+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
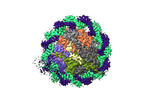
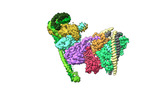
| Title | Structure of the human CCAN CENP-A alpha-satellite complex | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
| Function / homology |  Function and homology information Function and homology informationspindle attachment to meiosis I kinetochore / inner kinetochore / centromeric DNA binding / CENP-A containing chromatin assembly / protein localization to chromosome, centromeric region / attachment of mitotic spindle microtubules to kinetochore /  kinetochore assembly / condensed chromosome, centromeric region / establishment of mitotic spindle orientation / mitotic cytokinesis ...spindle attachment to meiosis I kinetochore / inner kinetochore / centromeric DNA binding / CENP-A containing chromatin assembly / protein localization to chromosome, centromeric region / attachment of mitotic spindle microtubules to kinetochore / kinetochore assembly / condensed chromosome, centromeric region / establishment of mitotic spindle orientation / mitotic cytokinesis ...spindle attachment to meiosis I kinetochore / inner kinetochore / centromeric DNA binding / CENP-A containing chromatin assembly / protein localization to chromosome, centromeric region / attachment of mitotic spindle microtubules to kinetochore /  kinetochore assembly / condensed chromosome, centromeric region / establishment of mitotic spindle orientation / mitotic cytokinesis / kinetochore assembly / condensed chromosome, centromeric region / establishment of mitotic spindle orientation / mitotic cytokinesis /  chromosome, centromeric region / negative regulation of megakaryocyte differentiation / protein localization to CENP-A containing chromatin / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / pericentric heterochromatin / Replacement of protamines by nucleosomes in the male pronucleus / CENP-A containing nucleosome / Packaging Of Telomere Ends / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Recognition and association of DNA glycosylase with site containing an affected purine / Cleavage of the damaged purine / Deposition of new CENPA-containing nucleosomes at the centromere / Resolution of Sister Chromatid Cohesion / Recognition and association of DNA glycosylase with site containing an affected pyrimidine / Cleavage of the damaged pyrimidine / Inhibition of DNA recombination at telomere / Meiotic synapsis / telomere organization / RNA Polymerase I Promoter Opening / SUMOylation of chromatin organization proteins / Assembly of the ORC complex at the origin of replication / chromosome, centromeric region / negative regulation of megakaryocyte differentiation / protein localization to CENP-A containing chromatin / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / pericentric heterochromatin / Replacement of protamines by nucleosomes in the male pronucleus / CENP-A containing nucleosome / Packaging Of Telomere Ends / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Recognition and association of DNA glycosylase with site containing an affected purine / Cleavage of the damaged purine / Deposition of new CENPA-containing nucleosomes at the centromere / Resolution of Sister Chromatid Cohesion / Recognition and association of DNA glycosylase with site containing an affected