[English] 日本語
 Yorodumi
Yorodumi- EMDB-26952: Local refinement of ankyrin-1 (C-terminal half), class 1 of eryth... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Local refinement of ankyrin-1 (C-terminal half), class 1 of erythrocyte ankyrin-1 complex | |||||||||
 Map data Map data | Main map used for model building/refinement. Density modified and cropped with phenix.resolve_cryo_em, and resampled on fine grid with relion_image_handler. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords |  Membrane Protein / Membrane Protein /  ankyrin complex / ankyrin complex /  Erythrocyte / Erythrocyte /  STRUCTURAL PROTEIN STRUCTURAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationspectrin-associated cytoskeleton / positive regulation of organelle organization / maintenance of epithelial cell apical/basal polarity / NrCAM interactions /  ankyrin-1 complex / Neurofascin interactions / CHL1 interactions / cytoskeletal anchor activity / ankyrin-1 complex / Neurofascin interactions / CHL1 interactions / cytoskeletal anchor activity /  M band / Interaction between L1 and Ankyrins ...spectrin-associated cytoskeleton / positive regulation of organelle organization / maintenance of epithelial cell apical/basal polarity / NrCAM interactions / M band / Interaction between L1 and Ankyrins ...spectrin-associated cytoskeleton / positive regulation of organelle organization / maintenance of epithelial cell apical/basal polarity / NrCAM interactions /  ankyrin-1 complex / Neurofascin interactions / CHL1 interactions / cytoskeletal anchor activity / ankyrin-1 complex / Neurofascin interactions / CHL1 interactions / cytoskeletal anchor activity /  M band / Interaction between L1 and Ankyrins / M band / Interaction between L1 and Ankyrins /  spectrin binding / spectrin binding /  exocytosis / exocytosis /  axolemma / endoplasmic reticulum to Golgi vesicle-mediated transport / COPI-mediated anterograde transport / cytoskeleton organization / axolemma / endoplasmic reticulum to Golgi vesicle-mediated transport / COPI-mediated anterograde transport / cytoskeleton organization /  sarcoplasmic reticulum / protein localization to plasma membrane / sarcoplasmic reticulum / protein localization to plasma membrane /  sarcolemma / cytoplasmic side of plasma membrane / structural constituent of cytoskeleton / Z disc / sarcolemma / cytoplasmic side of plasma membrane / structural constituent of cytoskeleton / Z disc /  ATPase binding / ATPase binding /  postsynaptic membrane / basolateral plasma membrane / postsynaptic membrane / basolateral plasma membrane /  protein phosphatase binding / transmembrane transporter binding / protein phosphatase binding / transmembrane transporter binding /  cytoskeleton / neuron projection / structural molecule activity / cytoskeleton / neuron projection / structural molecule activity /  enzyme binding / enzyme binding /  signal transduction / signal transduction /  plasma membrane / plasma membrane /  cytosol cytosolSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
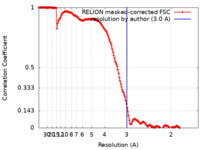
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.0 Å cryo EM / Resolution: 3.0 Å | |||||||||
 Authors Authors | Vallese F / Kim K / Yen LY / Johnston JD / Noble AJ / Cali T / Clarke OB | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2022 Journal: Nat Struct Mol Biol / Year: 2022Title: Architecture of the human erythrocyte ankyrin-1 complex. Authors: Francesca Vallese / Kookjoo Kim / Laura Y Yen / Jake D Johnston / Alex J Noble / Tito Calì / Oliver Biggs Clarke /   Abstract: The stability and shape of the erythrocyte membrane is provided by the ankyrin-1 complex, but how it tethers the spectrin-actin cytoskeleton to the lipid bilayer and the nature of its association ...The stability and shape of the erythrocyte membrane is provided by the ankyrin-1 complex, but how it tethers the spectrin-actin cytoskeleton to the lipid bilayer and the nature of its association with the band 3 anion exchanger and the Rhesus glycoproteins remains unknown. Here we present structures of ankyrin-1 complexes purified from human erythrocytes. We reveal the architecture of a core complex of ankyrin-1, the Rhesus proteins RhAG and RhCE, the band 3 anion exchanger, protein 4.2, glycophorin A and glycophorin B. The distinct T-shaped conformation of membrane-bound ankyrin-1 facilitates recognition of RhCE and, unexpectedly, the water channel aquaporin-1. Together, our results uncover the molecular details of ankyrin-1 association with the erythrocyte membrane, and illustrate the mechanism of ankyrin-mediated membrane protein clustering. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26952.map.gz emd_26952.map.gz | 63.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26952-v30.xml emd-26952-v30.xml emd-26952.xml emd-26952.xml | 32.7 KB 32.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_26952_fsc.xml emd_26952_fsc.xml | 15.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_26952.png emd_26952.png | 41.4 KB | ||
| Filedesc metadata |  emd-26952.cif.gz emd-26952.cif.gz | 7.2 KB | ||
| Others |  emd_26952_additional_1.map.gz emd_26952_additional_1.map.gz emd_26952_additional_2.map.gz emd_26952_additional_2.map.gz emd_26952_additional_3.map.gz emd_26952_additional_3.map.gz emd_26952_additional_4.map.gz emd_26952_additional_4.map.gz emd_26952_half_map_1.map.gz emd_26952_half_map_1.map.gz emd_26952_half_map_2.map.gz emd_26952_half_map_2.map.gz | 322.3 MB 322.3 MB 510.1 KB 328.2 MB 63.6 MB 63.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26952 http://ftp.pdbj.org/pub/emdb/structures/EMD-26952 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26952 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26952 | HTTPS FTP |
-Related structure data
| Related structure data |  7v0xMC  7uz3C  7uzeC  7uzqC  7uzsC  7uzuC  7uzvC  7v07C  7v0kC  7v0mC  7v0qC  7v0sC  7v0tC  7v0uC  7v0yC  7v19C  8crqC  8crrC  8crtC  8cs9C  8cslC  8csvC  8cswC  8csxC  8csyC  8ct2C  8ct3C  8cteC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26952.map.gz / Format: CCP4 / Size: 68.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26952.map.gz / Format: CCP4 / Size: 68.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Main map used for model building/refinement. Density modified and cropped with phenix.resolve_cryo_em, and resampled on fine grid with relion_image_handler. | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.415 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Half map 1 (unmodified)
| File | emd_26952_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 (unmodified) | ||||||||||||
| Projections & Slices |
| ||||||||||||
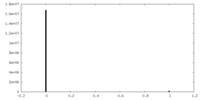
| Density Histograms |
-Additional map: Half map 2 (unmodified)
| File | emd_26952_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 (unmodified) | ||||||||||||
| Projections & Slices |
| ||||||||||||
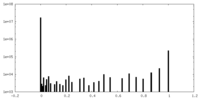
| Density Histograms |
-Additional map: Mask used for FSC calculation
| File | emd_26952_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Mask used for FSC calculation | ||||||||||||
| Projections & Slices |
| ||||||||||||
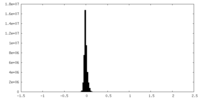
| Density Histograms |
-Additional map: B-factor sharpened map.
| File | emd_26952_additional_4.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | B-factor sharpened map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
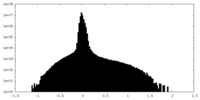
| Density Histograms |
-Half map: Half map 1 (cropped and resampled to match main map).
| File | emd_26952_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 (cropped and resampled to match main map). | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 2 (cropped and resampled to match main map).
| File | emd_26952_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 (cropped and resampled to match main map). | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Erythrocyte ankyrin-1 complex
| Entire | Name: Erythrocyte ankyrin-1 complex |
|---|---|
| Components |
|
-Supramolecule #1: Erythrocyte ankyrin-1 complex
