[English] 日本語
 Yorodumi
Yorodumi- EMDB-26767: The 2.19-angstrom CryoEM structure of the [NiFe]-hydrogenase Huc ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | The 2.19-angstrom CryoEM structure of the [NiFe]-hydrogenase Huc from Mycobacterium smegmatis - Complex minus stalk | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationhydrogenase (acceptor) / [Ni-Fe] hydrogenase complex / ferredoxin hydrogenase complex / hydrogenase (acceptor) activity / ferredoxin hydrogenase activity / anaerobic respiration / 3 iron, 4 sulfur cluster binding / nickel cation binding / 4 iron, 4 sulfur cluster binding / electron transfer activity ...hydrogenase (acceptor) / [Ni-Fe] hydrogenase complex / ferredoxin hydrogenase complex / hydrogenase (acceptor) activity / ferredoxin hydrogenase activity / anaerobic respiration / 3 iron, 4 sulfur cluster binding / nickel cation binding / 4 iron, 4 sulfur cluster binding / electron transfer activity / membrane / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Mycolicibacterium smegmatis (bacteria) / Mycolicibacterium smegmatis (bacteria) /  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.19 Å | |||||||||
 Authors Authors | Grinter R / Venugopal H / Kropp A / Greening C | |||||||||
| Funding support |  Australia, 2 items Australia, 2 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: Structural basis for bacterial energy extraction from atmospheric hydrogen. Authors: Rhys Grinter / Ashleigh Kropp / Hari Venugopal / Moritz Senger / Jack Badley / Princess R Cabotaje / Ruyu Jia / Zehui Duan / Ping Huang / Sven T Stripp / Christopher K Barlow / Matthew ...Authors: Rhys Grinter / Ashleigh Kropp / Hari Venugopal / Moritz Senger / Jack Badley / Princess R Cabotaje / Ruyu Jia / Zehui Duan / Ping Huang / Sven T Stripp / Christopher K Barlow / Matthew Belousoff / Hannah S Shafaat / Gregory M Cook / Ralf B Schittenhelm / Kylie A Vincent / Syma Khalid / Gustav Berggren / Chris Greening /       Abstract: Diverse aerobic bacteria use atmospheric H as an energy source for growth and survival. This globally significant process regulates the composition of the atmosphere, enhances soil biodiversity and ...Diverse aerobic bacteria use atmospheric H as an energy source for growth and survival. This globally significant process regulates the composition of the atmosphere, enhances soil biodiversity and drives primary production in extreme environments. Atmospheric H oxidation is attributed to uncharacterized members of the [NiFe] hydrogenase superfamily. However, it remains unresolved how these enzymes overcome the extraordinary catalytic challenge of oxidizing picomolar levels of H amid ambient levels of the catalytic poison O and how the derived electrons are transferred to the respiratory chain. Here we determined the cryo-electron microscopy structure of the Mycobacterium smegmatis hydrogenase Huc and investigated its mechanism. Huc is a highly efficient oxygen-insensitive enzyme that couples oxidation of atmospheric H to the hydrogenation of the respiratory electron carrier menaquinone. Huc uses narrow hydrophobic gas channels to selectively bind atmospheric H at the expense of O, and 3 [3Fe-4S] clusters modulate the properties of the enzyme so that atmospheric H oxidation is energetically feasible. The Huc catalytic subunits form an octameric 833 kDa complex around a membrane-associated stalk, which transports and reduces menaquinone 94 Å from the membrane. These findings provide a mechanistic basis for the biogeochemically and ecologically important process of atmospheric H oxidation, uncover a mode of energy coupling dependent on long-range quinone transport, and pave the way for the development of catalysts that oxidize H in ambient air. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26767.map.gz emd_26767.map.gz | 88.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26767-v30.xml emd-26767-v30.xml emd-26767.xml emd-26767.xml | 21.8 KB 21.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_26767_fsc.xml emd_26767_fsc.xml | 11.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_26767.png emd_26767.png | 209.2 KB | ||
| Others |  emd_26767_half_map_1.map.gz emd_26767_half_map_1.map.gz emd_26767_half_map_2.map.gz emd_26767_half_map_2.map.gz | 164.8 MB 164.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26767 http://ftp.pdbj.org/pub/emdb/structures/EMD-26767 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26767 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26767 | HTTPS FTP |
-Validation report
| Summary document |  emd_26767_validation.pdf.gz emd_26767_validation.pdf.gz | 883.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_26767_full_validation.pdf.gz emd_26767_full_validation.pdf.gz | 882.9 KB | Display | |
| Data in XML |  emd_26767_validation.xml.gz emd_26767_validation.xml.gz | 20.7 KB | Display | |
| Data in CIF |  emd_26767_validation.cif.gz emd_26767_validation.cif.gz | 26.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26767 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26767 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26767 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26767 | HTTPS FTP |
-Related structure data
| Related structure data |  7utdMC  7uurC  7uusC  8dqvC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26767.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26767.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.82 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_26767_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_26767_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of the type 2 [NiFe]-hydrogenase Huc from Mycobacterium s...