pyrimidine / Cleavage of the damaged pyrimidine / Inhibition of DNA recombination at telomere / Meiotic synapsis / telomere organization / RNA Polymerase I Promoter Opening / SUMOylation of chromatin organization proteins / Assembly of the ORC complex at the origin of replication /  DNA methylation / Condensation of Prophase Chromosomes / HCMV Late Events / Chromatin modifications during the maternal to zygotic transition (MZT) / ERCC6 (CSB) and EHMT2 (G9a) positively regulate rRNA expression / SIRT1 negatively regulates rRNA expression / DNA methylation / Condensation of Prophase Chromosomes / HCMV Late Events / Chromatin modifications during the maternal to zygotic transition (MZT) / ERCC6 (CSB) and EHMT2 (G9a) positively regulate rRNA expression / SIRT1 negatively regulates rRNA expression /  innate immune response in mucosa / PRC2 methylates histones and DNA / Defective pyroptosis / innate immune response in mucosa / PRC2 methylates histones and DNA / Defective pyroptosis /  chromosome segregation / RHO GTPases Activate Formins / HDACs deacetylate histones / RNA Polymerase I Promoter Escape / Nonhomologous End-Joining (NHEJ) / Transcriptional regulation by small RNAs / Formation of the beta-catenin:TCF transactivating complex / RUNX1 regulates genes involved in megakaryocyte differentiation and platelet function / Activated PKN1 stimulates transcription of AR (androgen receptor) regulated genes KLK2 and KLK3 / NoRC negatively regulates rRNA expression / G2/M DNA damage checkpoint / B-WICH complex positively regulates rRNA expression / HDMs demethylate histones / DNA Damage/Telomere Stress Induced Senescence / Metalloprotease DUBs / chromosome segregation / RHO GTPases Activate Formins / HDACs deacetylate histones / RNA Polymerase I Promoter Escape / Nonhomologous End-Joining (NHEJ) / Transcriptional regulation by small RNAs / Formation of the beta-catenin:TCF transactivating complex / RUNX1 regulates genes involved in megakaryocyte differentiation and platelet function / Activated PKN1 stimulates transcription of AR (androgen receptor) regulated genes KLK2 and KLK3 / NoRC negatively regulates rRNA expression / G2/M DNA damage checkpoint / B-WICH complex positively regulates rRNA expression / HDMs demethylate histones / DNA Damage/Telomere Stress Induced Senescence / Metalloprotease DUBs /  kinetochore / PKMTs methylate histone lysines / RMTs methylate histone arginines / kinetochore / PKMTs methylate histone lysines / RMTs methylate histone arginines /  Meiotic recombination / Pre-NOTCH Transcription and Translation / Meiotic recombination / Pre-NOTCH Transcription and Translation /  nucleosome assembly / Activation of anterior HOX genes in hindbrain development during early embryogenesis / HCMV Early Events / Transcriptional regulation of granulopoiesis / structural constituent of chromatin / Separation of Sister Chromatids / UCH proteinases / nucleosome assembly / Activation of anterior HOX genes in hindbrain development during early embryogenesis / HCMV Early Events / Transcriptional regulation of granulopoiesis / structural constituent of chromatin / Separation of Sister Chromatids / UCH proteinases /  nucleosome / antimicrobial humoral immune response mediated by antimicrobial peptide / E3 ubiquitin ligases ubiquitinate target proteins / mitotic cell cycle / Recruitment and ATM-mediated