[English] 日本語
 Yorodumi
Yorodumi- PDB-8dw2: Cryo-EM structure of SARS-CoV-2 RBD in complex with anti-SARS-CoV... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8dw2 | ||||||
|---|---|---|---|---|---|---|---|
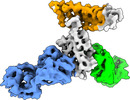
| Title | Cryo-EM structure of SARS-CoV-2 RBD in complex with anti-SARS-CoV-2 DARPin,SR22, and two antibody Fabs, S309 and CR3022 | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  VIRAL PROTEIN/IMMUNE SYSTEM / DARPins / Anti-SARS-CoV-2 / VIRAL PROTEIN/IMMUNE SYSTEM / DARPins / Anti-SARS-CoV-2 /  therapeutics / therapeutics /  COVID-19 / COVID-19 /  VIRAL PROTEIN-IMMUNE SYSTEM complex VIRAL PROTEIN-IMMUNE SYSTEM complex | ||||||
| Function / homology |  Function and homology information Function and homology informationMaturation of spike protein / viral translation / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular space / suppression by virus of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / entry receptor-mediated virion attachment to host cell ...Maturation of spike protein / viral translation / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular space / suppression by virus of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / entry receptor-mediated virion attachment to host cell / host cell endoplasmic reticulum-Golgi intermediate compartment membrane / receptor-mediated endocytosis of virus by host cell / Attachment and Entry /  membrane fusion / positive regulation of viral entry into host cell / receptor-mediated virion attachment to host cell / membrane fusion / positive regulation of viral entry into host cell / receptor-mediated virion attachment to host cell /  receptor ligand activity / host cell surface receptor binding / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / receptor ligand activity / host cell surface receptor binding / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane /  viral envelope / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / virion attachment to host cell / SARS-CoV-2 activates/modulates innate and adaptive immune responses / host cell plasma membrane / virion membrane / viral envelope / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / virion attachment to host cell / SARS-CoV-2 activates/modulates innate and adaptive immune responses / host cell plasma membrane / virion membrane /  membrane / identical protein binding / membrane / identical protein binding /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Severe acute respiratory syndrome coronavirus 2 Severe acute respiratory syndrome coronavirus 2  Homo sapiens (human) Homo sapiens (human) Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) | ||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 4.11 Å cryo EM / Resolution: 4.11 Å | ||||||
 Authors Authors | Kwon, Y.D. / Gorman, J. / Kwong, P.D. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nat Chem Biol / Year: 2023 Journal: Nat Chem Biol / Year: 2023Title: A potent and broad neutralization of SARS-CoV-2 variants of concern by DARPins. Authors: Vikas Chonira / Young D Kwon / Jason Gorman / James Brett Case / Zhiqiang Ku / Rudo Simeon / Ryan G Casner / Darcy R Harris / Adam S Olia / Tyler Stephens / Lawrence Shapiro / Michael F ...Authors: Vikas Chonira / Young D Kwon / Jason Gorman / James Brett Case / Zhiqiang Ku / Rudo Simeon / Ryan G Casner / Darcy R Harris / Adam S Olia / Tyler Stephens / Lawrence Shapiro / Michael F Bender / Hannah Boyd / I-Ting Teng / Yaroslav Tsybovsky / Florian Krammer / Ningyan Zhang / Michael S Diamond / Peter D Kwong / Zhiqiang An / Zhilei Chen /  Abstract: We report the engineering and selection of two synthetic proteins-FSR16m and FSR22-for the possible treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. FSR16m and ...We report the engineering and selection of two synthetic proteins-FSR16m and FSR22-for the possible treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. FSR16m and FSR22 are trimeric proteins composed of DARPin SR16m or SR22 fused with a T4 foldon. Despite selection by a spike protein from a now historical SARS-CoV-2 strain, FSR16m and FSR22 exhibit broad-spectrum neutralization of SARS-CoV-2 strains, inhibiting authentic B.1.351, B.1.617.2 and BA.1.1 viruses, with respective IC values of 3.4, 2.2 and 7.4 ng ml for FSR16m. Cryo-EM structures revealed that these DARPins recognize a region of the receptor-binding domain (residues 456, 475, 486, 487 and 489) overlapping a critical portion of the angiotensin-converting enzyme 2 (ACE2)-binding surface. K18-hACE2 transgenic mice inoculated with B.1.617.2 and receiving intranasally administered FSR16m showed less weight loss and 10-100-fold lower viral burden in upper and lower respiratory tracts. The strong and broad neutralization potency makes FSR16m and FSR22 promising candidates for the prevention and treatment of infection by SARS-CoV-2. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8dw2.cif.gz 8dw2.cif.gz | 218 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8dw2.ent.gz pdb8dw2.ent.gz | 174.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8dw2.json.gz 8dw2.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dw/8dw2 https://data.pdbj.org/pub/pdb/validation_reports/dw/8dw2 ftp://data.pdbj.org/pub/pdb/validation_reports/dw/8dw2 ftp://data.pdbj.org/pub/pdb/validation_reports/dw/8dw2 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 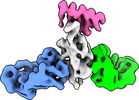 27749MC  7tyzC  7tz0C  8dw3C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 2 types, 2 molecules AB
| #1: Protein | Mass: 16428.541 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)  Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) |
|---|---|
| #2: Protein | Mass: 22100.758 Da / Num. of mol.: 1 / Fragment: receptor binding domain (UNP residues 330-526) Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Severe acute respiratory syndrome coronavirus 2 Severe acute respiratory syndrome coronavirus 2Gene: S, 2 / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host:   Homo sapiens (human) / References: UniProt: P0DTC2 Homo sapiens (human) / References: UniProt: P0DTC2 |
-Antibody , 4 types, 4 molecules CDHL
| #3: Antibody | Mass: 23204.697 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host: Homo sapiens (human) / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host:   Homo sapiens (human) Homo sapiens (human) |
|---|---|
| #4: Antibody | Mass: 24573.471 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host: Homo sapiens (human) / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host:   Homo sapiens (human) Homo sapiens (human) |
| #5: Antibody | Mass: 23382.369 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host: Homo sapiens (human) / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host:   Homo sapiens (human) Homo sapiens (human) |
| #6: Antibody | Mass: 24289.885 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host: Homo sapiens (human) / Plasmid: pVRC8400 / Cell line (production host): 293 Freestyle / Production host:   Homo sapiens (human) Homo sapiens (human) |
-Sugars , 1 types, 1 molecules
| #7: Polysaccharide | beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta- ...beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose / Mass: 586.542 Da / Num. of mol.: 1 / Mass: 586.542 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: DARPin SR22 in complex with SARS-CoV-2 RBD and antibody Fabs, S309 and CR3022 Type: COMPLEX Details: DARPin SR22 in complex with SARS-CoV-2 RBD and antibody Fabs, S309 and CR3022 Entity ID: #1-#6 / Source: MULTIPLE SOURCES |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:   Severe acute respiratory syndrome coronavirus 2 Severe acute respiratory syndrome coronavirus 2 |
| Source (recombinant) | Organism:   Homo sapiens (human) / Cell: Freestyle 293 / Plasmid Homo sapiens (human) / Cell: Freestyle 293 / Plasmid : pVRC8400 : pVRC8400 |
| Buffer solution | pH: 7.4 / Details: 10 mM HEPES, 7.4, 150 mM NaCl |
| Specimen | Conc.: 1 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES |
Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 95 % |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 2500 nm / Nominal defocus min: 1250 nm / Cs Bright-field microscopy / Nominal defocus max: 2500 nm / Nominal defocus min: 1250 nm / Cs : 2.7 mm / C2 aperture diameter: 100 µm : 2.7 mm / C2 aperture diameter: 100 µm |
| Image recording | Electron dose: 40.2 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.20.1_4487: / Classification: refinement | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 10327040 | ||||||||||||||||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | ||||||||||||||||||||||||||||
3D reconstruction | Resolution: 4.11 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 101417 / Symmetry type: POINT | ||||||||||||||||||||||||||||
| Atomic model building | B value: 134 / Protocol: RIGID BODY FIT / Space: REAL / Target criteria: CC | ||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 7BNO | ||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller



 PDBj
PDBj