| Entire | Name: Complex of the type 2 [NiFe]-hydrogenase Huc from Mycobacterium smegmatis |
|---|---|
| Components |
|
-Supramolecule #1: Complex of the type 2 [NiFe]-hydrogenase Huc from Mycobacterium s...
| Supramolecule | Name: Complex of the type 2 [NiFe]-hydrogenase Huc from Mycobacterium smegmatis type: complex / ID: 1 / Chimera: Yes / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Mycolicibacterium smegmatis (bacteria) / Strain: MC2 155 Mycolicibacterium smegmatis (bacteria) / Strain: MC2 155 |
| Molecular weight | Theoretical: 833 KDa |
-Macromolecule #1: Hydrogenase-2, large subunit
| Macromolecule | Name: Hydrogenase-2, large subunit / type: protein_or_peptide / ID: 1 / Number of copies: 8 / Enantiomer: LEVO / EC number: hydrogenase (acceptor) |
|---|---|
| Source (natural) | Organism:  Mycolicibacterium smegmatis MC2 155 (bacteria) / Strain: ATCC 700084 / mc(2)155 Mycolicibacterium smegmatis MC2 155 (bacteria) / Strain: ATCC 700084 / mc(2)155 |
| Molecular weight | Theoretical: 57.217078 KDa |
| Sequence | String: LDLFVSPLGR VEGDLDVRVT INDGVVTSAW TEAAMFRGFE IILRGKDPQA GLIVCPRICG ICGGSHLYKS AYALDTAWRT HMPPNATLI RNICQACETL QSIPRYFYAL FAIDLTNKNY AKSKLYDEAV RRFAPYVGTS YQPGVVLSAK PVEVYAIFGG Q WP(DHI)SSFMV ...String: LDLFVSPLGR VEGDLDVRVT INDGVVTSAW TEAAMFRGFE IILRGKDPQA GLIVCPRICG ICGGSHLYKS AYALDTAWRT HMPPNATLI RNICQACETL QSIPRYFYAL FAIDLTNKNY AKSKLYDEAV RRFAPYVGTS YQPGVVLSAK PVEVYAIFGG Q WP(DHI)SSFMV PGGVMSAPTL SDVTRAIAIL EHWNDNWLEK QWLGCSVDRW LENKTWNDVL AWVDENESQY NSDCGFFI R YCLDVGLDKY GQGVGNYLAT GTYFEPSLYE NPTIEGRNAA LIGRSGVFAD GRYFEFDQAN VTEDVTHSFY EGNRPLHPF EGETIPVNPE DGRRQGKYSW AKSPRYAVPG LGNVPLETGP LARRMAASAP DAETHQDDDP LFADIYNAIG PSVMVRQLAR MHEGPKYYK WVRQWLDDLE LKESFYTKPV EYAEGKGFGS TEAARGALSD WIVIEDSKIK NYQVVTPTAW NIGPRDASEV L GPIEQALV GSPIVDAEDP VELGHVARSF DSCLVCTVH |
-Macromolecule #2: Hydrogenase-2, small subunit
| Macromolecule | Name: Hydrogenase-2, small subunit / type: protein_or_peptide / ID: 2 / Number of copies: 8 / Enantiomer: LEVO / EC number: hydrogenase (acceptor) |
|---|---|
| Source (natural) | Organism:  Mycolicibacterium smegmatis MC2 155 (bacteria) / Strain: ATCC 700084 / mc(2)155 Mycolicibacterium smegmatis MC2 155 (bacteria) / Strain: ATCC 700084 / mc(2)155 |
| Molecular weight | Theoretical: 39.65977 KDa |
| Sequence | String: MSAWSHPQFE KGGGSGGGSG GSAWSHPQFE KSGGGGGENL YFQGSGGASV LWFQGGACSG NTMSFLNADE PNVVDLIVDF GLDLLWHPS LGLELGNNAQ KVFWDCAKGE RPLDIFVFEG TVIEAPNGTG QMDMFAGRPM KDWVTDLAGA AQIVVAIGDC A CFGGIPAM ...String: MSAWSHPQFE KGGGSGGGSG GSAWSHPQFE KSGGGGGENL YFQGSGGASV LWFQGGACSG NTMSFLNADE PNVVDLIVDF GLDLLWHPS LGLELGNNAQ KVFWDCAKGE RPLDIFVFEG TVIEAPNGTG QMDMFAGRPM KDWVTDLAGA AQIVVAIGDC A CFGGIPAM EPNPSGSTGL QFHKREKGGF LGPDFRSKMG LPVINVPGCP AHPDWITQIL VALATGRAGD ITLDDLHRPE TF FKTFTQT GCTRVQFFEY KQSTLSFGEG TRTGCLFYEF GCRGPMTHSP CNRILWNRQS SKTRAGMPCL GCTEPEFPHF DLA PGTVFK TQKVSGMIPK EVPEGTDHLT YMGLAAAARI AAPQWSKEDM FVV |