phosphorylation of repair and signaling proteins at DNA double strand breaks / RUNX1 regulates transcription of genes involved in differentiation of HSCs / chromatin organization / Processing of DNA double-strand break ends / HATs acetylate histones / midbody / antibacterial humoral response / Senescence-Associated Secretory Phenotype (SASP) / Oxidative Stress Induced Senescence / Estrogen-dependent gene expression / nucleosome / antimicrobial humoral immune response mediated by antimicrobial peptide / E3 ubiquitin ligases ubiquitinate target proteins / mitotic cell cycle / Recruitment and ATM-mediated phosphorylation of repair and signaling proteins at DNA double strand breaks / RUNX1 regulates transcription of genes involved in differentiation of HSCs / chromatin organization / Processing of DNA double-strand break ends / HATs acetylate histones / midbody / antibacterial humoral response / Senescence-Associated Secretory Phenotype (SASP) / Oxidative Stress Induced Senescence / Estrogen-dependent gene expression /  chromosome, telomeric region / chromosome, telomeric region /  nuclear body / Ub-specific processing proteases / defense response to Gram-positive bacterium / Amyloid fiber formation / protein heterodimerization activity / nuclear body / Ub-specific processing proteases / defense response to Gram-positive bacterium / Amyloid fiber formation / protein heterodimerization activity /  cell division / negative regulation of cell population proliferation / cell division / negative regulation of cell population proliferation /  chromatin binding / protein-containing complex / chromatin binding / protein-containing complex /  DNA binding / DNA binding /  extracellular space / extracellular space /  RNA binding / extracellular exosome / extracellular region / RNA binding / extracellular exosome / extracellular region /  nucleoplasm / nucleoplasm /  membrane / identical protein binding membrane / identical protein bindingSimilarity search - Function | ||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.68 Å cryo EM / Resolution: 2.68 Å | ||||||||||||
 Authors Authors | Yatskevich S / Muir KW / Bellini D / Barford D | ||||||||||||
| Funding support |  Germany, 3 items Germany, 3 items
| ||||||||||||
 Citation Citation |  Journal: Science / Year: 2022 Journal: Science / Year: 2022Title: Structure of the human inner kinetochore bound to a centromeric CENP-A nucleosome. Authors: Stanislau Yatskevich / Kyle W Muir / Dom Bellini / Ziguo Zhang / Jing Yang / Thomas Tischer / Masa Predin / Tom Dendooven / Stephen H McLaughlin / David Barford /  Abstract: Kinetochores assemble onto specialized centromeric CENP-A (centromere protein A) nucleosomes (CENP-A) to mediate attachments between chromosomes and the mitotic spindle. We describe cryo-electron ...Kinetochores assemble onto specialized centromeric CENP-A (centromere protein A) nucleosomes (CENP-A) to mediate attachments between chromosomes and the mitotic spindle. We describe cryo-electron microscopy structures of the human inner kinetochore constitutive centromere associated network (CCAN) complex bound to CENP-A reconstituted onto α-satellite DNA. CCAN forms edge-on contacts with CENP-A, and a linker DNA segment of the α-satellite repeat emerges from the fully wrapped end of the nucleosome to thread through the central CENP-LN channel that tightly grips the DNA. The CENP-TWSX histone-fold module further augments DNA binding and partially wraps the linker DNA in a manner reminiscent of canonical nucleosomes. Our study suggests that the topological entrapment of the linker DNA by CCAN provides a robust mechanism by which kinetochores withstand both pushing and pulling forces exerted by the mitotic spindle. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13437.map.gz emd_13437.map.gz | 14.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13437-v30.xml emd-13437-v30.xml emd-13437.xml emd-13437.xml | 18.8 KB 18.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_13437_fsc.xml emd_13437_fsc.xml | 13.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_13437.png emd_13437.png | 81.2 KB | ||
| Others |  emd_13437_additional_1.map.gz emd_13437_additional_1.map.gz | 170.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13437 http://ftp.pdbj.org/pub/emdb/structures/EMD-13437 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13437 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13437 | HTTPS FTP |
-Related structure data
| Related structure data |  7piiMC  7pb4C  7pb8C  7pknC 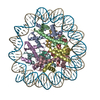 7r5rC  7r5sC  7r5vC  7ywxC  7yyhC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_13437.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13437.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.831 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_13437_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : CENP-A nucleosome in complex with CENP-C
| Entire | Name: CENP-A nucleosome in complex with CENP-C |
|---|---|
| Components |
|
-Supramolecule #1: CENP-A nucleosome in complex with CENP-C
| Supramolecule | Name: CENP-A nucleosome in complex with CENP-C / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#7 |
|---|
-Macromolecule #1: Histone H3-like centromeric protein A
| Macromolecule | Name: Histone H3-like centromeric protein A / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 16.02363 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MGPRRRSRKP EAPRRRSPSP TPTPGPSRRG PSLGASSHQH SRRRQGWLKE IRKLQKSTHL LIRKLPFSRL AREICVKFTR GVDFNWQAQ ALLALQEAAE AFLVHLFEDA YLLTLHAGRV TLFPKDVQLA RRIRGLEEGL G |
-Macromolecule #2: Histone H4
| Macromolecule | Name: Histone H4 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 11.394426 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MSGRGKGGKG LGKGGAKRHR KVLRDNIQGI TKPAIRRLAR RGGVKRISGL IYEETRGVLK VFLENVIRDA VTYTEHAKRK TVTAMDVVY ALKRQGRTLY GFGG |
-Macromolecule #3: Histone H2A type 1-C
| Macromolecule | Name: Histone H2A type 1-C / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 16.666334 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MGSSHHHHHH SPGLEVLFQG PRGMSGRGKQ GGKARAKAKS RSSRAGLQFP VGRVHRLLRK GNYAERVGAG APVYLAAVLE YLTAEILEL AGNAARDNKK TRIIPRHLQL AIRNDEELNK LLGRVTIAQG GVLPNIQAVL LPKKTESHHK AKGK |
-Macromolecule #4: Histone H2B type 1-C/E/F/G/I
| Macromolecule | Name: Histone H2B type 1-C/E/F/G/I / type: protein_or_peptide / ID: 4 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 13.937213 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MPEPAKSAPA PKKGSKKAVT KAQKKDGKKR KRSRKESYSV YVYKVLKQVH PDTGISSKAM GIMNSFVNDI FERIAGEASR LAHYNKRST ITSREIQTAV RLLLPGELAK HAVSEGTKAV TKYTSSK |
-Macromolecule #7: Centromere protein C