| Supramolecule | Name: Erythrocyte ankyrin-1 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: Purified by density gradient centrifugation and size exclusion chromatography from digitonin-solubilized erythrocyte membranes. |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) / Organ: Blood / Tissue: Erythrocytes / Location in cell: Plasma membrane Homo sapiens (human) / Organ: Blood / Tissue: Erythrocytes / Location in cell: Plasma membrane |
-Macromolecule #1: Ankyrin-1
| Macromolecule | Name: Ankyrin-1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 206.522625 KDa |
| Sequence | String: MPYSVGFREA DAATSFLRAA RSGNLDKALD HLRNGVDINT CNQNGLNGLH LASKEGHVKM VVELLHKEII LETTTKKGNT ALHIAALAG QDEVVRELVN YGANVNAQSQ KGFTPLYMAA QENHLEVVKF LLENGANQNV ATEDGFTPLA VALQQGHENV V AHLINYGT ...String: MPYSVGFREA DAATSFLRAA RSGNLDKALD HLRNGVDINT CNQNGLNGLH LASKEGHVKM VVELLHKEII LETTTKKGNT ALHIAALAG QDEVVRELVN YGANVNAQSQ KGFTPLYMAA QENHLEVVKF LLENGANQNV ATEDGFTPLA VALQQGHENV V AHLINYGT KGKVRLPALH IAARNDDTRT AAVLLQNDPN PDVLSKTGFT PLHIAAHYEN LNVAQLLLNR GASVNFTPQN GI TPLHIAS RRGNVIMVRL LLDRGAQIET KTKDELTPLH CAARNGHVRI SEILLDHGAP IQAKTKNGLS PIHMAAQGDH LDC VRLLLQ YDAEIDDITL DHLTPLHVAA HCGHHRVAKV LLDKGAKPNS RALNGFTPLH IACKKNHVRV MELLLKTGAS IDAV TESGL TPLHVASFMG HLPIVKNLLQ RGASPNVSNV KVETPLHMAA RAGHTEVAKY LLQNKAKVNA KAKDDQTPLH CAARI GHTN MVKLLLENNA NPNLATTAGH TPLHIAAREG HVETVLALLE KEASQACMTK KGFTPLHVAA KYGKVRVAEL LLERDA HPN AAGKNGLTPL HVAVHHNNLD IVKLLLPRGG SPHSPAWNGY TPLHIAAKQN QVEVARSLLQ YGGSANAESV QGVTPLH LA AQEGHAEMVA LLLSKQANGN LGNKSGLTPL HLVAQEGHVP VADVLIKHGV MVDATTRMGY TPLHVASHYG NIKLVKFL L QHQADVNAKT KLGYSPLHQA AQQGHTDIVT LLLKNGASPN EVSSDGTTPL AIAKRLGYIS VTDVLKVVTD ETSFVLVSD KHRMSFPETV DEILDVSEDE GEELISFKAE RRDSRDVDEE KELLDFVPKL DQVVESPAIP RIPCAMPETV VIRSEEQEQA SKEYDEDSL IPSSPATETS DNISPVASPV HTGFLVSFMV DARGGSMRGS RHNGLRVVIP PRTCAAPTRI TCRLVKPQKL S TPPPLAEE EGLASRIIAL GPTGAQFLSP VIVEIPHFAS HGRGDRELVV LRSENGSVWK EHRSRYGESY LDQILNGMDE EL GSLEELE KKRVCRIITT DFPLYFVIMS RLCQDYDTIG PEGGSLKSKL VPLVQATFPE NAVTKRVKLA LQAQPVPDEL VTK LLGNQA TFSPIVTVEP RRRKFHRPIG LRIPLPPSWT DNPRDSGEGD TTSLRLLCSV IGGTDQAQWE DITGTTKLVY ANEC ANFTT NVSARFWLSD CPRTAEAVNF ATLLYKELTA VPYMAKFVIF AKMNDPREGR LRCYCMTDDK VDKTLEQHEN FVEVA RSRD IEVLEGMSLF AELSGNLVPV KKAAQQRSFH FQSFRENRLA MPVKVRDSSR EPGGSLSFLR KAMKYEDTQH ILCHLN ITM PPCAKGSGAE DRRRTPTPLA LRYSILSEST PGSLSGTEQA EMKMAVISEH LGLSWAELAR ELQFSVEDIN RIRVENP NS LLEQSVALLN LWVIREGQNA NMENLYTALQ SIDRGEIVNM LEGSGRQSRN LKPDRRHTDR DYSLSPSQMN GYSSLQDE L LSPASLGCAL SSPLRADQYW NEVAVLDAIP LAATEHDTML EMSDMQVWSA GLTPSLVTAE DSSLECSKAE DSDATGHEW KLEGALSEEP RGPELGSLEL VEDDTVDSDA TNGLIDLLEQ EEGQRSEEKL PGSKRQDDAT GAGQDSENEV SLVSGHQRGQ ARITHSPTV SQVTERSQDR LQDWDADGSI VSYLQDAAQG SWQEEVTQGP HSFQGTSTMT EGLEPGGSQE YEKVLVSVSE H TWTEQPEA ESSQADRDRR QQGQEEQVQE AKNTFTQVVQ GNEFQNIPGE QVTEEQFTDE QGNIVTKKII RKVVRQIDLS SA DAAQEHE EVTVEGPLED PSELEVDIDY FMKHSKDHTS TPNP UniProtKB:  Ankyrin-1 Ankyrin-1 |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 8 mg/mL |
|---|---|
| Buffer | pH: 7.4 Details: Final gel filtration buffer contained 0.05% w/v digitonin, 130 mM KCl, 20 mM HEPES, pH 7.4, 1 mM ATP, 1 mM MgCl2, 1 mM PMSF. Peak fractions were concentrated to 8 mg/mL, and 0.01% w/v ...Details: Final gel filtration buffer contained 0.05% w/v digitonin, 130 mM KCl, 20 mM HEPES, pH 7.4, 1 mM ATP, 1 mM MgCl2, 1 mM PMSF. Peak fractions were concentrated to 8 mg/mL, and 0.01% w/v glycyrrhizic acid was added immediately prior to vitrification. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV / Details: 4-6 seconds, wait time 30 seconds.. |
| Details | Ankyrin complex mixture purified from digitonin-solubilized erythrocyte ghost membranes |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm |
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number grids imaged: 2 / Number real images: 14464 / Average exposure time: 2.5 sec. / Average electron dose: 58.0 e/Å2 / Details: Two grids were imaged in a single session. |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller








































 Z
Z Y
Y X
X