-Macromolecule #3: Type 2 [NiFe]-hydrogenase Huc membrane adapter subunit
| Macromolecule | Name: Type 2 [NiFe]-hydrogenase Huc membrane adapter subunit type: protein_or_peptide / ID: 3 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Mycolicibacterium smegmatis MC2 155 (bacteria) / Strain: MC2 155 Mycolicibacterium smegmatis MC2 155 (bacteria) / Strain: MC2 155 |
| Molecular weight | Theoretical: 6.416264 KDa |
| Sequence | String: SPVDGIRRRL DDPQVAEALN SLLDHADLLA VLVKGLDGFV RRGDDIANNL TSAIGELKAL |
-Macromolecule #4: NICKEL (III) ION
| Macromolecule | Name: NICKEL (III) ION / type: ligand / ID: 4 / Number of copies: 8 / Formula: 3NI |
|---|---|
| Molecular weight | Theoretical: 58.693 Da |
| Chemical component information |  ChemComp-3NI: |
-Macromolecule #5: CARBONMONOXIDE-(DICYANO) IRON
| Macromolecule | Name: CARBONMONOXIDE-(DICYANO) IRON / type: ligand / ID: 5 / Number of copies: 8 / Formula: FCO |
|---|---|
| Molecular weight | Theoretical: 135.89 Da |
| Chemical component information | 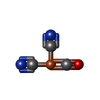 ChemComp-FCO: |
-Macromolecule #6: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 6 / Number of copies: 8 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #7: MENAQUINONE-9
| Macromolecule | Name: MENAQUINONE-9 / type: ligand / ID: 7 / Number of copies: 8 / Formula: MQ9 |
|---|---|
| Molecular weight | Theoretical: 785.233 Da |
| Chemical component information | 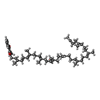 ChemComp-MQ9: |
-Macromolecule #8: FE3-S4 CLUSTER
| Macromolecule | Name: FE3-S4 CLUSTER / type: ligand / ID: 8 / Number of copies: 24 / Formula: F3S |
|---|---|
| Molecular weight | Theoretical: 295.795 Da |
| Chemical component information |  ChemComp-F3S: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 5.0 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.9 Component:
Details: pH 7.9 | |||||||||
| Grid | Model: Quantifoil / Material: GOLD / Support film - Material: GOLD / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295 K / Instrument: FEI VITROBOT MARK III |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number real images: 3113 / Average exposure time: 6.0 sec. / Average electron dose: 66.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Cooling holder cryogen: HELIUM |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: BACKBONE TRACE / Target criteria: Correlation coefficient |
|---|---|
| Output model |  PDB-7utd: |
 Movie
Movie Controller
Controller



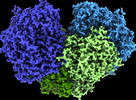

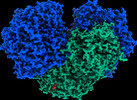


 Z
Z Y
Y X
X


















