[English] 日本語
 Yorodumi
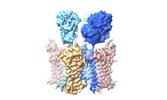
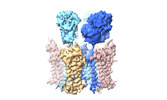
Yorodumi- PDB-8c1p: Active state homomeric GluA1 AMPA receptor in complex with TARP g... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8c1p | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Active state homomeric GluA1 AMPA receptor in complex with TARP gamma 3 | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords |  MEMBRANE PROTEIN / MEMBRANE PROTEIN /  AMPAR / AMPAR /  ion channels / ion channels /  neurotransmission neurotransmission | |||||||||
| Function / homology |  Function and homology information Function and homology informationCargo concentration in the ER / cellular response to amine stimulus / axonal spine / COPII-mediated vesicle transport / positive regulation of membrane potential / cellular response to ammonium ion / neurotransmitter receptor transport, postsynaptic endosome to lysosome / neurotransmitter receptor activity involved in regulation of postsynaptic cytosolic calcium ion concentration / LGI-ADAM interactions /  myosin V binding ...Cargo concentration in the ER / cellular response to amine stimulus / axonal spine / COPII-mediated vesicle transport / positive regulation of membrane potential / cellular response to ammonium ion / neurotransmitter receptor transport, postsynaptic endosome to lysosome / neurotransmitter receptor activity involved in regulation of postsynaptic cytosolic calcium ion concentration / LGI-ADAM interactions / myosin V binding ...Cargo concentration in the ER / cellular response to amine stimulus / axonal spine / COPII-mediated vesicle transport / positive regulation of membrane potential / cellular response to ammonium ion / neurotransmitter receptor transport, postsynaptic endosome to lysosome / neurotransmitter receptor activity involved in regulation of postsynaptic cytosolic calcium ion concentration / LGI-ADAM interactions /  myosin V binding / neuron spine / Trafficking of AMPA receptors / myosin V binding / neuron spine / Trafficking of AMPA receptors /  regulation of AMPA receptor activity / neurotransmitter receptor internalization / channel regulator activity / response to arsenic-containing substance / cellular response to dsRNA / dendritic spine membrane / postsynaptic neurotransmitter receptor diffusion trapping / Synaptic adhesion-like molecules / glutamate-gated calcium ion channel activity / neurotransmitter receptor localization to postsynaptic specialization membrane / long-term synaptic depression / cellular response to peptide hormone stimulus / regulation of AMPA receptor activity / neurotransmitter receptor internalization / channel regulator activity / response to arsenic-containing substance / cellular response to dsRNA / dendritic spine membrane / postsynaptic neurotransmitter receptor diffusion trapping / Synaptic adhesion-like molecules / glutamate-gated calcium ion channel activity / neurotransmitter receptor localization to postsynaptic specialization membrane / long-term synaptic depression / cellular response to peptide hormone stimulus /  beta-2 adrenergic receptor binding / beta-2 adrenergic receptor binding /  protein kinase A binding / neuronal cell body membrane / spinal cord development / Activation of AMPA receptors / perisynaptic space / transmission of nerve impulse / AMPA glutamate receptor activity / Trafficking of GluR2-containing AMPA receptors / response to lithium ion / protein kinase A binding / neuronal cell body membrane / spinal cord development / Activation of AMPA receptors / perisynaptic space / transmission of nerve impulse / AMPA glutamate receptor activity / Trafficking of GluR2-containing AMPA receptors / response to lithium ion /  immunoglobulin binding / AMPA glutamate receptor complex / immunoglobulin binding / AMPA glutamate receptor complex /  adenylate cyclase binding / adenylate cyclase binding /  excitatory synapse / cellular response to organic cyclic compound / excitatory synapse / cellular response to organic cyclic compound /  ionotropic glutamate receptor complex / asymmetric synapse / G-protein alpha-subunit binding / regulation of receptor recycling / neuronal action potential / ionotropic glutamate receptor complex / asymmetric synapse / G-protein alpha-subunit binding / regulation of receptor recycling / neuronal action potential /  voltage-gated calcium channel activity / Unblocking of NMDA receptors, glutamate binding and activation / regulation of postsynaptic membrane potential / voltage-gated calcium channel activity / Unblocking of NMDA receptors, glutamate binding and activation / regulation of postsynaptic membrane potential /  glutamate receptor binding / glutamate receptor binding /  postsynaptic density, intracellular component / postsynaptic density, intracellular component /  protein targeting / positive regulation of synaptic transmission / protein targeting / positive regulation of synaptic transmission /  long-term memory / response to electrical stimulus / presynaptic active zone membrane / glutamate-gated receptor activity / response to fungicide / somatodendritic compartment / cellular response to brain-derived neurotrophic factor stimulus / long-term memory / response to electrical stimulus / presynaptic active zone membrane / glutamate-gated receptor activity / response to fungicide / somatodendritic compartment / cellular response to brain-derived neurotrophic factor stimulus /  synapse assembly / dendrite membrane / ligand-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential / synapse assembly / dendrite membrane / ligand-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential /  ionotropic glutamate receptor binding / positive regulation of synaptic transmission, glutamatergic / dendritic shaft / response to cocaine / ionotropic glutamate receptor binding / positive regulation of synaptic transmission, glutamatergic / dendritic shaft / response to cocaine /  synaptic membrane / synaptic membrane /  synaptic transmission, glutamatergic / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential / synaptic transmission, glutamatergic / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential /  PDZ domain binding / long-term synaptic potentiation / cellular response to amino acid stimulus / postsynaptic density membrane / Schaffer collateral - CA1 synapse / PDZ domain binding / long-term synaptic potentiation / cellular response to amino acid stimulus / postsynaptic density membrane / Schaffer collateral - CA1 synapse /  regulation of synaptic plasticity / modulation of chemical synaptic transmission / regulation of synaptic plasticity / modulation of chemical synaptic transmission /  neuromuscular junction / neuromuscular junction /  protein localization / protein localization /  receptor internalization / response to organic cyclic compound / recycling endosome / response to toxic substance / cerebral cortex development / synaptic vesicle membrane / cellular response to growth factor stimulus / receptor internalization / response to organic cyclic compound / recycling endosome / response to toxic substance / cerebral cortex development / synaptic vesicle membrane / cellular response to growth factor stimulus /  small GTPase binding / response to peptide hormone / recycling endosome membrane / G-protein beta-subunit binding / cell-cell junction / small GTPase binding / response to peptide hormone / recycling endosome membrane / G-protein beta-subunit binding / cell-cell junction /  synaptic vesicle / presynapse / response to estradiol / synaptic vesicle / presynapse / response to estradiol /  presynaptic membrane / presynaptic membrane /  amyloid-beta binding / amyloid-beta binding /  cell body / early endosome membrane / cell body / early endosome membrane /  scaffold protein binding / postsynapse / chemical synaptic transmission / scaffold protein binding / postsynapse / chemical synaptic transmission /  postsynaptic membrane postsynaptic membraneSimilarity search - Function | |||||||||
| Biological species |   Rattus norvegicus (Norway rat) Rattus norvegicus (Norway rat) | |||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.9 Å cryo EM / Resolution: 2.9 Å | |||||||||
 Authors Authors | Zhang, D. / Ivica, J. / Krieger, J.M. / Ho, H. / Yamashita, K. / Cais, O. / Greger, I. | |||||||||
| Funding support |  United Kingdom, 2items United Kingdom, 2items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: Structural mobility tunes signalling of the GluA1 AMPA glutamate receptor. Authors: Danyang Zhang / Josip Ivica / James M Krieger / Hinze Ho / Keitaro Yamashita / Imogen Stockwell / Rozbeh Baradaran / Ondrej Cais / Ingo H Greger /   Abstract: AMPA glutamate receptors (AMPARs), the primary mediators of excitatory neurotransmission in the brain, are either GluA2 subunit-containing and thus Ca-impermeable, or GluA2-lacking and Ca-permeable. ...AMPA glutamate receptors (AMPARs), the primary mediators of excitatory neurotransmission in the brain, are either GluA2 subunit-containing and thus Ca-impermeable, or GluA2-lacking and Ca-permeable. Despite their prominent expression throughout interneurons and glia, their role in long-term potentiation and their involvement in a range of neuropathologies, structural information for GluA2-lacking receptors is currently absent. Here we determine and characterize cryo-electron microscopy structures of the GluA1 homotetramer, fully occupied with TARPγ3 auxiliary subunits (GluA1/γ3). The gating core of both resting and open-state GluA1/γ3 closely resembles GluA2-containing receptors. However, the sequence-diverse N-terminal domains (NTDs) give rise to a highly mobile assembly, enabling domain swapping and subunit re-alignments in the ligand-binding domain tier that are pronounced in desensitized states. These transitions underlie the unique kinetic properties of GluA1. A GluA2 mutant (F231A) increasing NTD dynamics phenocopies this behaviour, and exhibits reduced synaptic responses, reflecting the anchoring function of the AMPAR NTD at the synapse. Together, this work underscores how the subunit-diverse NTDs determine subunit arrangement, gating properties and ultimately synaptic signalling efficiency among AMPAR subtypes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8c1p.cif.gz 8c1p.cif.gz | 504 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8c1p.ent.gz pdb8c1p.ent.gz | 377.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8c1p.json.gz 8c1p.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/c1/8c1p https://data.pdbj.org/pub/pdb/validation_reports/c1/8c1p ftp://data.pdbj.org/pub/pdb/validation_reports/c1/8c1p ftp://data.pdbj.org/pub/pdb/validation_reports/c1/8c1p | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  16379MC  8c1qC  8c1rC  8c1sC  8c2hC  8c2iC  8p3qC  8p3sC  8p3tC  8p3uC  8p3vC  8p3wC  8p3xC  8p3yC  8p3zC  8pivC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments: Refine code: 1
|
 Movie
Movie Controller
Controller















 PDBj
PDBj












