.gz | 84.6 MB 84.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36001 http://ftp.pdbj.org/pub/emdb/structures/EMD-36001 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36001 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36001 | HTTPS FTP |
-Related structure data
| Related structure data |  8j6dMC  8i95C  8i97C  8i9aC  8i9lC  8i9sC  8ia2C  8jzzC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_36001.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36001.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.15 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_36001_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_36001_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Structure of EP141-C3aR-Go complex
| Entire | Name: Structure of EP141-C3aR-Go complex |
|---|---|
| Components |
|
-Supramolecule #1: Structure of EP141-C3aR-Go complex
| Supramolecule | Name: Structure of EP141-C3aR-Go complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Guanine nucleotide-binding protein G(o) subunit alpha
| Macromolecule | Name: Guanine nucleotide-binding protein G(o) subunit alpha / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 28.193939 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MGHHHHHHEN LYFQGTLSAE ERAALERSKA IEKNLKEDGI SAAKDVKLLL LGADNSGKST IVKQMKIIHG GSGGSGGTTG IVETHFTFK NLHFRLFDVG GQRSERKKWI HCFEDVTAII FCVDLSDYDQ VLHEDETTNR MHESLMLFDS ICNNKFFIDT S IILFLNKK ...String: MGHHHHHHEN LYFQGTLSAE ERAALERSKA IEKNLKEDGI SAAKDVKLLL LGADNSGKST IVKQMKIIHG GSGGSGGTTG IVETHFTFK NLHFRLFDVG GQRSERKKWI HCFEDVTAII FCVDLSDYDQ VLHEDETTNR MHESLMLFDS ICNNKFFIDT S IILFLNKK DLFGEKIKKS PLTICFPEYT GPNTYEDAAA YIQAQFESKN RSPNKEIYCH MTCATDTNNA QVIFDAVTDI II ANNLRGC GLY UniProtKB: Guanine nucleotide-binding protein G(o) subunit alpha, Guanine nucleotide-binding protein G(o) subunit alpha |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 38.534062 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MHHHHHHGSS GSELDQLRQE AEQLKNQIRD ARKACADATL SQITNNIDPV GRIQMRTRRT LRGHLAKIYA MHWGTDSRLL VSASQDGKL IIWDSYTTNK VHAIPLRSSW VMTCAYAPSG NYVACGGLDN ICSIYNLKTR EGNVRVSREL AGHTGYLSCC R FLDDNQIV ...String: MHHHHHHGSS GSELDQLRQE AEQLKNQIRD ARKACADATL SQITNNIDPV GRIQMRTRRT LRGHLAKIYA MHWGTDSRLL VSASQDGKL IIWDSYTTNK VHAIPLRSSW VMTCAYAPSG NYVACGGLDN ICSIYNLKTR EGNVRVSREL AGHTGYLSCC R FLDDNQIV TSSGDTTCAL WDIETGQQTT TFTGHTGDVM SLSLAPDTRL FVSGACDASA KLWDVREGMC RQTFTGHESD IN AICFFPN GNAFATGSDD ATCRLFDLRA DQELMTYSHD NIICGITSVS FSKSGRLLLA GYDDFNCNVW DALKADRAGV LAG HDNRVS CLGVTDDGMA VATGSWDSFL KIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: C3a anaphylatoxin chemotactic receptor
| Macromolecule | Name: C3a anaphylatoxin chemotactic receptor / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 59.808422 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MGKTIIALSY IFCLVFADYK DDDDAANFTP VNGSSGNQSV RLVTSSSLEV LFQGPGSASF SAETNSTDLL SQPWNEPPVI LSMVILSLT FLLGLPGNGL VLWVAGLKMQ RTVNTIWFLH LTLADLLCCL SLPFSLAHLA LQGQWPYGRF LCKLIPSIIV L NMFASVFL ...String: MGKTIIALSY IFCLVFADYK DDDDAANFTP VNGSSGNQSV RLVTSSSLEV LFQGPGSASF SAETNSTDLL SQPWNEPPVI LSMVILSLT FLLGLPGNGL VLWVAGLKMQ RTVNTIWFLH LTLADLLCCL SLPFSLAHLA LQGQWPYGRF LCKLIPSIIV L NMFASVFL LTAISLDRCL VVFKPIWCQN HRNVGMACSI CGCIWVVAFV MCIPVFVYRE IFTTDNHNRC GYKFGLSSSL DY PDFYGDP LENRSLENIV QPPGEMNDRL DPSSFQTNDH PWTVPTVFQP QTFQRPSADS LPRGSARLTS QNLYSNVFKP ADV VSPKIP SGFPIEDHET SPLDNSDAFL STHLKLFPSA SSNSFYESEL PQGFQDYYNL GQFTDDDQVP TPLVAITITR LVVG FLLPS VIMIACYSFI VFRMQRGRFA KSQSKTFRVA VVVVAVFLVC WTPYHIFGVL SLLTDPETPL GKTLMSWDHV CIALA SANS CFNPFLYALL GKDFRKKARQ SIQGILEAAF SEELTRSTHC PSNNVISERN STTV UniProtKB: C3a anaphylatoxin chemotactic receptor |
-Macromolecule #4: EP141 peptide agonist
| Macromolecule | Name: EP141 peptide agonist / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 1.825188 KDa |
| Sequence | String: WWGKKYRASK LGLAR |
-Macromolecule #5: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #6: Antibody fragment - ScFv16
| Macromolecule | Name: Antibody fragment - ScFv16 / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Mus musculus (house mouse) Mus musculus (house mouse) |
| Molecular weight | Theoretical: 26.466486 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFTI SRDDPKNTLF LQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSDIV MTQATSSVPV TPGESVSISC R SSKSLLHS ...String: DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFTI SRDDPKNTLF LQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSDIV MTQATSSVPV TPGESVSISC R SSKSLLHS NGNTYLYWFL QRPGQSPQLL IYRMSNLASG VPDRFSGSGS GTAFTLTISR LEAEDVGVYY CMQHLEYPLT FG AGTKLEL K |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS GLACIOS |
|---|---|
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.8 µm Bright-field microscopy / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.8 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 50.0 e/Å2 |
 Movie
Movie Controller
Controller














































 Z
Z Y
Y X
X


















