[English] 日本語
 Yorodumi
Yorodumi- PDB-6vz8: Arabidopsis thaliana acetohydroxyacid synthase complex with valin... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6vz8 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Arabidopsis thaliana acetohydroxyacid synthase complex with valine bound | |||||||||
 Components Components | (Acetolactate ...) x 2 | |||||||||
 Keywords Keywords |  PLANT PROTEIN / PLANT PROTEIN /  Complex Complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationacetolactate synthase regulator activity /  acetolactate synthase activity / acetolactate synthase activity /  acetolactate synthase / acetolactate synthase /  regulation of catalytic activity / valine biosynthetic process / isoleucine biosynthetic process / response to herbicide / regulation of catalytic activity / valine biosynthetic process / isoleucine biosynthetic process / response to herbicide /  thiamine pyrophosphate binding / thiamine pyrophosphate binding /  chloroplast stroma / chloroplast stroma /  chloroplast ...acetolactate synthase regulator activity / chloroplast ...acetolactate synthase regulator activity /  acetolactate synthase activity / acetolactate synthase activity /  acetolactate synthase / acetolactate synthase /  regulation of catalytic activity / valine biosynthetic process / isoleucine biosynthetic process / response to herbicide / regulation of catalytic activity / valine biosynthetic process / isoleucine biosynthetic process / response to herbicide /  thiamine pyrophosphate binding / thiamine pyrophosphate binding /  chloroplast stroma / chloroplast stroma /  chloroplast / chloroplast /  peroxisome / peroxisome /  : / : /  flavin adenine dinucleotide binding / response to hypoxia / magnesium ion binding flavin adenine dinucleotide binding / response to hypoxia / magnesium ion bindingSimilarity search - Function | |||||||||
| Biological species |   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) | |||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.45 Å cryo EM / Resolution: 3.45 Å | |||||||||
 Authors Authors | Guddat, L.W. / Low, Y.S. | |||||||||
| Funding support |  Australia, 2items Australia, 2items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2020 Journal: Nature / Year: 2020Title: Structures of fungal and plant acetohydroxyacid synthases. Authors: Thierry Lonhienne / Yu Shang Low / Mario D Garcia / Tristan Croll / Yan Gao / Quan Wang / Lou Brillault / Craig M Williams / James A Fraser / Ross P McGeary / Nicholas P West / Michael J ...Authors: Thierry Lonhienne / Yu Shang Low / Mario D Garcia / Tristan Croll / Yan Gao / Quan Wang / Lou Brillault / Craig M Williams / James A Fraser / Ross P McGeary / Nicholas P West / Michael J Landsberg / Zihe Rao / Gerhard Schenk / Luke W Guddat /    Abstract: Acetohydroxyacid synthase (AHAS), also known as acetolactate synthase, is a flavin adenine dinucleotide-, thiamine diphosphate- and magnesium-dependent enzyme that catalyses the first step in the ...Acetohydroxyacid synthase (AHAS), also known as acetolactate synthase, is a flavin adenine dinucleotide-, thiamine diphosphate- and magnesium-dependent enzyme that catalyses the first step in the biosynthesis of branched-chain amino acids. It is the target for more than 50 commercial herbicides. AHAS requires both catalytic and regulatory subunits for maximal activity and functionality. Here we describe structures of the hexadecameric AHAS complexes of Saccharomyces cerevisiae and dodecameric AHAS complexes of Arabidopsis thaliana. We found that the regulatory subunits of these AHAS complexes form a core to which the catalytic subunit dimers are attached, adopting the shape of a Maltese cross. The structures show how the catalytic and regulatory subunits communicate with each other to provide a pathway for activation and for feedback inhibition by branched-chain amino acids. We also show that the AHAS complex of Mycobacterium tuberculosis adopts a similar structure, thus demonstrating that the overall AHAS architecture is conserved across kingdoms. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6vz8.cif.gz 6vz8.cif.gz | 977.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6vz8.ent.gz pdb6vz8.ent.gz | 778.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6vz8.json.gz 6vz8.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/vz/6vz8 https://data.pdbj.org/pub/pdb/validation_reports/vz/6vz8 ftp://data.pdbj.org/pub/pdb/validation_reports/vz/6vz8 ftp://data.pdbj.org/pub/pdb/validation_reports/vz/6vz8 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  21487MC  6u9dC  6u9hC  6wo1C C: citing same article ( M: map data used to model this data |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Acetolactate ... , 2 types, 16 molecules DEHILMPQFGJKNORS
| #1: Protein |  / AtALS / Acetohydroxy-acid synthase / Protein CHLORSULFURON RESISTANT 1 / AtALS / Acetohydroxy-acid synthase / Protein CHLORSULFURON RESISTANT 1Mass: 64025.203 Da / Num. of mol.: 8 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Arabidopsis thaliana (thale cress) / Gene: ALS, AHAS, CSR1, TZP5, At3g48560, T8P19.70 / Production host: Arabidopsis thaliana (thale cress) / Gene: ALS, AHAS, CSR1, TZP5, At3g48560, T8P19.70 / Production host:   Escherichia coli (E. coli) / References: UniProt: P17597, Escherichia coli (E. coli) / References: UniProt: P17597,  acetolactate synthase acetolactate synthase#2: Protein |  / Acetohydroxy-acid synthase small subunit / ALS / Acetohydroxy-acid synthase small subunit / ALSMass: 53948.906 Da / Num. of mol.: 8 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Arabidopsis thaliana (thale cress) / Gene: At2g31810, F20M17.15 / Production host: Arabidopsis thaliana (thale cress) / Gene: At2g31810, F20M17.15 / Production host:   Escherichia coli (E. coli) / References: UniProt: Q93YZ7 Escherichia coli (E. coli) / References: UniProt: Q93YZ7 |
|---|
-Non-polymers , 4 types, 32 molecules 


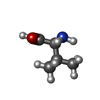



| #3: Chemical | ChemComp-MG / #4: Chemical | ChemComp-FAD /  Flavin adenine dinucleotide Flavin adenine dinucleotide#5: Chemical | ChemComp-TPP /  Thiamine pyrophosphate Thiamine pyrophosphate#6: Chemical | ChemComp-VAL /  Valine Valine |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Acetohydroxyacid synthase quaternary complex with valine bound Type: COMPLEX / Details: Recombinant expression from E. coli. / Entity ID: #1-#2 / Source: RECOMBINANT |
|---|---|
| Molecular weight | Value: 0.72 MDa / Experimental value: NO |
| Source (natural) | Organism:   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) |
| Source (recombinant) | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Buffer solution | pH: 7.5 |
| Specimen | Conc.: 0.5 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES / Details: Glutaraldehyde cross linked : YES / Details: Glutaraldehyde cross linked |
| Specimen support | Grid material: COPPER / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R1.2/1.3 |
Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy Bright-field microscopy |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 3.2 sec. / Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) / Num. of grids imaged: 3 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.17.1_3660: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| Symmetry | Point symmetry : C4 (4 fold cyclic : C4 (4 fold cyclic ) ) | ||||||||||||||||||||||||
3D reconstruction | Resolution: 3.45 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 290516 / Symmetry type: POINT | ||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL | ||||||||||||||||||||||||
| Atomic model building |
| ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller







 PDBj
PDBj