| Macromolecule | Name: Centromere protein C / type: protein_or_peptide / ID: 7 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 61.943082 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MAASGLDHLK NGYRRRFCRP SRARDINTEQ GQNVLEILQD CFEEKSLAND FSTNSTKSVP NSTRKIKDTC IQSPSKECQK SHPKSVPVS SKKKEASLQF VVEPSEATNR SVQAHEVHQK ILATDVSSKN TPDSKKISSR NINDHHSEAD EEFYLSVGSP S VLLDAKTS ...String: MAASGLDHLK NGYRRRFCRP SRARDINTEQ GQNVLEILQD CFEEKSLAND FSTNSTKSVP NSTRKIKDTC IQSPSKECQK SHPKSVPVS SKKKEASLQF VVEPSEATNR SVQAHEVHQK ILATDVSSKN TPDSKKISSR NINDHHSEAD EEFYLSVGSP S VLLDAKTS VSQNVIPSSA QKRETYTFEN SVNMLPSSTE VSVKTKKRLN FDDKVMLKKI EIDNKVSDEE DKTSEGQERK PS GSSQNRI RDSEYEIQRQ AKKSFSTLFL ETVKRKSESS PIVRHAATAP PHSCPPDDTK LIEDEFIIDE SDQSFASRSW ITI PRKAGS LKQRTISPAE STALLQGRKS REKHHNILPK TLANDKHSHK PHPVETSQPS DKTVLDTSYA LIGETVNNYR STKY EMYSK NAEKPSRSKR TIKQKQRRKF MAKPAEEQLD VGQSKDENIH TSHITQDEFQ RNSDRNMEEH EEMGNDCVSK KQMPP VGSK KSSTRKDKEE SKKKRFSSES KNKLVPEEVT STVTKSRRIS RRPSDWWVVK SEESPVYSNS S |
-Macromolecule #5: DNA (122-MER)
| Macromolecule | Name: DNA (122-MER) / type: dna / ID: 5 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 52.747801 KDa |
| Sequence | String: (DC)(DT)(DA)(DC)(DA)(DA)(DA)(DA)(DA)(DG) (DA)(DG)(DT)(DG)(DT)(DT)(DT)(DC)(DA)(DA) (DA)(DA)(DC)(DT)(DG)(DC)(DT)(DC)(DT) (DA)(DT)(DC)(DA)(DA)(DA)(DA)(DG)(DG)(DA) (DA) (DT)(DG)(DT)(DT)(DC)(DA) ...String: (DC)(DT)(DA)(DC)(DA)(DA)(DA)(DA)(DA)(DG) (DA)(DG)(DT)(DG)(DT)(DT)(DT)(DC)(DA)(DA) (DA)(DA)(DC)(DT)(DG)(DC)(DT)(DC)(DT) (DA)(DT)(DC)(DA)(DA)(DA)(DA)(DG)(DG)(DA) (DA) (DT)(DG)(DT)(DT)(DC)(DA)(DA)(DC) (DT)(DC)(DT)(DG)(DT)(DG)(DA)(DG)(DT)(DT) (DG)(DA) (DA)(DT)(DG)(DC)(DA)(DA)(DT) (DC)(DA)(DT)(DC)(DA)(DC)(DA)(DA)(DA)(DG) (DA)(DA)(DG) (DT)(DT)(DT)(DC)(DT)(DG) (DA)(DG)(DA)(DA)(DT)(DG)(DC)(DT)(DT)(DC) (DT)(DG)(DT)(DT) (DT)(DA)(DG)(DT)(DT) (DT)(DT)(DT)(DA)(DT)(DG)(DT)(DG)(DA)(DA) (DG)(DA)(DT)(DA)(DT) (DT)(DC)(DC)(DC) (DG)(DT)(DT)(DT)(DC)(DC)(DA)(DA)(DC)(DG) (DA)(DA)(DG)(DG)(DC)(DC) (DT)(DC)(DA) (DA)(DA)(DG)(DC)(DG)(DG)(DT)(DC)(DC)(DA) (DA)(DA)(DT)(DA)(DT)(DC)(DC) (DA)(DC) (DT)(DT)(DG)(DC)(DA)(DG)(DA)(DT)(DT) |
-Macromolecule #6: DNA (123-MER)
| Macromolecule | Name: DNA (123-MER) / type: dna / ID: 6 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 52.800816 KDa |
| Sequence | String: (DA)(DA)(DT)(DC)(DT)(DG)(DC)(DA)(DA)(DG) (DT)(DG)(DG)(DA)(DT)(DA)(DT)(DT)(DT)(DG) (DG)(DA)(DC)(DC)(DG)(DC)(DT)(DT)(DT) (DG)(DA)(DG)(DG)(DC)(DC)(DT)(DT)(DC)(DG) (DT) (DT)(DG)(DG)(DA)(DA)(DA) ...String: (DA)(DA)(DT)(DC)(DT)(DG)(DC)(DA)(DA)(DG) (DT)(DG)(DG)(DA)(DT)(DA)(DT)(DT)(DT)(DG) (DG)(DA)(DC)(DC)(DG)(DC)(DT)(DT)(DT) (DG)(DA)(DG)(DG)(DC)(DC)(DT)(DT)(DC)(DG) (DT) (DT)(DG)(DG)(DA)(DA)(DA)(DC)(DG) (DG)(DG)(DA)(DA)(DT)(DA)(DT)(DC)(DT)(DT) (DC)(DA) (DC)(DA)(DT)(DA)(DA)(DA)(DA) (DA)(DC)(DT)(DA)(DA)(DA)(DC)(DA)(DG)(DA) (DA)(DG)(DC) (DA)(DT)(DT)(DC)(DT)(DC) (DA)(DG)(DA)(DA)(DA)(DC)(DT)(DT)(DC)(DT) (DT)(DT)(DG)(DT) (DG)(DA)(DT)(DG)(DA) (DT)(DT)(DG)(DC)(DA)(DT)(DT)(DC)(DA)(DA) (DC)(DT)(DC)(DA)(DC) (DA)(DG)(DA)(DG) (DT)(DT)(DG)(DA)(DA)(DC)(DA)(DT)(DT)(DC) (DC)(DT)(DT)(DT)(DT)(DG) (DA)(DT)(DA) (DG)(DA)(DG)(DC)(DA)(DG)(DT)(DT)(DT)(DT) (DG)(DA)(DA)(DA)(DC)(DA)(DC) (DT)(DC) (DT)(DT)(DT)(DT)(DT)(DG)(DT)(DA)(DG) |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy Bright-field microscopy |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




















 Z
Z Y
Y X
X